2.12.0
Fact or Fiction? The Truth About Dorsal Vagal Shutdown
Porges and Deb Dana describe the dorsal vagal shutdown as a primitive response to a lethal threat– a survival mode. This functional freeze includes collapse, immobility, depression, and dissociation. Here, I critically take a position and review the freezing response, tonic immobility, learned helplessness, hibernation, and the diving reflex based on current scientific literature.
Dissociation has been discussed in a separate chapter.
Freezing vs. Tonic Immobility
Periaqueductal Gray Matter PAG
Immobility and Meditation
Quiescence
Sleep
Dive Reflex
Hibernation
Learned Helplessness
Depression
2.12.1
Freezing vs. Tonic Immobility
Toward the end of the last century, the more dramatic expression “freezing” gradually replaced the neutral term “tonic immobility” which was more specifically related to the motor system and suggested readiness for action. Freezing evokes a motor arrest and a radical metabolic change - from vapor to ice. A simultaneous drop in blood pressure and slowing of the heart (bradycardia) are described (see literature below). However, various authors, such as Nieujenhuys, have strongly criticized this model, arguing that the first phase of tonic immobilization during an inescapable encounter is accompanied by a high sympathetic tone (e.g., tachycardia). In the second phase, the surrender phase, accompanied by a high parasympathetic tone, bradycardia, and hypotension occur, with a decrease in muscle tone.
Some authors (Roelofs, 2017) make a mistake in describing the motor arrest (behavior) as controlled by the parasympathetic system. This arrest is a joint response of the somatic and autonomic systems, initiated by the PAG in the midbrain (or even higher, in the amygdala, hypothalamus, or neocortex). Somatic and visceral (autonomic) territories are thus confused. From a physiological perspective, it's obvious that efferent somatic and postganglionic visceral fibers target different receptors.
Several authors have tried to make a clear distinction between “freezing” and TI (tonic immobility) (Leite-Panissi, 2001). Carli and Farabollini, in their must-read Defense from Invertebrates to Mammals: Focus on Tonic Immobility (2022), clarify the terms extensively:
Freezing is pre-contact. Prey can still fly. The purpose of freezing is to reduce the detection risk and assess the situation. This brief immobilization (a few seconds) with a brief slowing of the heartbeat is related to the “Orientation response » described by Sokolov. This short time allows decisions to be made.
Tonic immobility (TI) is caused by prolonged physical contact (e.g., the searcher holding the animal by the neck or in an inverted position) with no possibility for the prey to escape. This behavior may also occur before contact if the situation is hopeless.
What is the difference between freezing and tonic immobility? In TI, immobility persists (from a few seconds to 30') after the threat (e.g., restraining the animal) has ceased.
Hofer has shown in Cardiac and Respiratory Function During Sudden Prolonged Immobility in Wild Rodents (Hofer, 1970) that the cardio-respiratory response differs between rodent species (tachycardia vs. bradycardia). In the wild, a long period of immobilization to avoid detection, prepare for the next step, or play dead is of adaptive value. During this period, breathing is most rapid and shallow. This is not fainting. It is characterized by a reversible state of profound physical inactivity and a relative lack of awareness of the environment.
The immobility may be waxy rather than tonic, as in alligators lying on their backs. In Hemodynamics of tonic immobility in the American alligator, Young (2018) describes a hemodynamic mechanism similar to vasovagal syncope.
In Endogenous opiate analgesia induced by tonic immobility in guinea pigs, Leite-Panissi et al. (2001) show that restraint, pressure, and inversion produce combined tonic immobility (which persists after the procedure) and opioid-based anesthesia. The opioid antagonist naloxone suppresses the anesthesia but not the immobility. As we can see, immobility, anesthesia, unresponsiveness, and cardiovascular responses each represent a distinct action. A confusing description such as polyvagal “shutdown,” which mixes motor, autonomic, and psychological responses, is not helpful.
Different authors argue for the correctness of their concepts, but no academy can settle the debate. As I argue later (What else?), the best solution is to disentangle the various systems involved and deconstruct the responses.
2.12.2
Periaqueductal Gray Matter (PAG) – why should we know this funny word?
Whether fight, fly, or freeze, the Periaqueductal Gray Matter is the most essential component of defensive behavior: it is responsible for rapid responses to threats, stress, and challenges.
