2.5.0
Ventral Vagal Complex (VVC)
This chapter examines the polyvagal hypothesis of a Ventral Vagal Complex (VVC) that provides anatomical support for the Social Engagement System. We compare this to the current scientific description of the Nucleus Ambiguus (NA) and the External Formation (NAext).
The term Ventral Vagal Complex, invented by Porges, is not used in the current literature, except by polyvagal authors. Here we show how the PVT has created a fictional narrative around the ventral parasympathetic branch with unrealistic magical properties.
2.5.1
Ventral Vagal Complex (VVC) : a polyvagal perspective
This chapter compares the Ventral Vagal Complex (VVC) – the brain stem anatomical support of the Social Engagement System – and the Nucleus Ambiguus, described by current anatomists and physiologists. The term Ventral Vagal complex forged by Porges is not used in the current literature – aside from polyvagal engaged authors.
In Love: An Emergent Property of the Mammalian Autonomic Nervous System (1998) Porges shapes the contours of his new creation: the Ventral Vagal Complex. “The VVC represents the phylogenetically most recent stage and is unique to mammals (…) it consists of the myelinated vagus and portions of other cranial nerves (i.e. V, VII, IX, XI) that regulate structures derived from the embryonic branchial arches (i.e. ancient gill arches). Collectively, these neural pathways regulate the branchiomeric muscles and have been categorized as special visceral efferent fibers. In mammals the branchiomeric muscles control facial expression, sucking, swallowing, breathing, listening and vocalization (…) The VVC fosters the development of complex social behaviors.”
The article uses "primitive" fourteen times and "old" eleven times to describe the "vestigial" organization of non-mammalian species. Porges unfolded his design apodictically. His construction is simple, relying only on the branchiomotor nerves, supplemented by the myelinated parasympathetic nerve.
As the following figure shows, the VVC includes the four branchiomotor (pharyngeomotor) nerves that innervate the pharyngeal arches (I, II, III, IV, VI). The CN XI (accessory nerve), which has a dual origin — medulla and cervical cord — has been added to the group. The ventral myelinated parasympathetic branch innervates the bronchi and the heart.
Neither oculomotor nerves (III, IV, VI), tongue (XII), sympathetic, or respiratory centers are part of the plan. In his scheme, a simple rectangle represents the cortex. The hypothalamus and the PAG (midbrain) are also missing.
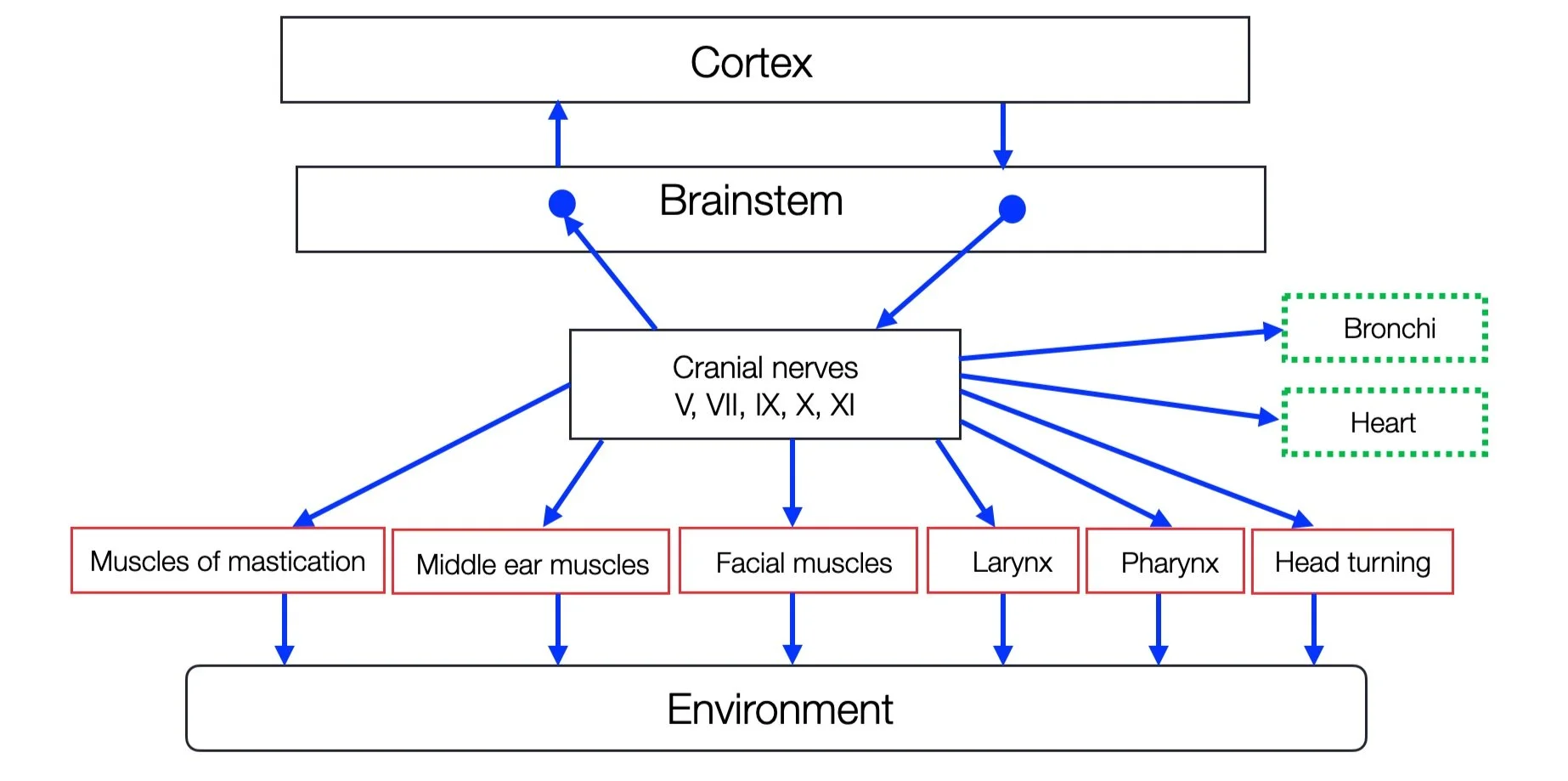
The Social Engagement System according to Porges. Our modified version shows somatomotor components in red and visceromotor components in green (adapted from Porges, 2012 by ©absb, 2024).
In Polyvagal Safety (2021, p. 24), Porges describes how the source nuclei of the cranial nerves IX, X, and XI “migrate to form the nucleus ambiguus (a portion of the vagal preganglionic neurons that forms the nucleus ambiguus in mammals and regulate striated muscles including the larynx and pharynx….)” an evolutionary progression. The whole paragraph is a nightmare of logical reasoning because the author confuses the somatic (NA proper) and visceral (NAext) parts of the nucleus ambiguus. The branchiomotor nerves are not preganglionic neurons. Parasympathetic neurons (with the source in the external formation) are. Moreover, the arrangement of branchiomotor nerves within the pharyngeal apparatus is very ancient (identical or homologous). Branchial motor nerves in cartilaginous (e.g., sharks) and bony fishes were present. This is a typical example of the confusing ambiguity we regularly encounter in the polyvagal discourse about the nucleus ambiguus (see the chapter on fallacies).
2.5.2
Nucleus Ambiguus
Like most nuclei in the lower brainstem, the nucleus ambiguus (NA) is a bilateral log-shaped structure, with, above, the nucleus of the CN XII, the DVMN, and the NTS above and the respiratory centers below.
