2.4.0
Does the Dorsal Vagal Cause Sudden Death?
This topic is the PVT's first stumbling block for two reasons:
“First” because “sudden infant death syndrome” (SID) was what motivated Porges to start his crusade against the “dorsal vagus.”
“Stumbling” because Porges fell into a false dilemma fallacy, assuming that one of the two parasympathetic branches should be the culprit while the other was the savior. It didn't occur to him that other factors might be at play. Unfortunately, recent research contradicting this theory hasn't made him change his mind.
2.4.1
The Neonatologist
In his Pocket Guide to the Polyvagal Theory, Porges tells us how the PVT was born (Porges, 2017, p. 59). In his research about the autonomic system, he wrote a paper published in Paediatrics (Porges, 1992) to educate neonatologists about the utility of assessing vagal tone (via RSA) in the neonatal nursery. « Following publication of the paper, I received a letter from a neonatologist » writing that «the notion of vagal activity being protective was not consistent with his training (…) In medical school, he had learned that the vagus could kill you.» Porges adds, «I immediately understood what the neonatologist meant. From his perspective, the vagus was capable of promoting life-threatening bradycardia and apnea, characterized by massive slowing of heart rate and cessation of breathing (…). For months, I carried the neonatologist’s letter in my briefcase. I tried to explain the paradox.» Eventually, Porges concluded that the efferent vagus had a double anatomic origin (dorsal and ventral): «One system was potentially lethal, while the other system was potentially protective.» This was the beginning of a long journey. Unfortunately, the very first premise of his theory was wrong. The vagus doesn’t kill babies.
In a non-explicit way, Porges suggests that sudden death, either in infants or in “voodoo death,” is caused by a “dorsal-vagal” fatal slowing of the heartbeat (bradycardia).
After discovering the vagal paradox, he never stopped looking for cases of sudden death that could justify his theory. Because the death of a newborn or infant is probably the most devastating experience of loss, PVT operates in a very emotional field. No one can remain indifferent. Therefore, the search for the culprit (“the Dorsal”) gave the PVT a massive popular resonance from the beginning. Unfortunately for the PVT, the Dorsal is only the scapegoat. As new techniques inexorably improve our knowledge of the brain, a very different story comes to light.
Porges uses two examples of sudden death to prove his point: Sudden Infant Death Syndrome and Voodoo Death. To complete the picture, we add the cases of young athletes who die of sudden cardiac arrest during or after intense physical exertion.
2.4.2
Sudden Infant Death (SIDS)
Sudden Infant Death Syndrome (SIDS) is the leading cause of death in infants aged 1 month to 1 year, and it is estimated that there are approximately 2,500 cases of SIDS in the United States each year. SIDS occurs primarily in an (apparently) healthy infant during sleep. In recent years, public health campaigns have more than halved the incidence of SIDS; however, despite this dramatic decline in incidence, SIDS remains the leading cause of sudden and unexpected infant death in Western countries, accounting for nearly 50% of all post-neonatal deaths. In 60-80% of cases, the cause remains unexplained, even after postmortem examination.
While researchers have been searching for explanations for decades, more and more risk factors have been identified. It is now believed that a single factor does not cause SIDS but is multifactorial. Several models have been proposed to explain the multifactorial nature of SIDS, with the most widely accepted model being the “triple risk model.” According to this model, three factors must be present in an at-risk infant: a critical developmental period for homeostatic control and exogenous stressors (e.g., smoking parents or incorrect bed positioning). A benign cause-such as the flu or exposure of the face to cold water (Ottaviani)-may serve as the ultimate lethal trigger in an infant with an invisible vulnerability (e.g., an underdeveloped arcuate nucleus in the midbrain).
Most researchers have rejected the hypothesis of autonomic causality (vagal input) from the beginning — in the 50s. As it seems now, the origin of bradycardia is the external formation (NAext) of the ambiguous nucleus (NAext). It's impossible to make a “dorsal” diving reflex the cause of SID, as it has been proven that the human diving reflex is “ventral.” However, neither ventral nor dorsal vagal can cause death. There are many other factors involved!
The first international conference on SIDS was held in Seattle in 1963. In 1984, during the International Symposium on Sudden Infant Death Syndrome, Sydney Segal summarized: Twenty Years of Progress in Research on Sudden Infant Death Syndrome. (Harper, 1988, pp. 21-26), describing different epochs focusing successively on morphology, function, sleep physiology, arousal failure, infant apnea, vagal stimulation, airway obstruction, or epidemiology. Later, Hannah C. Kinney, in The Brainstem in the Sudden Infant Death Syndrome: a Review (ibid. pp. 115-134), presents various morphological and biochemical findings as possible causes of SIDS, without being conclusive. A year earlier (Tildon, 1983), 300 scientists and health professionals from 13 countries had already concluded how elusive the research results were. It quickly became apparent that SIDS was the product of many factors. In 1994, Filiano and Kinney introduced the triple risk model: a) vulnerable infant, b) critical developmental period, and c) exogenous stressors (e.g., smoke, heat, position) (Singh, 2016).