Aqueduct is an old word from when Romans built canals (= ducts) or bridges to bring water (= aqua) from one place to another. The Aqueduct of Silvius connects the third ventricle (chamber) and the fourth ventricle. A group of nuclei form a gray matter sleeve around (= peri) the canal.

This is an acqueduct bringing water!
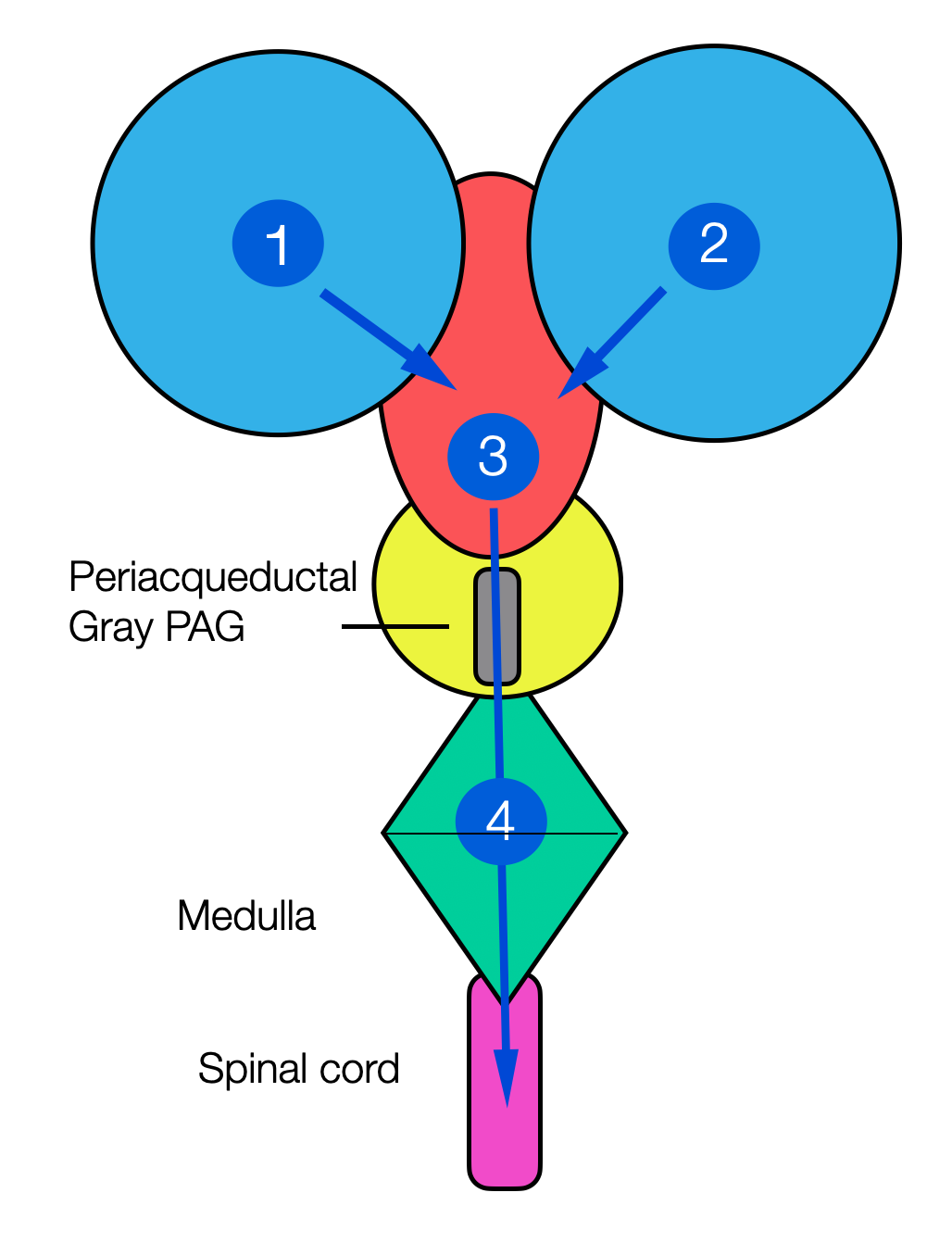
These schematic diagrams show the location of the PAG. The first illustration-reminiscent of Mickey Mouse-illustrates the circulation of cerebrospinal fluid from the first two ventricles to the third ventricle. It then travels through the upper midbrain to the fourth ventricle and then down to the spinal canal.