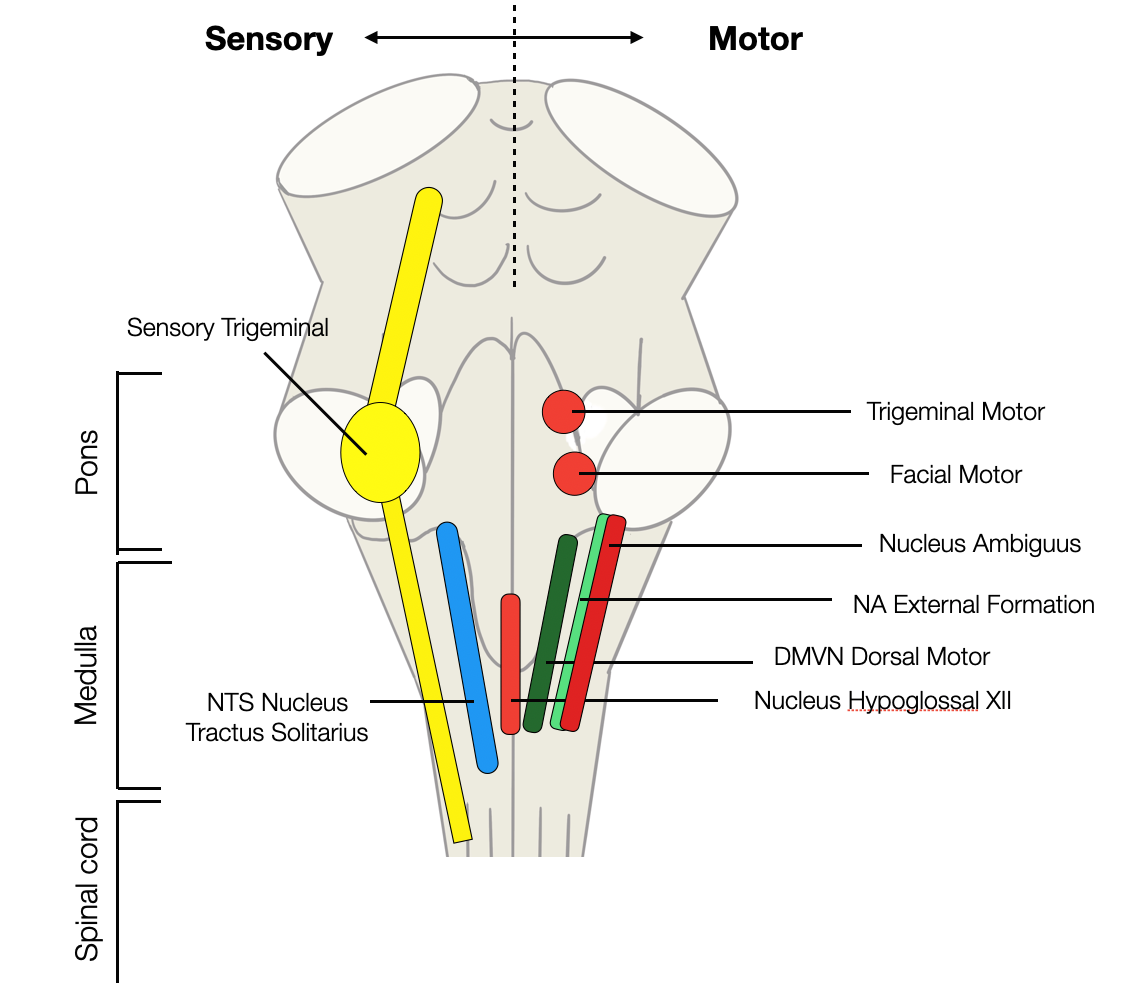
Sensory and motor nuclei in the lower part of the brainstem (rhombencephalon). The nucleus ambiguus is a double column (red=branchiomotor, green=parasympathetic) (©absb, 2024).
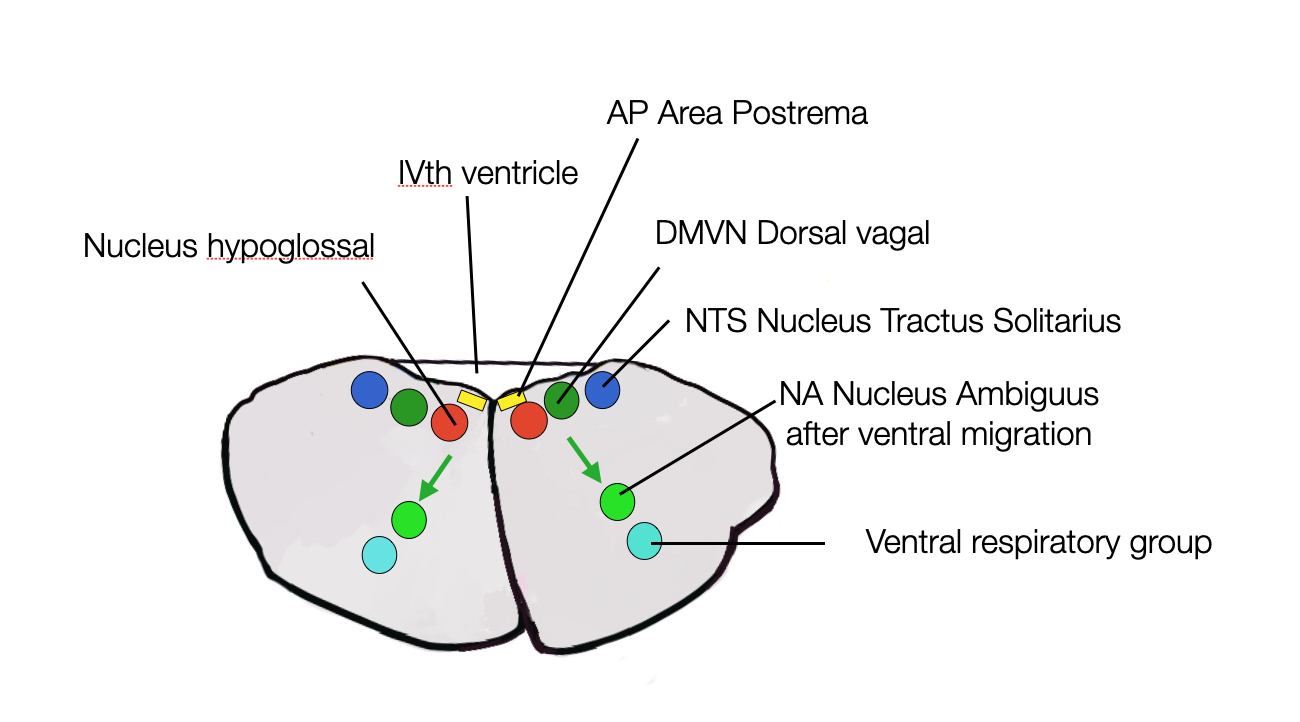
This diagram shows the superposition of nuclei in the spinal cord. This organization results from the early migration of neurons from the wall of the neural tube to a ventral location, just above the ventral respiratory group (©absb, 2024).
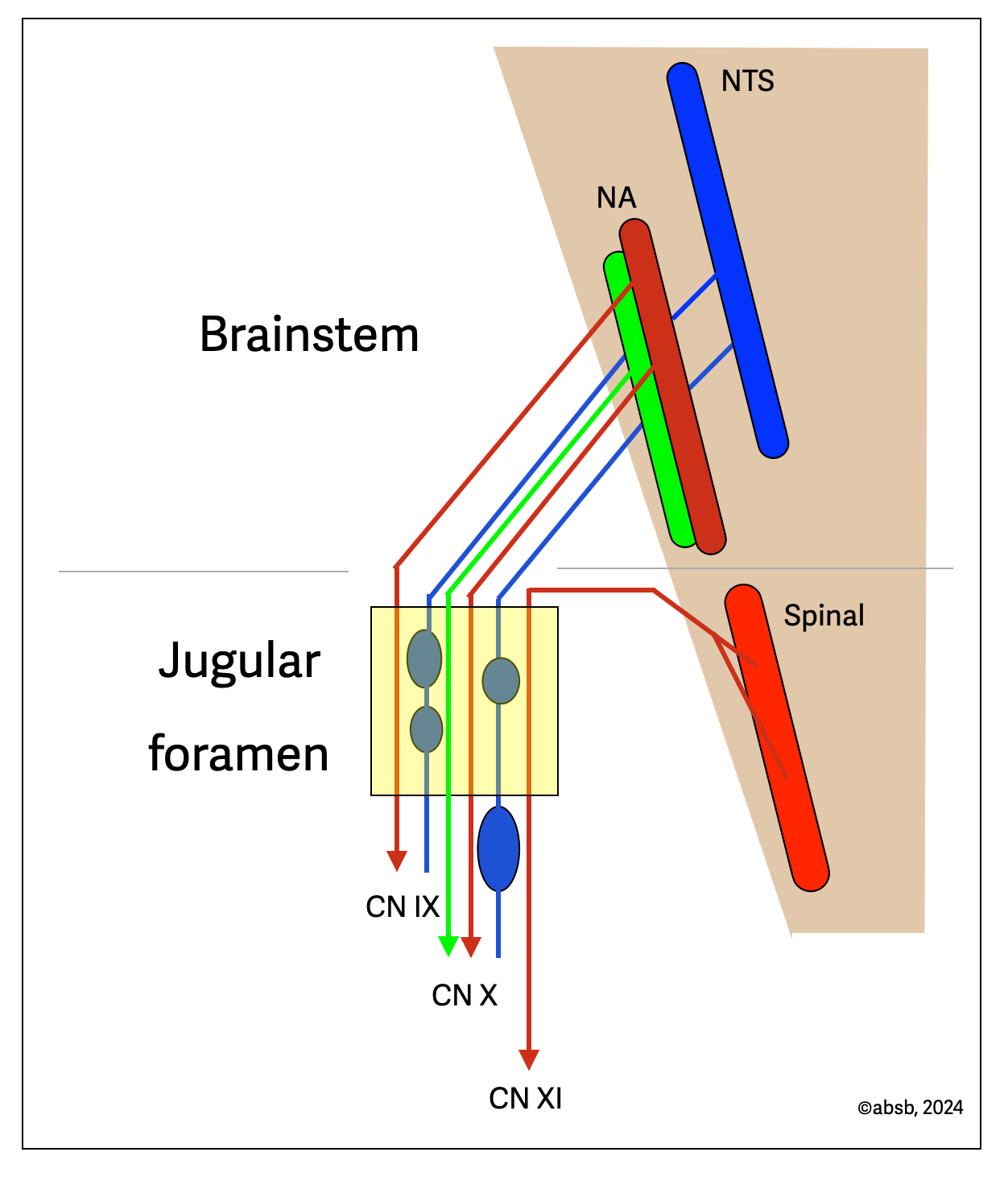
The nucleus ambiguus (NA) is the origin of three different cranial nerves: CN IX (glossopharyngeal), CN X (vagal), and CN XI (accessory). However, the combined diagram shows that most accessory roots originate in the spinal cord. The three nerves exit the skull together through the same opening, the jugular foramen. CN IX and X have both efferent motor and afferent sensory fibers. CN X contains parasympathetic fibers (green) that originate in the ventral part of the nucleus ambiguus (NAext). As we will see in the next section, the nucleus ambiguus has two regions:
a dorsal region that innervates striated (voluntary) muscles for swallowing and vocalization
a ventral parasympathetic region called the external formation (NAext).