Surprisingly, the same hypotheses persist 40 years later (Ottaviani, 2014). Multiple congenital anomalies (in the brainstem, but mostly higher, in the arcuate nucleus), molecular anomalies, or delayed myelination are often observed. Although the technology has improved significantly (e.g., genetics, molecular biology, and optogenetics), the problem remains challenging for all researchers, as no simple explanation fits all cases. One thing is for sure: the vagal brake hypothesis is never the cause.
Worldwide, about 2.6 million infants die at birth (Urdal, 2021). 98% are in low- and middle-income countries (LMIC). The leading causes are asphyxia and prematurity (Hunt, 2006).
During fetal development, the heart rate is 120-160 (Pildner, 2013), slowing from an average of 134 to 119 in the last 150 minutes before death. Immediately after birth, the heart rate increases to a mean of 168 and slowly declines to 136 60 minutes later (Munyaw, 2023).
For more information: fetal cardiac pathology.
2.4.3
The Serotonin Hypothesis of Sudden Death
Because serotoninergic centers in the brainstem play a critical role in helplessness (hopelessness) and breathing, researchers have recently begun to explore their role in sudden infant death (SID).
Take a deep breath and wake up: The protean role of serotonin preventing sudden death in infancy (Cummings, 2020)
Recordings of infants who died suddenly and unexpectedly show recurrent apneas, ineffective gasping, and failure to reestablish eupnea and arousal before death—a constellation of serotonergic defects in the caudal raphe nuclei of these infants. Similar symptoms are observed in animals with the same defects. The caudal serotonergic system provides an organized defense against cardiorespiratory reflex inhibition and hypoxia associated with prolonged apnea, bradycardia, and hypotension. Deficiency of caudal serotonergic function increases the propensity for sudden, unexplained infant death.
Update on congenital heart disease and sudden infant/perinatal death: from history to future trends (Ottaviani, 2017)
Unexplained stillbirth and SIDS share brainstem developmental anomalies such as hypoplasia of the arcuate, hypoglossal, inferior olivary, pre-Bötzinger, dorsal and ventral cochlear, medial inferior vestibular, obscurus and pallidus raphé nuclei in the medulla oblongata; Locus coeruleus, facial/parafacial complex, retrotrapezoid nucleus, superior olivary complex, superior and lateral vestibular nuclei, Kölliker fuse, median and magnus raphé nuclei in the pons; inferior colliculus, substantia nigra, dorsal and caudal linear raphé nuclei in the caudal mesencephalon (Figure 2). The detection of frequent congenital anomalies, similar in both SIDS and SIUDS ( sudden intrauterine unexpected death syndrome) sudden intrauterine unexpected death syndrome victims, indicates continuity between these two fatal forms, considered the SIDS-SIUDS complex.
Overall, abnormalities of serotonergic neurotransmission in the caudal brainstem are the most consistent findings relevant to the pathogenesis of SIDS, likely resulting in a reduced ability to respond appropriately to periods of cardiorespiratory stress or reduced arousal.
2.4.4
Dive Reflex and Sudden Death
The dive reflex (DR) is characterized by breath-holding (apnea), bradycardia, and vasoconstriction, which leads to elevated blood pressure.
Several authors have questioned the role of the dive reflex in sudden death. This would fit well with the idea of reptilian dorsal activity. However, according to Molecular defined circuits of cardiovascular and cardiopulmonary control (Veerakumar, 2022), the mammalian dive reflex is mediated by a group of neurons located in the external formation of the nucleus ambiguus (NAext). This formation contains two distinct populations of neurons: cardiovascular (ACV) and cardiopulmonary (ACP) neurons. The second population is active in the diving reflex, triggering the simultaneous bradycardia and bronchoconstriction following water immersion. The diving response in mammals is primarily physiological (Ponganis, 2011), (Panneton, 2013), (Ponganis, 2017), (Panneton, 2020), (Williams, 2021). The sperm whale — a mammal — has an average dive duration of about 40–60 minutes, reaching depths of 400-900 m. Find more in the Shutdown chapter.