Here, we see the “sleeve” of gray matter (collection of neuronal cell bodies around the canal)

This is a section through the midbrain (mesencephalon) with the tectum (= roof) and the tegmentum (= cover).
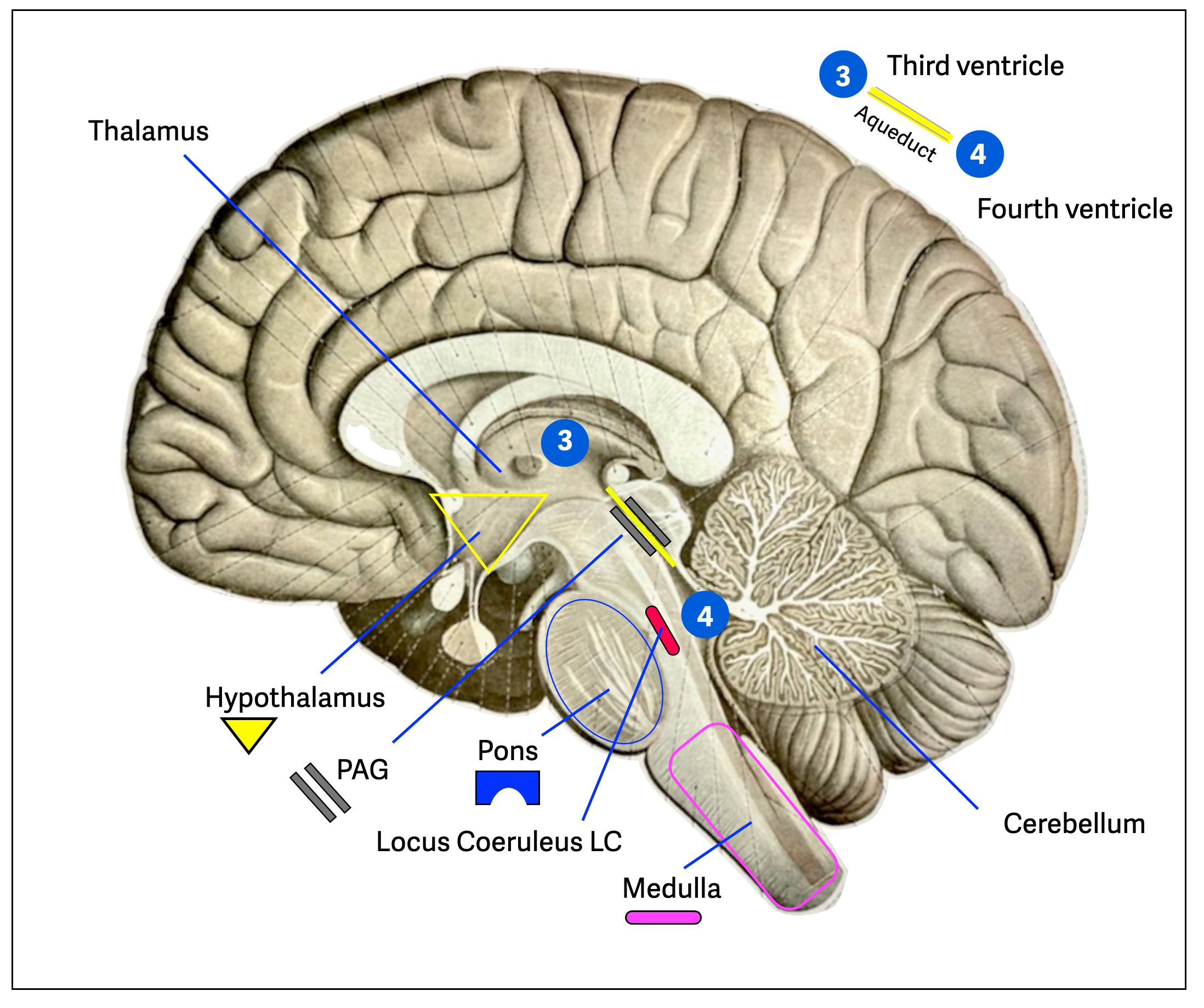
This illustration (background by Sobotta, 1922) shows the main components of the defensive reflex, which can be activated in three possible scenarios: fight, flight, and freeze. This is where the magic happens, but also the tragedy. These reactions are barely under the control of the cortical zones. While they can save a life, they can also wholly surprise the victim of an attack, who will wonder why he/she felt paralyzed and unable to react. This could be a civilian victim of sexual assault or a soldier/police officer under heavy fire.