The NTS (nucleus of the solitary tract) is a significant component of the system, as it receives afferent sensory fibers from the CN IX and CN X.
Source: Clinical Neuroanatomy and Neuroscience (Mtui, 2021, p. 216).
In 1987, Bieger and Hopkins published a seminal and comprehensive article on the nucleus ambiguus (NA), a pivotal moment in our research when we first read it. Viscerotopic representation of the upper digestive tract in the medulla oblongata of the rat: The Nucleus Ambiguus is probably the best description of the NA you can find — except for very recent studies based on optogenetics.
The human ventral parasympathetic branch of the vagal nerve originates in the nucleus ambiguus, a collection of neurons located ventrally in the lower part of the spinal cord. As the name “ambiguus” implies, the NA is not a precise structure. Besides the ventral part of the Xth nerve, two other cranial nerves (the IXth and the medullary part of the XIth) originate from it. Bieger and Hopkins (Bieger, 1987) explain in their article that “the dorsal division corresponds to the nucleus ambiguus in the narrow sense and comprises a rostral esophagomotor compact formation, an intermediate pharyngolaryngomotor semicompact formation, and a caudal laryngomotor loose formation … The ventral division of the nucleus ambiguus corresponds to the external formation. It extends along the entire length of the medulla oblongata and contains preganglionic neurons innervating the heart and supra diaphragmatic structures innervated by the glossopharyngeal and the superior laryngeal nerves” (out of the abstract, bold characters are ours).
Nieuwenhuys, Voogd, and van Huizen reproduced the main diagram in The Human Central Nervous System (fourth edition, 2008), one of the best handbooks about neuroanatomy. Recently, Molecularly defined circuits for cardiovascular and cardiopulmonary control (Veerakumar, 2022), a study using optogenetics, confirmed those observations with even more certainty.
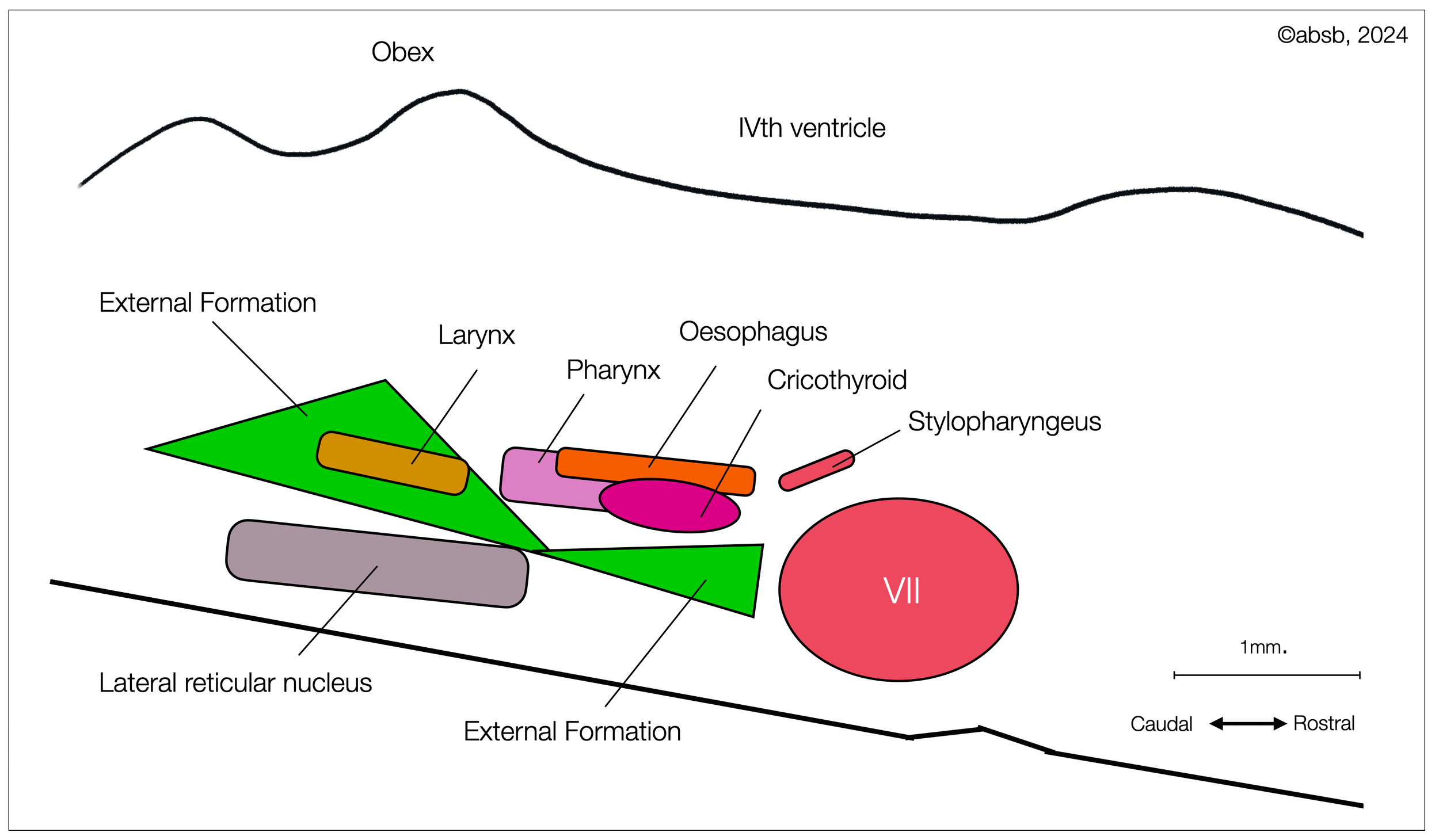
Schematic representation of the viscerotopy of the ambiguus nucleus of a rat, based on the diagram of Bieger and Hopkins (1987), as redrawn by Nieuwenhuis (2008, p. 868). Our diagram adopts the general elements of Bieger and Hopkins, with new colors and simplified shapes. We encourage you to consult the original article and book (see above). This diagram shows the duality of the NA (branchiomotor vs. parasympathetic external formation in green)(©absb, 2024).
If we invert the schematic diagram (originally in Bieger, 1987) and rotate it to a half vertical position, we can better understand how it would look in a human brain. This is also more consistent with other illustrations on this website (head profile looking to the left, brainstem in half-upright position).
The orange rectangle shows the location of the nucleus ambiguus (NA + NAext) and some adjacent structures.
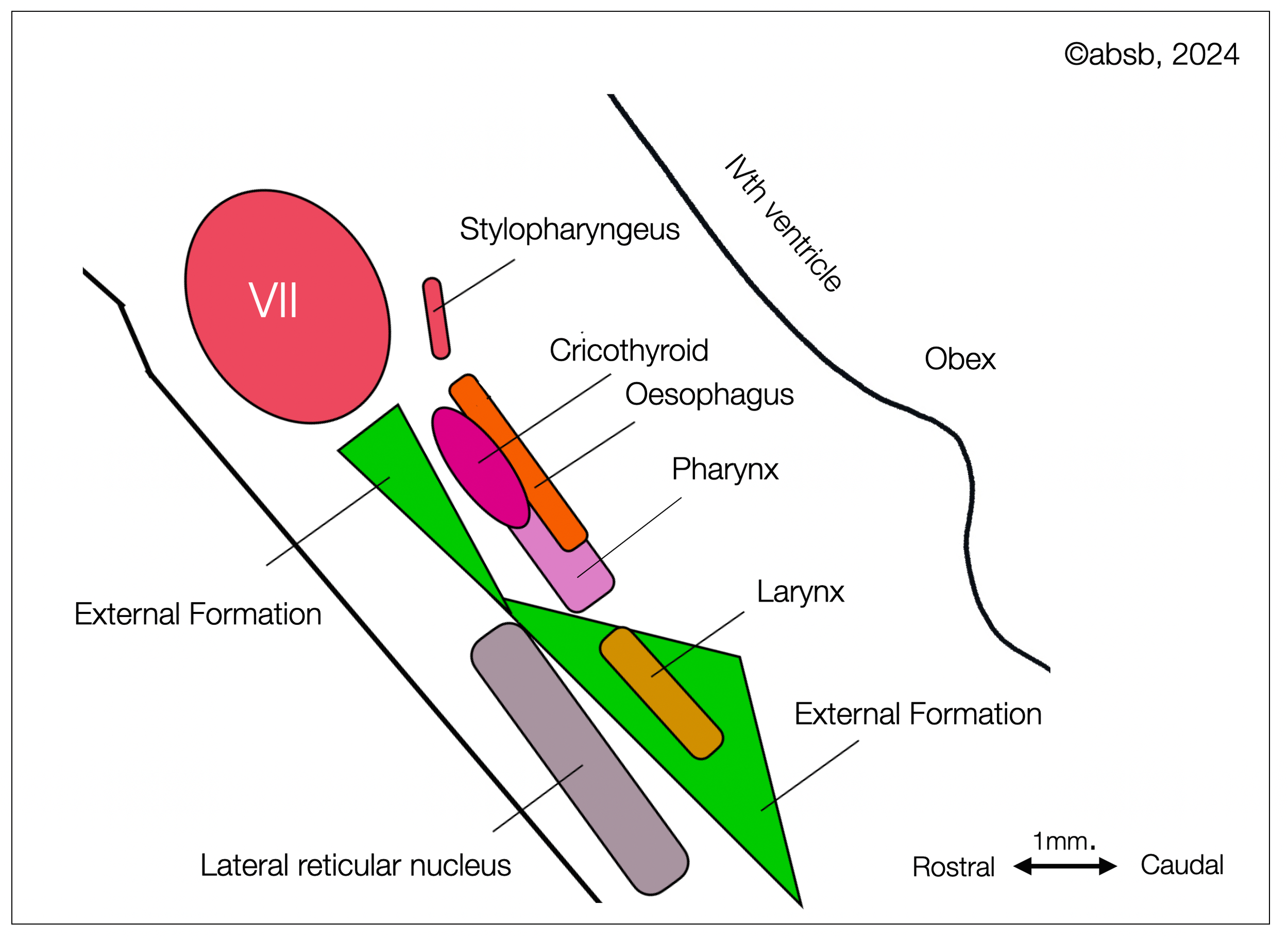
Inverted and rotated replicate of the Bieger-Hopkins diagram.