2.4.5
Trigeminal Reflex
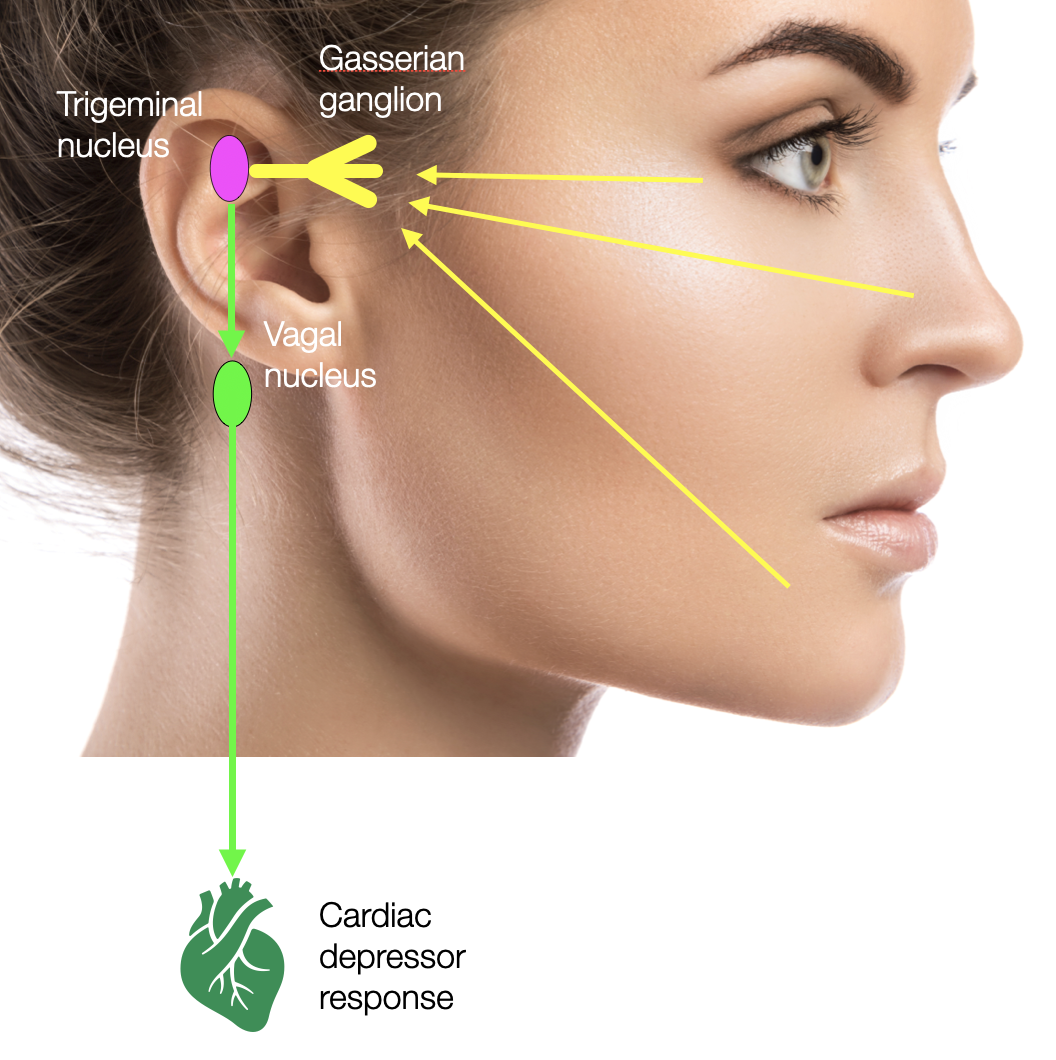
Simplified representation of the Trigeminal Reflex, triggered by a strong sensory stimulation at the face level (e.g., surgery, or cold water).
Another explanation for SIDS is the trigeminal reflex. The sensory portion of the 5th cranial nerve, the trigeminal nerve, brings information from the periphery (e.g., the face and cavities of the mouth, nose, and eye) to the brain. It relates to the trigeminal centers in the spinal cord, the NTS, the nucleus ambiguus, the DVMN, and the respiratory centers. Sudden trigeminal nerve stimulation (e.g., cold water or surgery) can cause bradycardia, asystole, hypotension, apnea, or gastric hypermotility.
It has been compared to the dive reflex, which includes apnea, bradycardia, and bronchoconstriction. However, as we will see later, the dive reflex is a physiological reflex triggered by a deep dive that causes hypoxia. It involves the sympathetic and parasympathetic (nucleus ambiguus) systems.
Sudden infant death triggered by dive reflex, the role of the trigeminal reflex (Matturi, 2005). Three Italian searchers, Matturri, Ottaviani, and Lavezzi, narrate an intriguing case of Sudden Infant Death triggered by immersion in cold water. It has been reported that washing the face with cold water or plunging into cold water can provoke cardiac deceleration. This report describes a case of SIDS “that offers a unique insight into the role of the dive reflex in determining a lethal outcome.”
A 10-month-old male infant presented with vomiting and dysentery during a swimming lesson after being immersed in cold water. He was taken to the emergency room, where a gastroenteric disturbance of possible viral origin was diagnosed. He was rehydrated and discharged. The following night, while lying in his mother’s arms, the baby suddenly became unresponsive. Emergency help was summoned. Resuscitation efforts were attempted by external cardiac massage and assisted ventilation, followed by the repeated administration of adrenaline. Still, the child remained unconscious and was pronounced dead a few minutes later.