In the following sections, we will focus on immobilization. The PVT describes immobilization as part of the "dorsal" metabolic shutdown, which is strange. The DVC stimulates and regulates digestion, a major metabolic act, while the sympathetic antagonizes digestion.
In (Porges, 2012), p. 195), the author quotes Leite-Panissi et al. (2003) and then states: “Consistent with the polyvagal theory, the vlPAG communicates with dorsal vagal complex, whereas the lPAG and dlPAG communicate with the sympathetic nervous system.” Porges doesn't explain how he suddenly came to this conclusion, which is pure wishful thinking - neither "vagal" nor "dorsal" appear in the article. Perhaps he is referring to another article (Keay, Bandler, 2001), a little higher up, that attributes the three defensive patterns to three discrete components of the PAG. This localization is tempting, but as we will see later, recent research confuses this order.
Moreover, the dorsal parasympathetic (DVMN) is not involved in the immobilization defensive pattern, which is a somatomotor (muscular) response accompanied by a ventral parasympathetic (NAext) bradycardia. Yes, you read that right: the nucleus ambiguus (NAext) slows down the heartbeat during immobilization.
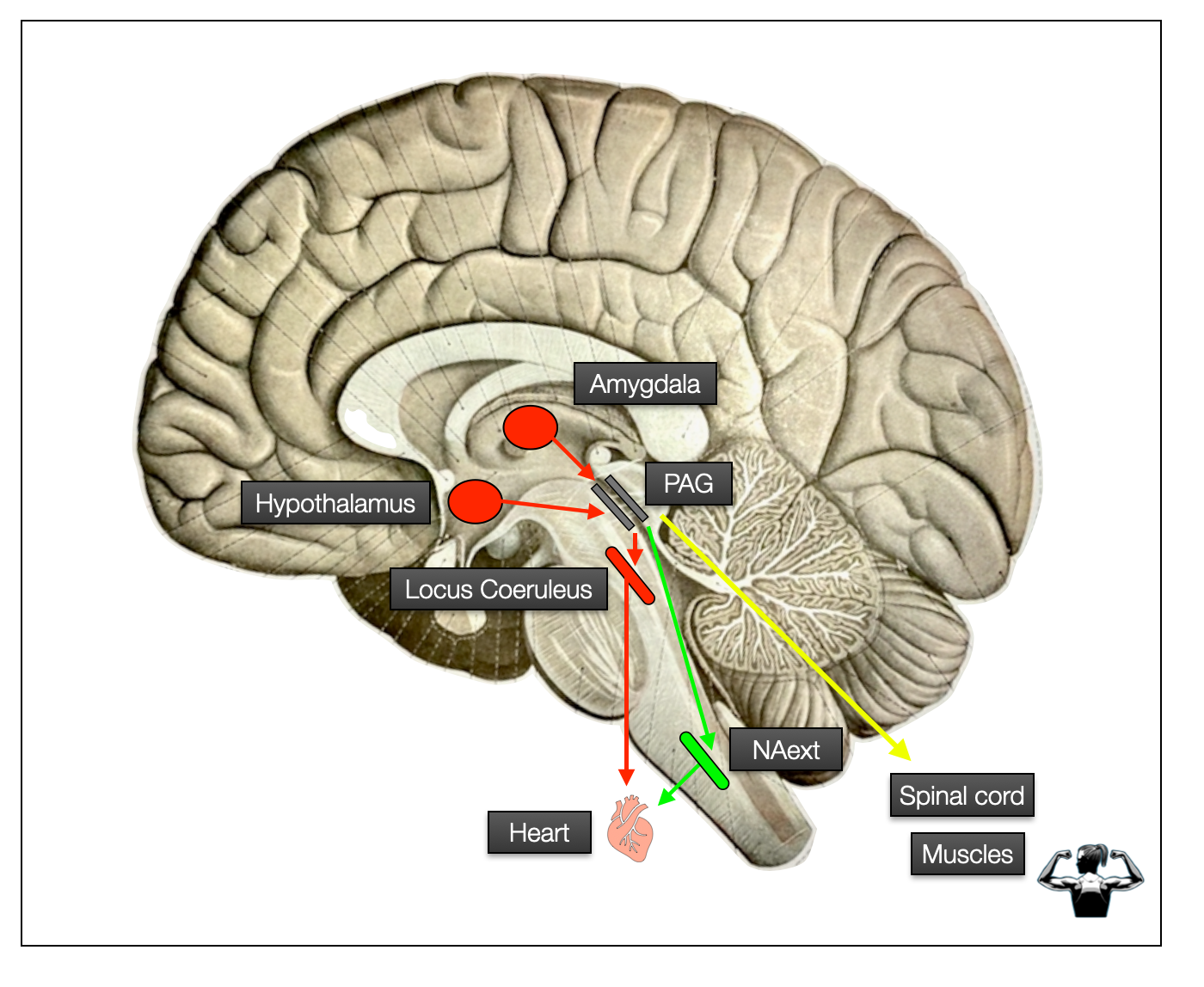
This next schematic (brain shape based on Sobotta, 1922) illustrates some parts of the chain of command from the hypothalamus or amygdala to the PAG and then down to the heart (autonomic pathway) or muscles (somatomotor pathway).