1. The dorsal part allows voluntary actions such as swallowing and speaking. It contains the nuclei of three nerves: CN IX, X, and XI. The first two divisions are branchiomotor (pharyngomotor), i.e., related to their phylogenetic and embryological development, as they innervate the sixth branchial arch. This part resembles the NA in other vertebrates (Birinyi, 2011) (Matesz, 1996). The third nucleus is the cranial, part of CN XI, which appears in amniotes.
The cranial nerves have two main functions:
Feeding (locating, catching, ingesting, tasting, and swallowing)
Coordination of swallow-breathing and vocalization (in land animals)
When leaving the sea for the land, the intestines were relocated and oxygen was extracted from the gills to the lungs. In the same process, the muscles involved in breathing moved from the neck to the thorax. We don't know how and when the NA transformation occurred (perhaps 100 mya or more). Because the cardiac and cardiopulmonary neurons are now just below the branchiomotor parts of the NA that innervate the pharynx (swallowing) and larynx (phonation, vocalization), they are also just above the ventral respiratory centers. This arrangement may help to coordinate breathing and swallowing better. But let's not forget that the central coordinator is the NTS in the dorsal region, not the “external formation.”
2. The ventral part of the nucleus ambiguus or external formation (NAext) is the visceral part of the nucleus ambiguus.
As Bieger and Hopkins describe: “The external formation of the nucleus ambiguus differs from the dorsal division, i.e., the nucleus ambiguus in the narrow sense, in that the former does not innervate special visceral structures, i.e., striated musculature of the pharynx, larynx, and esophagus. Thus, they are most likely general visceral efferent motoneurons and, in the aggregate, would constitute the general visceral efferent column of the nucleus ambiguus.”
We must remember that although modern anatomy and neurophysiology books show nuclei as compact, colorful structures, this is not reality. The brain is dull beige until it is stained with fluorescent dyes, and the boundaries between different nuclei are blurred.
2.5.3
Functionality and Interactions
The dorsal and ventral parts of the NA may be close, but that doesn't mean they are functionally related. They are genetically, biologically, and physiologically distinct. The presence of cardiovagal (CVN) and pulmonary (PVN) neurons results in a progressive migration of these neurons from a more dorsal position (in the dorsal vagal motor nucleus or DMVN) to a ventral position. This migration is incomplete in most mammals, as we still find some cardiac vagal neurons (CVN) in the DVMN or the “intermediate zone” between the NA and the DMVN. While the PVT suggests that the reason for this migration is an evolutionary agenda towards socialization, we can think that the presence of cardiovagal and pulmonary vagal neurons in the NAext allows them to better cooperate with the respiratory centers.
In his reply to Grossman, Porges argues that the novelty of the “smart vagus” is more than neuronal migration: the myelination of the ventral parasympathetic fibers is essential. This is mainly true, but not entirely. We find a myelinated parasympathetic vagus in elasmobranchs (e.g., sharks) and certain reptiles. A myelinated dorsal vagal nerve has been described in sheep (Booth, 2021).
Dividing the NA into two parts allows the orthovagal model (see below) to draw a different schema. The “branchial” or “pharyngeal apparatus” allows humans sophisticated communication but was initially designed for catching, biting, tasting, and swallowing. Food was undoubtedly the first function. When the first fishes developed a jaw (gnathostomes), they made a significant leap from passive to active feeding. They became predators.