The baby had been born at term after an uncomplicated pregnancy. He seemed normal at birth and continued in apparent good health until the day he died. The baby was bottle-fed, and both parents were cigarette smokers (two packs/day). An autopsy revealed bilateral underdevelopment (hypoplasia) of the arcuate nucleus (part of the hypothalamus) and atherosclerotic lesions in the heart. Parental smoking may have played a role here, too.
Our comment: There is confusion between the trigeminal reflex (triggered by contact with cold water) and the diving reflex (activated by diving and consequent hypoxia). The child was not diving, so this was not a hypoxic situation.
Trigeminal Cardiac Reflex: New Thinking Model About the Definition Based on a Literature Review Meuwly, 2015). After the pioneering work of Schaller and colleagues, the trigeminocardiac reflex (TCR) is now a well-established brain stem phenomenon in skull-base surgery and related disciplines. It is also the only documented cardiac arrest caused by the vagus nerve, notably in SDI.

Prolonged asystole induced by trigeminocardiac reflex accompanied with abnormal heart rate variability during percutaneous balloon compression: a case report (Zhang, 2023). The trigeminal cardiac reflex (TCR) can cause bradycardia and even cardiac arrest, and is reversible with removal of the stimulus. A 68-year-old man experienced cardiac arrest during percutaneous balloon compression for the treatment of trigeminal neuralgia. This patient did not regain sinus rhythm with cardiopulmonary resuscitation (CPR). The patient regained sinus rhythm and was premedicated with atropine 0.5 mg, allowing the operation to resume. The operation was successfully completed without complications. Heart rate variability (HRV) analysis showed that parasympathetic activity was predominant before induction of anesthesia and after tracheal intubation. It was further increased during foramen ovale puncture, resulting in prolonged asystole. Sympathetic activity was predominant after atropine administration. This may be beneficial in patients with TCR, as atropine plays a key role in preventing TCR. In addition, HRV analysis may be essential for preoperative screening of high-risk patients.
Sudden Infant Death Syndrome — Role of the Trigeminocardiac Reflex: A Review (Singh, 2016). The TCR is a neurogenic reflex that manifests as bradycardia, hypotension, apnea, and gastric hypermotility. It shares many similarities with DR, an important physiological adaptation to hypoxia during apnea in many animal species, including humans. Several studies have described congenital abnormalities of the autonomic nervous system in the pathogenesis of SIDS (e.g., hypoplasia, delayed neuronal maturation, decreased neuronal density of the arcuate nucleus, hypoplasia and neuronal immaturity of the hypoglossal nucleus). HR is regulated by sympathetic and parasympathetic input to the sinoatrial node. The last part of the reflex arc of the TCR is formed by the cardioinhibitory input of the ventral vagal branch, which is distributed to the sinoatrial and atrioventricular nodes and not to the ventricles. The main action of the vagal nerve is chronotropic.
The trigeminocardiac reflex – a comparison with the diving reflex in humans (Lemaitre, 2015)
Serotonergic Modulation of the Trigeminocardiac Reflex Neurotransmission to Cardiac Vagal Neurons in the Nucleus Ambiguus (Gorini, 2009)
Trigeminocardiac Reflex: The Current Clinical and Physiological Knowledge (Chowdhury, 2015)
Trigeminocardiac reflex during maxillary third molar extraction: Our experience (Agarwal, 2022)
Autonomic regulation and bradycardia during the neonatal period (Patural, 2014). Nasal tube passages, autonomic regulation, and neonatal bradycardia.
2.4.6
Voodoo Death

Walter Cannon, an American physiologist, first described the phenomenon of voodoo death in 1942. He observed that some people in traditional African or Haitian societies who believed they were under a curse or spell would die shortly after being condemned by the tribe's sorcerer, even if they were not physically ill. Cannon suggested that intense psychological stress could lead to death under these dramatic circumstances. The unquestioned belief in a curse can cause a massive response in the sympathetic system, resulting in a critically elevated heart rate and blood pressure, known to cardiologists as paroxysmal tachycardia (= fast heart). Irregularities (arrhythmias) and inadequate contractions of the heart lead to reduced performance and the formation of blood clots, which may block the coronary arteries. The risk of myocardial infarction (stroke) is high.
Porges raised the question of the mechanism of death: if the heart of the poor victim was dilated at death (diastole), this would support the dorsal-vagal explanation. In the case of a final contraction (systole), the sympathetic hypothesis would prevail. Without being a cardiologist, everyone should understand that this question is irrelevant. Since the heart is under massive sympathetic stimulation — caused by a misleading belief system — it is slowly being knocked down. No chance. This is not the fault of the sympathetic system, for it requires a special cultural impregnation — superstition — supported by higher centers such as the anterior cingulate cortex (ACC). You can't curse a dog or a mouse to death!