Chain of command: Hypothalamus, Amygdala, PAG, and Periphery
The PAG is the command center for pre-ordered responses to threats. Here are the fight, flight, and freeze reflexes. These patterns are protocol-driven responses, similar to firefighters following a predefined procedure in an emergency. SWAT would also be a good comparison. Once higher centers have given the green light (“It's a go!”), there is no reassessment, which would take time and energy. These pre-programmed responses act on two primary axes: somatic motor (muscles) and autonomic (sympathetic vs. parasympathetic). Depending on which region is stimulated, it triggers specific motor behaviors (e.g., fight, flight, or immobilization) and neurovegetative responses in parallel — sympathetic for action and parasympathetic for immobilization. It's important to understand how the chain of command works: the PAG is the boss, NAext and LC execute, not the other way around, as the PVT suggests.
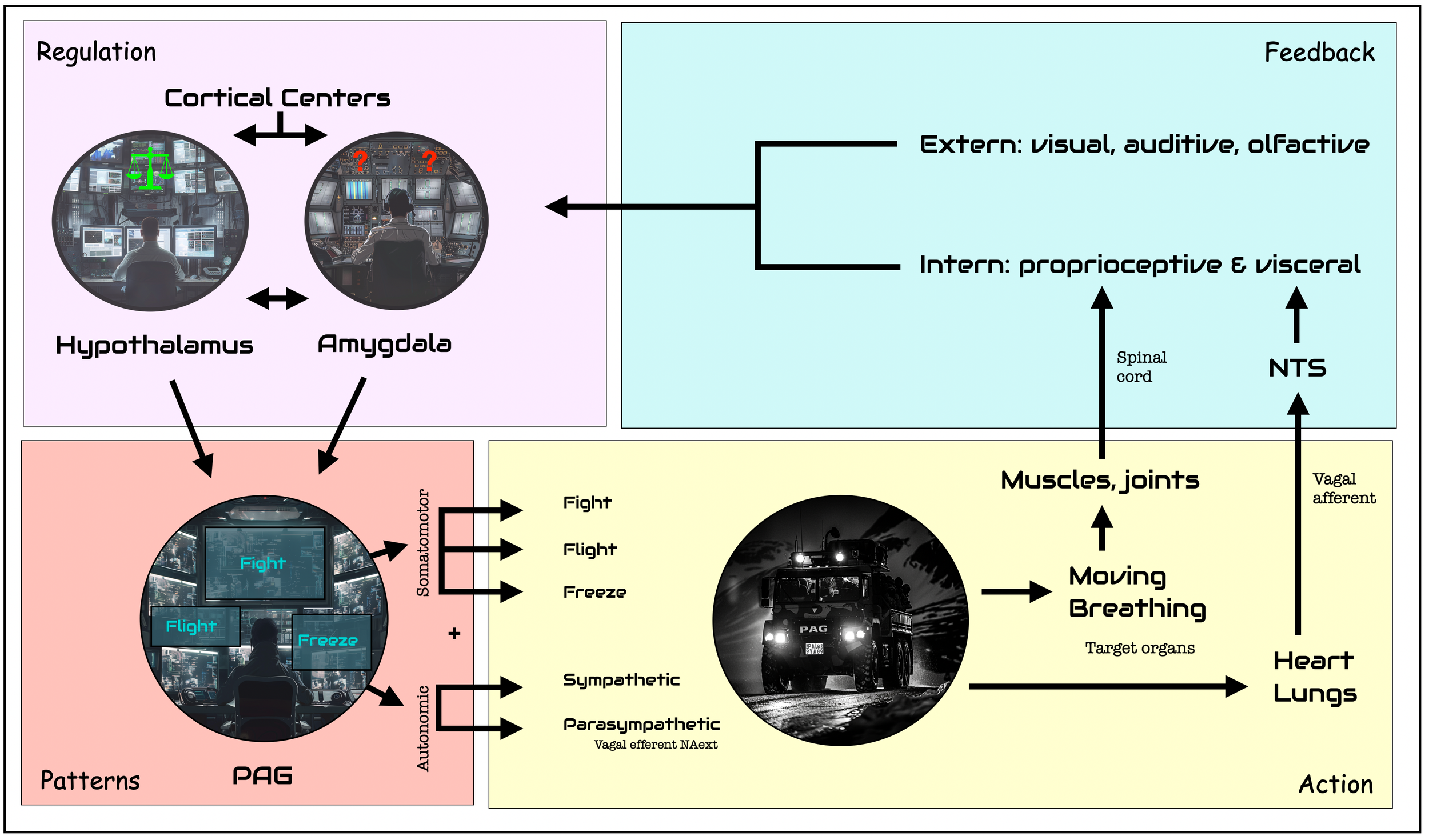
Action loop. In this schematic, we see the circuit of response to a critical incident, starting at a cortical or subcortical level, orchestrated by the PAG, and executed by the somatomotor and autonomic systems. A feedback loop runs up through the spinal cord and the sensory vagal nerve, parallel to the external perceptual pathway. The PAG is the operations center that immediately sends a SWAT team (truck) into action. It can react quickly thanks to predefined fight, flight or freeze patterns. The amygdala is specialized in detecting novelty (positive or negative) and threat levels. The hypothalamus' main goal is to maintain balance, but it is sometimes the source of aggressive behavior. Higher centers perform cognitive tasks, such as assessing the reality of the threat (e.g., ACC) or developing strategies for action (prefrontal cortex): ©drmuret, 2024.
2.12.3
Literature on Periaqueductal Gray Matter (PAG)
Although the literature on PAG and tonic immobility is not extensive, research shows promising potential as new techniques (e.g., optogenetics) are developed and sometimes controversial results are obtained. Most authors describe a somato- and viscerotopic organization of the PAG, mostly in parallel columns, with specific response patterns.