The NA's ventral “external formation” (NAext) is part of the visceral system and forms a sophisticated interconnected network with the respiratory groups, the DMVN, and the NTS. The purpose of migrating neurons within the hindbrain may fundamentally differ from the polyvagal proposal. Isn't the proximity of the underlying respiratory centers (pre-Bötzinger and Bötzinger), which generate the respiratory rhythm, a better explanation for this migration? To our knowledge, no direct connection of the NAext to the V, VII, IX, X, and XI has been demonstrated, while the NAext is in constant communication. The efferent parasympathetic system requires ascending afferent information in the NTS, which distributes inputs to the NA, DVMN, and several other structures (e.g., the hypothalamus and sympathetic relays). In addition, these multiple circuits involve local neurohormonal players (endorphins, endocannabinoids, serotoninergic and adrenergic sympathetic transmitters).
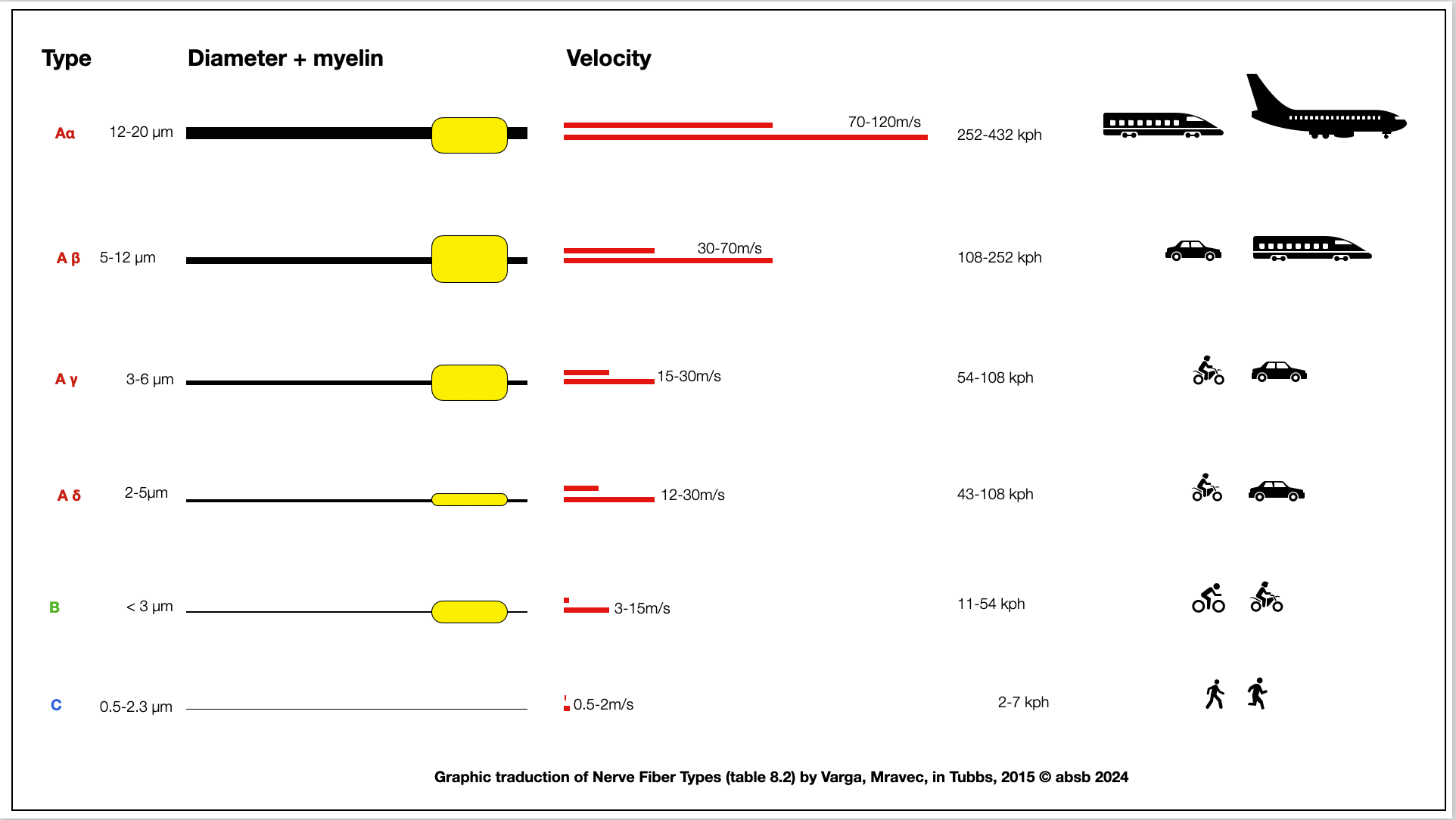
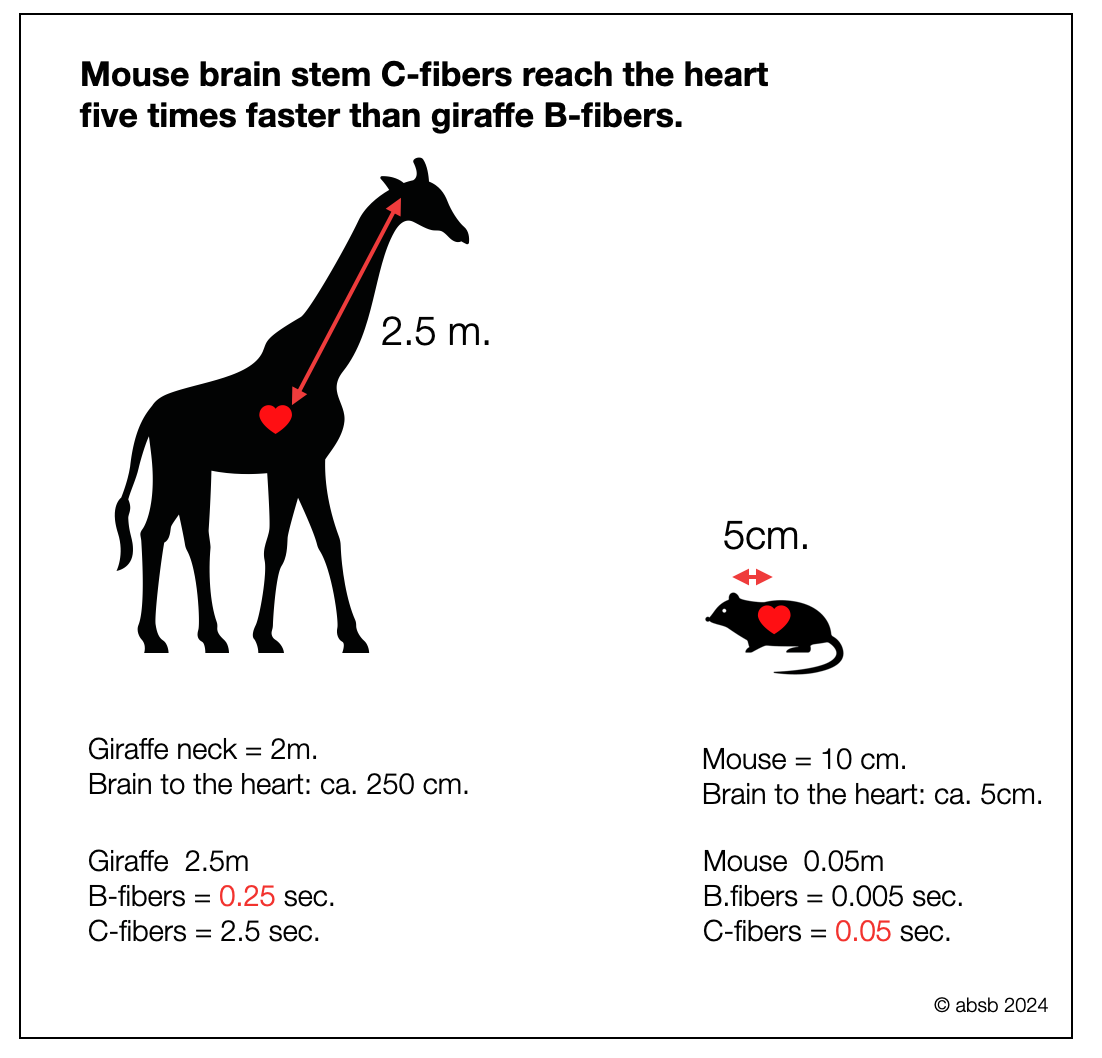
How much faster is the ventral parasympathetic?
The Polyvagal Theory (PVT) suggests that the myelinated B-fibers of the ventral vagal branch (NAext) provide a significant advantage over the non-myelinated C-fibers of the dorsal vagal branch (DVMN). At first glance, this seems straightforward, but a closer look challenges this assumption. The first diagram illustrates the speed of different nerve fibers and compares them to real-world objects in motion, like walking humans, bicycles, motorcycles, cars, trains, and airplanes.
Comparing C-fibers to B-fibers is like comparing a man on foot to one on a bicycle or a slow motorcycle. Cardiac parasympathetic function, however, primarily modulates the heart's intrinsic rhythm rather than requiring split-second reaction times, as seen in the somatic motor system for urgent actions like escaping predators or executing attacks.
The second diagram highlights another factor—the pathway length. It compares the time it takes for a signal to travel from the brainstem to the heart in a giraffe (via B-fibers) with that in a mouse (via C-fibers). Despite the myelination advantage, the signal in the mouse reaches its heart five times faster than in the giraffe. The results would be similar if we replaced the mouse with a lizard.
Therefore, we argue that myelination in the ventral parasympathetic pathway, while facilitating fine-tuning of vagal tone and conserving energy, does not provide the critical advantage suggested by PVT. It supports energy-efficient, rhythmic modulation (respiratory and baroreceptor input), which matches the energetic strategy of high-energy mammals (e.g., lions and gazelles).
2.5.4
The Orthovagal Model
In light of the scientific knowledge available today — and each year brings new astonishing discoveries — we no longer agree with a truncated representation of the brain stem. The mammalian ANS does not “retain vestiges of phylogenetically older autonomic nervous systems."(Porges, 2001, p. 126).
These systems (e.g., SNS, NTS, and DMVN) fit perfectly into the complexity of the brainstem as full partners. Therefore, we oppose an “orthovagal” model of the brain stem to the polyvagal model. “Orthovagal,” as in polyvagal, means “correct” model of the vagal nerve (Greek: ορθο ortho = correct, as in orthography, orthopedic, or orthostatic). Beyond a simple discussion of anatomical structures, this distinction will have implications in several areas: immunity, metabolism, psychology, and more.
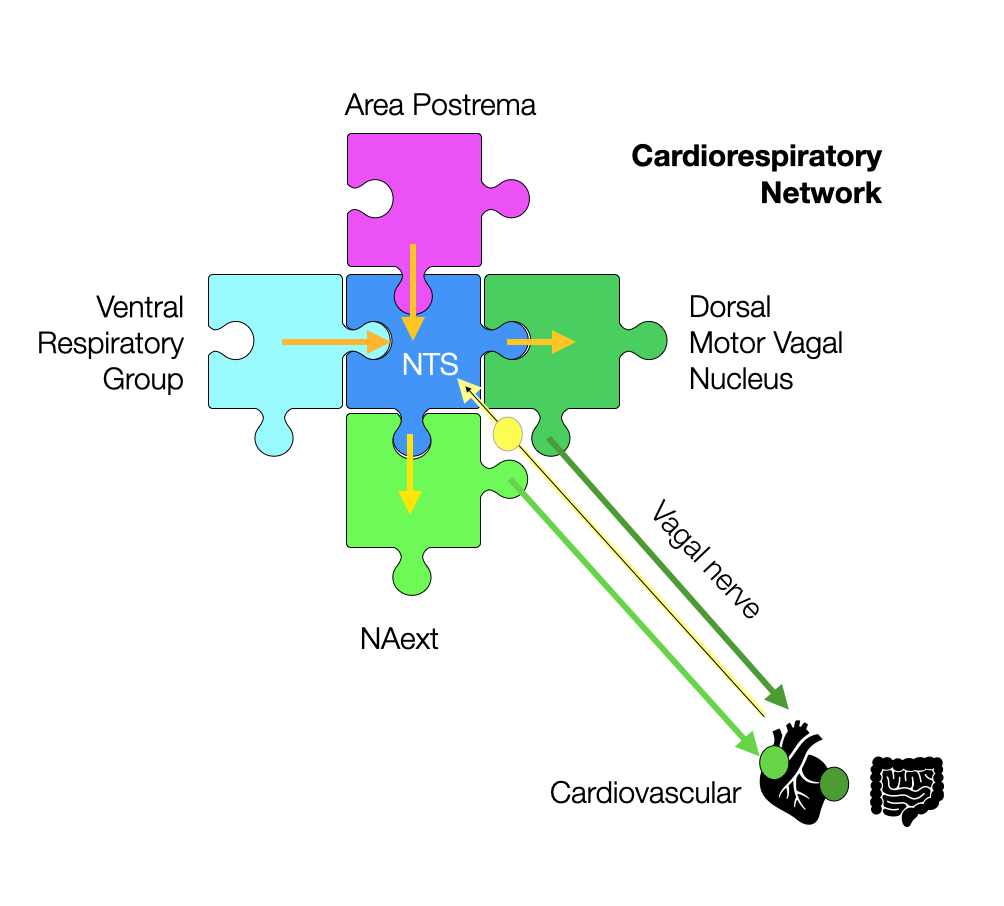
In this simplified model, the NTS fits perfectly as the central piece of the cardiorespiratory puzzle. The primary function of the external formation (NAext) is to slow the intrinsic rhythm of the heart, creating heart rate variability (HRV) and bradycardia (e.g., dive reflex). The NTS is the visceral coordinator — not the nucleus ambiguus.