Porges' fallacy is that the dorsal is the only cause. If he had opened a cardiology book, he would have understood that tachycardia is the main cause of death. Recent research based on optogenetic techniques shows that the “dorsal-vagal” hardly influences the heart rhythm. The DVMN innervates the ventricles (the lower part of the heart), reducing the intensity of contraction (regulating the work of the heart muscle), dilating the coronary arteries for better heart perfusion, and reducing the local inflammatory process after a heat stroke. If we had to personify the “dorsal”, it would undoubtedly be the protecting mother or the nurse.
Voodoo Death vs. Hopelessness vs. Helplessness
Later, Porges felt confident enough to offer a polyvagal explanation for the phenomenon of “voodoo death” presented by Walter Cannon (Cannon, 2002). Based on stories he read, Cannon, who coined the concept of fight-flight, postulates that the death of a poor man cursed by an Aboriginal sorcerer is a sympathetically caused process (Cannon, 1942). The veracity of this phenomenon has since been severely criticized (Sternberg, 2002). In Voodoo Death (Lester, 2009), Lester questions the existence of voodoo death as commonly described. Richter (1957) suggests a third explanation: hopelessness. Long before Seligman, Richter conducted experiments on rats swimming in a closed vessel and showed that various treatments (e.g., holding the rat firmly or cutting its whiskers) were crucial for survival. But while leaning toward a parasympathetic alternative to Cannon's sympathetic explanation, Richter introduces a new hypothesis: hopelessness. Several decades later, research will show how subcortical structures in the frontal brain regulate learned helplessness (Peterson, 1993) or learned hope (Maier, 2016) (Seligman, 2018). According to recent research (Andalman, 2019) using the zebrafish, a similar mechanism of learned conditioning appears in all vertebrates. It seems that the decision to surrender to death is not made in the brainstem but much higher up. A concomitant sympathetic deactivation and parasympathetic overtone may be present, but the autonomic structures execute orders; they are not the rulers.
Reading:
Learned helplessness : a theory for the age of personal control (Peterson, 1993), Learned helplessness at fifty: Insights from neuroscience (Maier, 2016), The hope circuit : a psychologist's journey from helplessness to optimism (Seligman, 2018).
More under Shutdown.

Survive!
Cardiological pathology
Too often the medical pathology (Edwards, 2006) is absent in the Voodoo death discussion. Lecomte (Lecomte, 1996) presents a study of 43 cases of sudden death occurring immediately after experiencing emotional stress (during or within two hours after the event). Ninety percent had no clinical antecedents, but at the autopsy, evidence of previous myocardial infarction was present in 92% of the patients with corresponding coronary atherosclerosis (clocked arteries of the heart). An enlarged heart (cardiomegaly) was present in most of the subjects. In this series, an acute coronary thrombosis was found in only 8 of 43 cases, suggesting that most subjects suffered an arrhythmic death in the setting of prior silent myocardial infarction.
2.4.7
Karoshi Death

Karoshi death is usually the extreme result of an acute cardiovascular event (heart attack), including stroke (Ke, 2012). This social medicine term first appeared in Japan in the late 1970s. Karoshi deaths are typically due to overwork. Two neuroendocrine systems are involved: epinephrine (adrenaline)/norepinephrine (noradrenaline) and cortisol. The first group (catecholamines) contributes to elevated blood lipid levels, increased blood clotting, and atherosclerosis.
In most cases, hypertension is associated with thickening of the artery walls, narrowing of the blood vessels, and increased peripheral resistance in the cardiovascular system. On the other hand, cortisol can affect cell metabolism and fat distribution, leading to cardiovascular disease, type 2 diabetes, and reduced immune function. Lundberg and Hellstrom found that women who worked more than 50 hours per week had twice the cortisol levels of women with more moderate workloads (Lundberg, 2002).
Grippo et al. researched a group of prairie voles that replicated some aspects of Richter's experiment. Unlike most other rodent species, the prairie vole is among only 3% of mammalian species that actively engage in long-term bonds, bi-parental care, or family groups (Carter, 2002). Social isolation and disruption of established social bonds in this species result in depressive and anxiety-like behaviors. Increased heart rate, decreased variability, and impaired vascular relaxation are observed. Social stressors also produce ventricular and supraventricular arrhythmias in prairie voles (sympathetic overdrive).

Molecular Mechanisms of Cardiac Death
Unlike the relatively slow synaptic transmission found in the nervous system, the heart uses a more straightforward depolarization mechanism to mediate the spread of electrical excitation from cell to cell. The diffusion of ions — the generation of current — occurs through junctional structures.