2.5.5
Ventral vagal induced bradycardia
cVPN = cardiac vagal preganglionic neurons (most of them in the NAext)
VPN = vagal preganglionic neurons (relating to lungs and digestive functions)
In the opening pages of Orienting in a Defensive World (1995), Porges clarifies the existence of two parasympathetic branches. The ventral branch produces heart rate variability (HRV), while the dorsal branch is responsible for neurogenic bradycardia — which can cause death in mammals. Twenty years later, more and more studies challenge this claim. The ventral parasympathetic branch, originating in the NAext, produces bradycardia. Moreover, most studies show that the dorsal branch has no (or weak) influence on the heartbeat (chronotropic).
According to CARDIAC VAGAL PREGANGLIONIC NEURONS: AN UPDATE. (Gourine, 2016), cVPNs of the NA have rhythmic, respiration-related discharge patterns, with B-fiber axons innervating the cardiac sinus (McAllen et al., 1976; McAllen et al., 1978a; Ciriello et al., 1982). cVPNs contain the postganglionic neurons that innervate the heart. The richest afferent input to the NA comes from the nucleus tractus solitarius (NTS), although it also receives input from other brain areas. There is also evidence in some species that there are cVPNs with C-fiber axons in the NA (Jordan et al., 1986).
Electrophysiological studies have shown that these VPNs affecting heart rate are localized in the ventrolateral part of the nucleus ambiguus, although some may be more widely distributed (McAllen et al., 1976; 1978b). These represent a subpopulation of NA neurons forming the vagus nerve's cardiac and pulmonary branches. In their electrophysiological description of cVPNs, McAllen & Spyer (McAllen et al., 1976; 1978b) showed that NA neurons are either silent or have a meager resting discharge rate under anesthesia. However, their discharge at rest or when facilitated by glutamate, a typical excitatory neurotransmitter, is rhythmic.
Two primary rhythms dominate:
First, cardiac rhythm is driven by glutamatergic projections from the NTS and is abolished by glutamate receptor antagonists. Robust baroreceptor modulation and respiratory-related activity distinguish cVPNs of the NA from cardiac-projecting neurons of the DVMN, which are unaffected by these inputs. The first is correlated with the arterial pulse and depends on inputs from arterial baroreceptors.
The second is related to the central respiratory rhythm (Gilbey et al., 1984).
Activation of arterial chemoreceptors has a strong effect on NA neurons. The direct effect is to excite cVPNs when a brief stimulus is delivered during expiration, but is ineffective when delivered during inspiration. While arterial baroreceptors' inputs largely control these neurons' excitability, their rhythmic pattern of respiratory-related discharge reflects strong excitatory and inhibitory inputs from adjacent respiratory centers (central oscillator). cVPNs receive an excitatory input during postinspiration and a strong inhibitory input during inspiration (Gilbey et al., 1984). These respiratory changes in cVPN excitability explain the “gating” of other excitatory and inhibitory inputs, including inputs from peripheral chemoreceptors.
This respiratory regulation (patterning)n by the cVPNs ensures cardiac output is closely linked to respiratory minute volume (Spyer, 1994). Suppression of respiratory activity, as in the diving reflex, causes cardiac deceleration and a decrease in arterial PO2, while breath-holding causes bradycardia. Conversely, high respiratory drive during exercise increases heart rate. As in meditation and yoga, a long post-inspiratory phase lowers the heart rate by increasing the release of cVPNs (stabilizing or lowering arterial blood pressure).
The prominent physiological role of NA neurons projecting to the sinus and controlling heart rate is well established. Some studies (Blinder et al., 2007; Gatti et al., 1996; Sampaio et al., 2014) have shown that stimulation of NA neurons can alter the rate of atrioventricular conduction (negative dromotropic effect, with a longer P-R interval), independent of changes in heart rate. Evidence from cats and dogs has also shown that activation of NA neurons may have a negative inotropic (decreased force) effect (Blinder, 1998, 2007). DVMN Neurons Control Ventricular Inotropy (Force of Ventricular Contraction)
2.5.6
The Optogenetic Revolution
How Does Optogenetics Reshape our View of the NA?
In the past, anatomists and physiologists had two ways of exploring the brain. The anatomists would dissect the brain with their scalpel and examine tissue probes under the microscope, using diverse dyes. It was possible to let the dye diffuse from the target to the origin of the axon, going upstream and marking the physical path. Physiologists, on their side, would implant tiny electrodes in the nerves or neuronal centers, send an electrical impulse, and observe what was happening. This later process is complicated and lacks precision, with the risk of stimulating surrounding neurons or nerves. Recently, researchers have genetically modified particular neuronal populations to make them produce light-sensitive pigments. You certainly know that the eye cells contain pigments (opsines) that react to the light. Depending on the wavelength of the light source, different cells react, sending an electrical signal to the brain. Researchers can introduce a skinny glass fiber through the brain and send a specific light stream (e.g., red, green, or blue), which activates the genetically modified neurons. It results in an electrical signal from the neuronal body to the receptor (synapse or organ). This process is far more precise and much less intrusive. In addition, animals are awake. They can be active and move around to some extent.
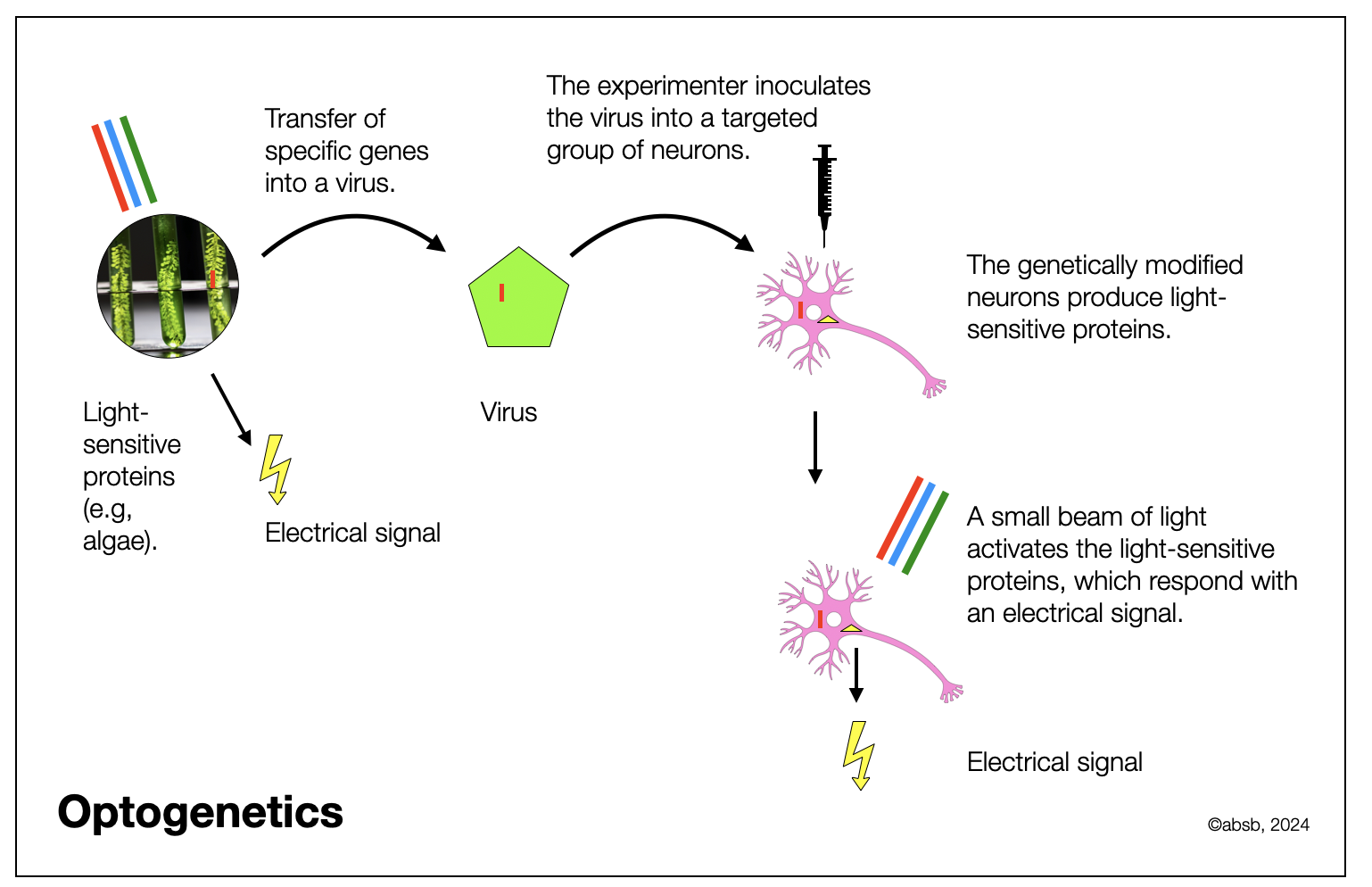
Around the world, teams (Veerakumar, 2022) and (Moreno 2018)—and see below—have been intensively exploring brain neurocircuits. They could confirm different hypotheses, among others, concerning the role of the Nucleus Ambiguus (NAext) in bradycardia, dive reflex, and control of bronchial activity.
Molecularly defined circuits for cardiovascular and cardiopulmonary control (Veerakumar, 2022). While the sympathetic nervous system exerts control of heart rate on a timescale of seconds, the parasympathetic nervous system controls an even faster, beat-to-beat timescale. It is thus thought to be responsible for the tight coordination between cardiac and pulmonary function. Cardiac parasympathetic outflow originates from brain stem preganglionic neurons, primarily in the nucleus ambiguus. The few cardiac parasympathetic neurons located in the DVMN (dorsal vagal motor nucleus) control the ventricular strength (inotropy) and excitability, but not the heart rate (chronotropic). The preganglionic neurons in the synapse on the surface of the heart (ganglia) with postganglionic neurons that innervate:
Sinoatrial (SA) node
Atrioventricular (AV) node
Myocardial tissue (cardiac muscle)
Further reading about Optogenetics
Bringing Light to Transcription: The Optogenetics Repertoire (De Mena, 2018).
Optogenetics (Deisseroth, 2011).
The Development and Application of Optogenetics (Feno, 2011).
Optogenetics in primates: a shining future? (Gerits, 2013).
Light Up the Brain: The Application of Optogenetics in Cell-Type Specific Dissection of Mouse Brain Circuits (Lee, 2020).
Sudden heart rate reduction upon optogenetic release of acetylcholine from cardiac parasympathetic neurons in perfused hearts (Moreno, 2018). Optogenetic activation of intrinsic parasympathetic neurons reduced heart rate in an immediate, dose-dependent manner similar to acetylcholine-induced sinus rate slowing. This is a novel approach to control parasympathetic modulation of cardiac function by selectively activating the endogenous release of acetylcholine from intrinsic cardiac cholinergic neurons (Chen, 2022).
Optophysiology: Illuminating cell physiology with optogenetics (Tan, 2022).
2.5.7
Genetic Dissection
Another new development in research is the ease of access to genetic information in microorganisms, plants, and animals. Recently, the cost and time required for genetic analysis has plummeted. A botanist in the jungle can scan a leaf or flower and get a result almost instantly. Neuroscience is taking advantage of this improvement, treating groups of neurons like a population geneticist studies the distribution of genomes within and between human populations — or viral strains.
Molecularly defined circuits for cardiovascular and cardiopulmonary control (Veerakumar, 2022). While the sympathetic nervous system controls heart rate on a timescale of seconds, the parasympathetic nervous system controls an even faster timescale, beat-to-beat. It is therefore thought to be responsible for the tight coordination between cardiac and pulmonary function. Cardiac parasympathetic outflow originates from preganglionic neurons in the brainstem, primarily in the nucleus ambiguus. The few cardiac parasympathetic neurons in the DVMN (dorsal vagal motor nucleus) control ventricular strength (inotropy) and excitability but not heart rate (chronotropic). The preganglionic neurons that synapse on the surface of the heart (ganglia) with postganglionic neurons that innervate:
Sinoatrial (SA) node
Atrioventricular (AV) node
Myocardial tissue (heart muscle)
In The Mammalian Diving Response: Inroads to Its Neural Control (Panneton, 2020), the authors examine the diving reflex and the genetic changes caused by oxygen deprivation. They cite several recent studies (see below):
Baranova, T. I., Berlo, D. N., Glotov, O. S., Korf, E. A., Minigali, A. D., Mitrofanova, A. V., et al. (2017). Genetic determination of the vascular reactions in humans in response to the diving reflex. Am. J. Physiol. Heart Circ. Physiol. 312, H622–H631.
Fahlman, A., Bostrom, B. L., Dillon, K. H., and Jones, D. R. (2011). The genetic component of the forced diving bradycardia response in mammals. Front. Physiol. 2:63. doi: 10.3389/fphys.2011.00063
Ilardo, M. A., Moltke, I., Korneliussen, T. S., Cheng, J., Stern, A. J., and Racimo, F. (2018). Physiological and genetic adaptations to diving in sea nomads. Cell 173, 569–580.
Using sRNA sequencing, the researchers targeted cardiac neurons in the nucleus ambiguus to exclude laryngeal neurons and found two types of neurons, ACP (cardiopulmonary) and ACV (cardiovascular). Since both populations in the external formation of the NA slow SA (sinus atrial) and AV (atrioventricular) nodes, the baroreflex selectively activates ACV neurons. In contrast, the dive reflex activates ACP neurons.
An increase in blood pressure selectively activates ACV neurons, which project directly to cholinergic cardiac ganglion neurons, which in turn slow SA node rate and AV node conduction velocity.
ACP neurons also project to cholinergic cardiac ganglion neurons that slow the SA and AV nodes. In addition, they activate the diving reflex, which is widely conserved in land vertebrates, causing bronchoconstriction (narrowing of the airways) and bradycardia.
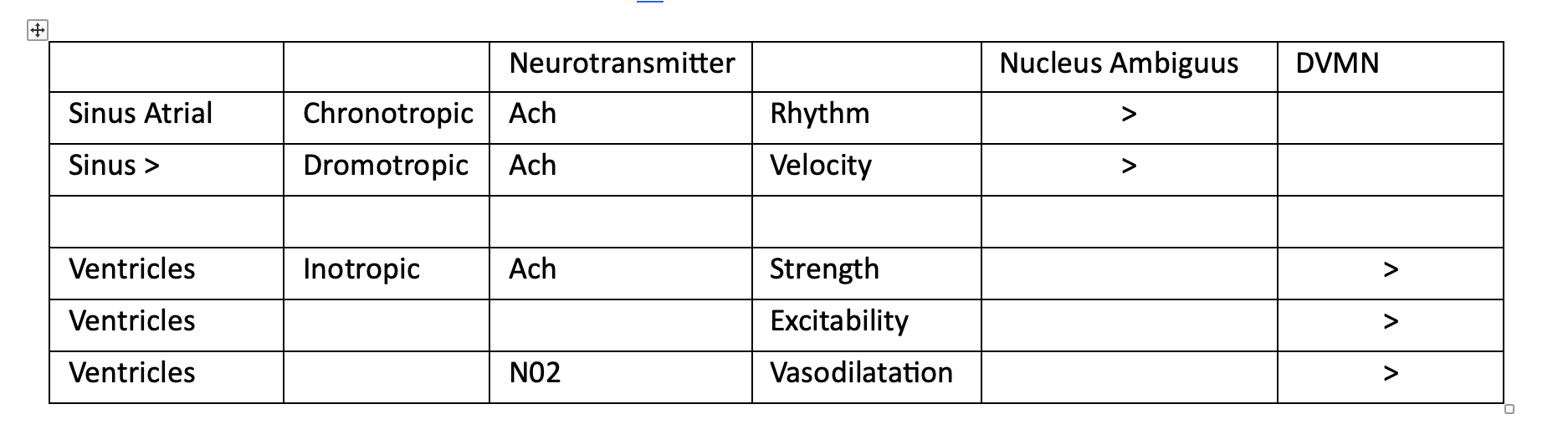
Activation of muscarinic receptors by acetylcholine released from postganglionic neurons has three effects
Reduces heart rate (chronotropic)
AV conduction velocity (dromotropic)
Myocardial contractility (inotropic).
Recent research (Machhada, 2020) has shown that the DVMN could increase the contractility of the left ventricle.
2.5.8
Respiratory Centers in the Brainstem
To get the full picture of the Nucleus Ambiguus, we need to understand the close connection of the NA (dorsal and ventral parts) with the NTS and the respiratory centers. The NA lies just above the pre-Bötzinger (pre-BOT), the main inspiratory group. With pacemaker properties, the preBOT generates a respiratory rhythm. The NA has no such quality, as it responds to external impulses, mainly from the NTS and respiratory centers.
As shown in Respiratory Modulation Of Premotor Cardiac Vagal Neurons In The Brainstem (Dergacheva, 2010), the respiratory and cardiovascular systems are highly intertwined anatomically and physiologically. Their neurons, often co-localized in the same brainstem regions, belong to very different systems:
Nucleus ambiguus (dorsal branchiomotor and ventral parasympathetic)
NTS (solitary tract nucleus) and DMVN (dorsal motor vagal nucleus)
Sympathetic centers in the ventral spinal cord (RVM and CVM)
Pontine, dorsal, and ventral respiratory groups
Selective transduction and photoinhibition of pre-Bötzinger neurons projecting to the facial nucleus in rats affect nasofacial activity (Melo, 2022) describes the interaction between the pre-Bötzinger respiratory center and the CNVII (facial). This is an interesting example of cooperation between respiratory and facial structures.
Massari (1995) in Cardiotopic organization of the nucleus ambiguus? An anatomical and physiological analysis of the neurons that regulate atrioventricular conduction cites the cases of swallowing syncope. In patients with “swallowing syncope”, the distension of the lower esophagus leads to the development of paroxysmal AV block and fainting during or immediately after swallowing. Esophageal motoneurons are located in the rostral part of the NA, next to the external formation (NAext).