These channels comprise two connexons of six proteins (connexin 43 = Cx43), forming a gap junction. The researchers observed that four weeks of isolation followed by one week of mild environmental stress (one hour of forced swim test FST per day) affected the composition of these channels. Cx43 proteins were partially replaced by Cx45 proteins, reducing the size of the gap junctions and possibly causing arrhythmia. The researchers found that social stress (but not the forced swim test, FST) alters Cx43 expression, while FST alters Cx45 and the Cx43/Cx45 ratio. Both social and physical stressors induce various depressive behaviors in rodent models, including anhedonia (lack of pleasure) and helplessness. The combination of the two factors has an additive effect. Social isolation alone leads to an increase in helpless behavior (immobility), but FST in combination produces more significant levels of immobility. Disruption of connexin proteins represents a cellular mechanism by which social stressors are arrhythmogenic and produce a cascade of cardiovascular dysfunction.
Similarly, in the human heart, ventricular myocytes express connexin 43 and only trace amounts of connexin 45. In congestive heart failure, connexin 43 levels decrease, connexin 45 levels increase, and gap junction size decreases (Grikscheit, 2008).
Xiao et al. (2019) investigated the molecular aspects of arrhythmias caused by overexertion leading to karoshi. They also found decreased expression of Cx43 (55.6% reduction) in the FST group compared to the control group. They observed that myocardial fibrosis decreased muscle contractility and affected cardiac conduction, causing arrhythmia and consequent cardiac dysfunction. In conclusion, long-term overexertion disrupts cardiac rhythm at the molecular level and increases myocardial fibrosis, leading to karoshi.
2.4.8
Sudden Cardiac Arrest

As a leading cause of mortality and morbidity in affluent societies, Sudden Cardiac Arrest (SCA) underscores the need to identify individuals at risk. However, the etiology of SCA is complex, with predisposing and precipitating factors interacting. Although anxiety and psychological stress have been associated with SCA for decades, their precise role and impact remain unclear, and the biological basis is poorly understood.
Anxiety may, in part, predispose to SCA by contributing to other risk factors such as cardiovascular disease and diabetes mellitus through mechanisms such as unhealthy lifestyles and metabolic abnormalities. Mental stress appears to precipitate SCA, presumably by more directly affecting the cardiac ion channels that control the heart's electrical properties. This may lead to ventricular fibrillation, the arrhythmia that underlies SCA. Experimental studies that elucidate the underlying biological mechanisms are essential to advance this field of research and are most easily designed for mental stress as a precipitating factor because of the short time frame.
Sudden Cardiac Arrest (SAC), which accounts for 20% of all deaths in industrialized societies, is mainly caused by cardiac arrhythmias (ventricular fibrillation), which may result from inherited cardiac ion channel dysfunction or cardiomyopathies. In recent decades, several searches have identified molecular pathologies called "channelopathies". The faulty channels that regulate the flow of ions (e.g., calcium, natrium or potassium) across the cell membrane can indeed be affected by genetic mutations (Jongsma, 1998), (Verberne, 2022), leading to an unstable sympathetic-parasympathetic regulation. On the other hand, mental stress also seems to trigger SCA (Batelaan, 2021), which alters cardiac ion channels, leading to ventricular fibrillation, which is an arrhythmia (de la Grandmaison, 2006), (Tester, 2009), (Guan, 2010), (Roberts, 2010), (Bastiaenen, 2011), (Napolitano, 2012), (Sridharan, 2022).
Electrocardiographic Repolarization During Stress From Awakening on Alarm Call (Toivonen, 1997). The ANS influences ventricular repolarization. Repolarization is a determinant of ventricular excitability and is fundamentally involved in arrhythmogenesis. Sudden stress evokes life-threatening arrhythmias in subjects with certain predisposing conditions, suggesting that autonomic strong nervous influences can abruptly render the heart vulnerable.
Stressful events as a trigger of sudden death: a study of 43 medico-legal autopsy cases (Lecomte, 1996). Post-myocardial infarction fibrosis was present in 25 cases (92%) of sudden coronary death. In conclusion, our autopsy study indicates that SCD occurs primarily in individuals with severe heart disease, particularly coronary artery disease.
Sudden cardiac death with stress and restraint: The association with sudden adult death syndrome, cardiomyopathy and coronary artery disease (Krexi, 2016).
Sudden and rapid death during psychological stress. Folklore or folk wisdom? (Engel (1971) presents several cases of sudden death. There is no mention of a vagal cause.
2.4.9
Athlete’s Heart Attack
What causes young athletes' heart failure and death during or after intensive training? Athletes were specifically studied to explain Sudden Cardiac Death (SCD), which often occurs during or after exercise. The first known victim was the Athenian soldier who ran from Marathon to Athens to deliver the news of victory and died on arrival. In athletes under 35, inherited heart disease predominates, with hypertrophic cardiomyopathy (HCM) and anomalous origin of a coronary artery being the two most common causes in the United States. In athletes older than 35 years, most SCD events are due to acquired atherosclerotic coronary artery disease (CAD) (Wasfy, 2016), (Fanous, 2019).