The Brainstem Respiratory Network (Bautista, 2014) provides a comprehensive list of respiratory centers. Three networks of neurons mediate the production and coordination of breathing, swallowing, and coughing.
Neurogenic control of respiration (pp. 311-348) in Introduction to basic aspects of the autonomic nervous system (Appenzeller, et al., 2022) also provides a remarkable account of the respiratory centers. Therefore, we have taken the latter authors as the source for the following list and graphic:
The Dorsal Respiratory Group is a subnucleus of the NTS, mainly involved in inspiration. Together with the rVRG (see below), it is a source of the phrenic nerve (PMN), which moves the diaphragm.
The Ventral Respiratory Group lies ventrolateral to the nucleus ambiguus for almost its entire length. It contains the cVRG and rVRG (caudal and rostral respiratory groups), the preBOT (pre-Bötzinger complex), the BOT (Bötzinger complex), and the retrotrapezoid nucleus (RTN).
The pontine respiratory group (PRG) includes the parabrachial and Kölliker-Fuse nuclei and the serotoninergic medullary raphe complex.
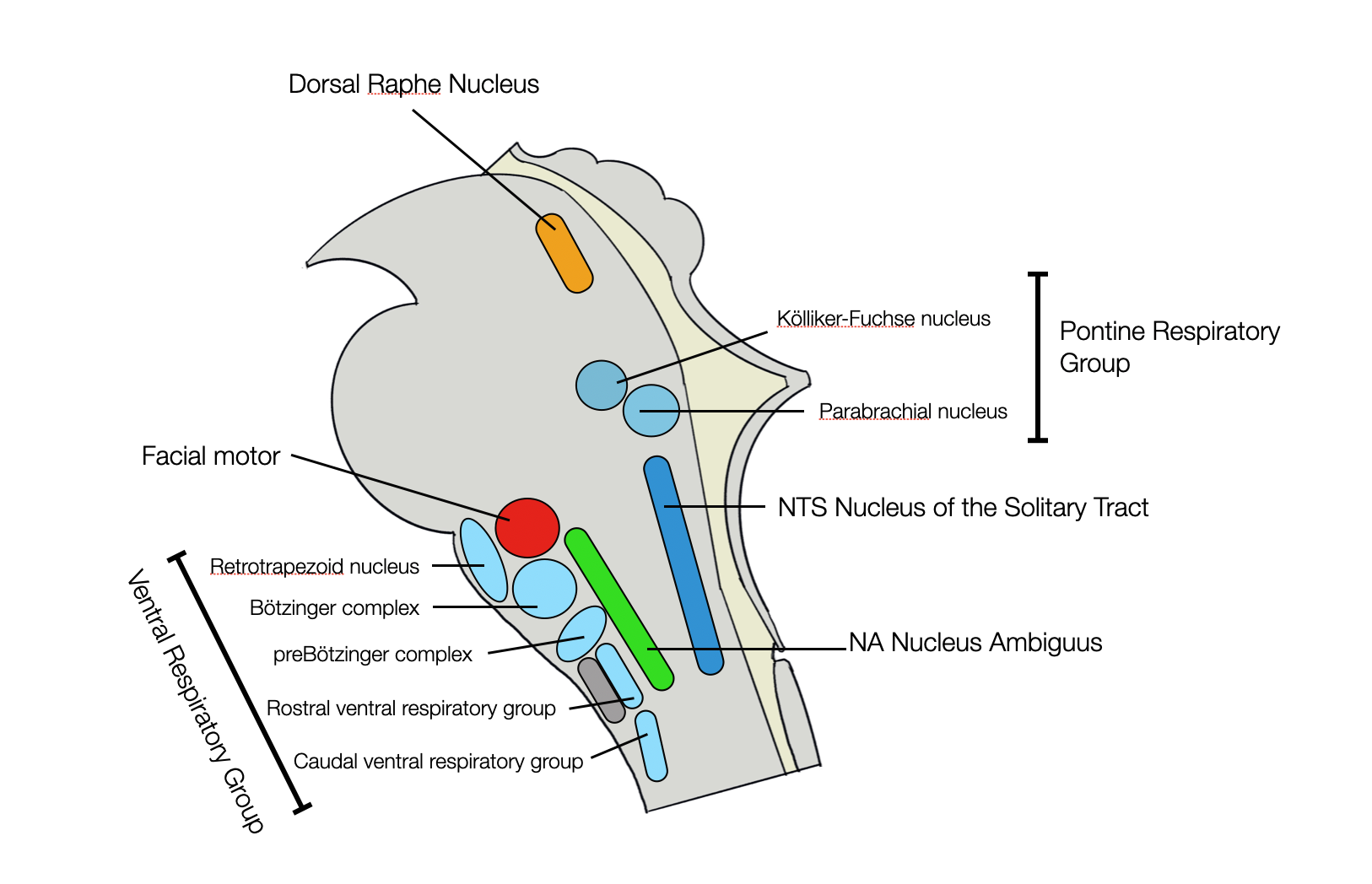
Lateral view of medulla and pons showing the three respiratory groups. We see the proximity of the NA and the ventral respiratory complexes. Adapted and simplified from Appenzeller (2022, p. 313). Please refer to the original book (see above). (©absb,2024).
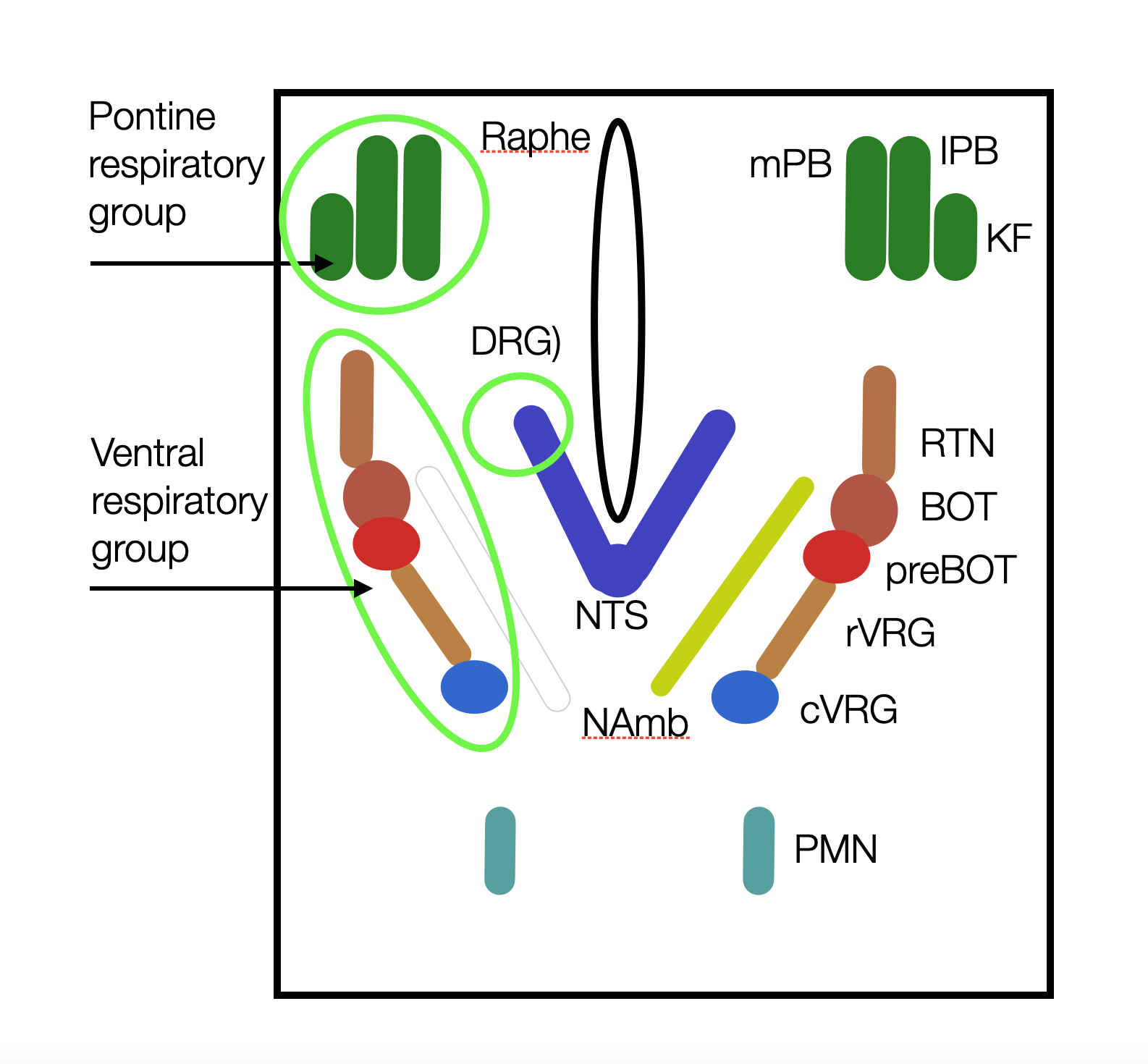
Schematic representation of the brainstem areas involved in respiratory control. Although not in the same area, the ventral NA and the dorsal NTS are in constant communication (©absb, 2024).
>> to the next chapter Dorsal