Everyone knows the story of someone who suddenly felt dead without any known pathological condition. In the case of infants (SIDS), the post-mortem examination usually reveals chronic, preexisting lesions that can't be evaluated in a routine medical examination. Is the evil “back” at work again?
There are several potential causes of heart failure and death in young athletes during or after intensive training, such as hypertrophic cardiomyopathy, congenital defects, inflammation (myocarditis), and coronary aortic stenosis.
One of the most common causes is hypertrophic cardiomyopathy (HCM), a genetic disorder that causes the heart muscle to thicken and become unable to pump blood effectively. Another cause is coronary artery anomalies (CAA), congenital abnormalities of the coronary arteries that can lead to myocardial ischemia or infarction (heart attack), and sudden cardiac death. Other potential causes include myocarditis (inflammation of the heart muscle), arrhythmias (abnormal heart rhythms), and aortic stenosis (narrowing of the aortic valve).
It is important to note that these conditions are rare and that most young athletes who participate in sports have a low risk of heart failure and death. However, a thorough medical evaluation before beginning an intensive training or exercise program can help identify any underlying conditions that may put an athlete at risk.
In conclusion, Porges' lack of knowledge in cardiology leads him to the wrong conclusions. The causes of sudden cardiac death are genetic factors, anatomical malformations, hyperactivation of the sympathetic system by physical and mental stress, or external toxic factors (such as smoke and heat).
2.4.10
Genetics and Sudden Death
Genetic Determinants of Sudden Unexpected Death (SUPD) in Pediatrics (Koh, 2022). More than 10% of infant and child deaths in the United States occur suddenly, unexpectedly, and without an established cause, exceeding child mortality from cancer and heart disease.
These deaths, which typically occur in healthy children during sleep, are known as sudden infant death syndrome (SIDS), sudden unexpected infant death (SUID), or sudden unexplained death in childhood (SUDC) and may be collectively termed sudden unexpected death in pediatrics (SUDP).
SIDS/SUID - the major component of SUDP - accounts for 1/6 of all infant deaths at a rate of 1 per 1000 live births. SIDS is the leading cause of postneonatal mortality.
These findings provide further evidence for the role of genetic factors in SUDP. It also supports the premise that children who die from SUDP may have intrinsic vulnerabilities that distinguish them from unaffected children. The finding of specific genetic contributions to SUDP in 11% of the cohort studied highlights the role of genetics in SUDP and suggests multiple mechanisms.

2.4.11
Did Kathleen Folbigg murder her four children?
Kathleen Folbigg: Woman jailed over infant deaths pardoned. See article.
The June 2023 BBC News article shows how new scientific evidence suggests she did not kill her four young children. A woman once branded “Australia's worst serial killer” has spent 20 years in prison after a jury found she killed sons Caleb and Patrick and daughters Sarah and Laura over a decade. The 55-year-old's case has been described as one of Australia's biggest miscarriages of justice. Professor Carola Vinuesa discovered that Kathleen Folbigg carried a unique and potentially pathogenic heart mutation, which her daughters Laura and Sarah also carried. She enlisted a colleague, Todor Arsov, to visit Folbigg in prison and obtain a DNA sample. When the two examined the sequence, they found a mutation in a gene, CALM2, that codes for a protein called calmodulin, the malfunction of which has been linked to cardiac arrhythmias and disorders and even death in infancy. Calmodulin is involved in the calcium-modulated transmission of cardiac depolarization, which is essential for keeping the heart beating.
In a recent investigation, a team of immunologists found that Ms. Folbigg's daughters shared a genetic mutation — called CALM2 G114R — that can cause sudden cardiac death. Genetic testing revealed a mutation (CALM2-G114R) in the calmodulin 2 gene in Ms. Folbigg and her daughters, Sarah and Laura. Her sons, Caleb and Patrick, did not carry the mutation.
What is CALM2?
Jozef Gecz, professor of human genetics at the University of Adelaide, said the essential role of calmodulin means the protein is so “protected” that any mutations in the gene are rare and have serious consequences. "Once you tamper with it, you introduce a problem and a potential explanation for why the heart can fail,” he said. There are 20,000 genes in the DNA of all humans, and the three calmodulin genes are among the top 15 that have remained unchanged throughout evolution. Calmodulinopathy, the name given to the abnormal heart rhythm caused by mutations in the calmodulin genes, affects only one in 35 million people. So far, only 135 cases have been reported worldwide.
Read also the report: How a geneticist led the effort to free a mother convicted of killing her kids.
Infanticide vs. inherited cardiac arrhythmias (Brohus, 2020). This article details how the genetic variations impaired calmodulin's ability to bind calcium and regulate two key calcium channels in the heart, causing sudden cardiac death in children.
Further reading
(Bacq, 2022), (Bove, 2018), (Brito, 2023), (Cerrone, 2012), (Crotti, 2019), (Engel, 1971 ), (Guan, 2010), (Hefti, 2016), (Hefti, 2016), (Kawanishi, 2019), (Kawanishi, 2020), (Koh, 2022), (Wang, 2007).
2.4.12
Caveats of the ”Vagal Paradox”
Asphyxia is the most common cause of SIDS
When oxygen levels in the body drop, such as during a stroke or asphyxia, it's similar to a motor running low on fuel. Oxygen is vital for the cardiac cells, the muscles that keep the heart pumping. Without enough oxygen, these muscles can’t function effectively. If breathing stops, the body doesn't fail due to a neurogenic 'dorsal death,' but rather from anoxia—where the heart muscles are deprived of the oxygen they need to continue working.
Following an ischemic or hemorrhage stroke, most patients experience cardiac arrhythmia. A network of cortical and subcortical brain sites regulates the autonomic nervous system. The chief hubs that regulate this network include the insular cortex, amygdala, hypothalamus, and paraventricular nucleus. It is thought that after a stroke, direct damage or secondary metabolic changes to any of these areas alter the regulatory balance of the sympathetic and parasympathetic nervous systems (Abrams, 2016).
Arrhythmias
In the case of a cardiac cause, rhythm disturbances (arrhythmias) correlate primarily with increased sympathetic tone. Due to a chaotic distribution of impulses from the sinus to the atrium and the ventricle, the heart muscle can't contract properly. The heart fails. The idea of a complete “vagal brake” on the heartbeat is unlikely because the heart has an intrinsic group of “pacemaker” cells with a relatively fast beat. The external vagal modulation can slow them down, but not block them.
The dorsal vagal doesn’t slow down the heart. In higher mammals, the ventral vagal branch (NAext or external formation of the nucleus ambiguus) is related to the frequency (chronotropic) and speed (dromotropic) of the heart. According to the latest findings, the dorsal part of the efferent vagus (DVMN) reduces the excitability of the ventricles. It protects them during a cardiac event (e.g., myocardial infarction).
PVT starts on the wrong foot with the first postulates:
An increase in vagal (dorsal) tone should be fatal. But fifty years of intensive research on SIDS has never come to that conclusion.
Vagal braking (supposedly by the DVMN) should cause respiratory arrest. But it does not: The breathing cycle is created and regulated by a group of six different centers in the brain stem (e.g., pre-Bötzinger). External causes such as smoke, heat, and position in bed are also critical factors. Some vagal efferent impulses (mainly from the nucleus ambiguus) may constrict the bronchial tubes (as in asthma), but respiratory arrest occurs abruptly, without precedent of dyspnea.
The “vagal paradox” offers a purely functional explanation of SIDS (the same mistake is later made with the phenomenon of autism). It doesn't consider the many genetic, morphological, or biomolecular anomalies (e.g., butyrylcholinesterase)(Harrington, 2022) that thousands of international researchers have already identified. The PVT sees the heart like the old cars from the sixties or before; when the driver stopped at a traffic light, he/she always had to step on the accelerator. Otherwise, the engine would have stalled. Later, manufacturers invented “idle,” which eliminated this problem. Continuing with this metaphor, a cardiac arrest is due to the engine itself (out of gas, dirty spark plugs, or “broken” machine), but certainly not to the driver's foot. The vagal and parasympathetic systems don't drive the heart; they modulate it! They can negatively contribute to a pre-existing pathology (see below).
The polyvagal explanation also doesn't consider the sympathetic control: tachycardia (accelerated rhythm), arrhythmia, clot formation, and more. Norepinephrine can cause the death (apoptosis) of heart muscle cells (myocytes) {Communal, 1998 #1156}, contributing to the progression of heart failure.
The PVT doesn't consider the role of the trigeminal cardiac reflex (Singh, 2016) (Lemaitre, 2015), (Gorini, 2009), which can cause a dramatic decrease in heart rate and blood pressure through an exaggerated vagal reflex involving the ventral vagus (NAext) — not the dorsal.
P.S. In Polyvagal Perspectives (Porges, 2024, p. 6), Porges suggests one more time that the dorsal vagal branch (DVMN) may create problems through “important influences of cardioinhibitory inotropic vagal fibers originating from the dorsal motor nucleus of the vagus.” The studies cited are (Gourine, 2016) (Machhada, 2015, 2016, 2017, 2020). Porges didn't read the articles carefully; the first one mentioned decreased contractility of the left ventricle, but Gourine (2019)–not quoted by Porges– and Machhada (2020) present studies based on the new optogenetic technology–animals are awake and no more under anesthesia. The DVMN increases the contractility! Porges then cites Richter (1957), who doesn’t describe the role of the DVMN but rather concludes with a psychological state of “hopelessness.” More in chapter dorsal, the DVMN.
Conclusion
The arguments proposed by the PVT to explain sudden death in infants, adults, or in “voodoo death” don't correspond to the current medical and neuroscientific knowledge. A modern approach is required.
>> to the next chapter Ventral Vagal Complex