Quiescence and hyporeactivity evoked by activation of cell bodies in the ventrolateral midbrain periaqueductal gray of the rat (Depaulis, 1994). Such injected rats became quiescent, i.e., all ongoing spontaneous activity ceased. These rats were also hyporeactive: the partner's exploratory approaches failed to elicit orientation, startle responses, or vocalization. In response to “deep” or “inescapable” pain, chronic injury, or defeat, animals often reduced their somatomotor activity, became more solitary, and were generally less responsive to their environment.
In Central circuits mediating patterned autonomic activity during active vs. passive emotional coping (Bandler, 2000), we see how rostral and caudal of the same column can evoke different patterns: confrontation vs. escape.
Fear bradycardia and activation of the human periaqueductal grey (PAG) (Hermans, 2013). A defense mode observed at intermediate threat levels is freezing: a cessation of locomotion characterized by a parasympathetically dominated autonomic nervous system response that causes a slowing of the heart rate or “fear bradycardia.” Studies in rodents have shown that freezing depends on projections from the amygdala to the PAG. The human PAG is involved in the parasympathetically dominated defense mode of attentive immobility.
Role played by periaqueductal gray neurons in parasympathetically mediated fear bradycardia in conscious rats (Koba, 2016). This article examines the role of PAG neurons in the induction of fear bradycardia. White noise sound (WNS) exposure at 90 dB induces freezing behavior, which elicits bradycardia in conscious rats. The bradycardia induced by WNS exposure is parasympathetic, abolished by atropine, and involves neurons of the lateral/ventrolateral PAG (l/vlPAG). Retrograde neuronal tracing shows that numerous l/vlPAG neurons send direct projections to the nucleus ambiguus (NA).
In Role played by periaqueductal gray neurons in parasympathetically mediated fear bradycardia in conscious rats (Koba, 2016), the authors describe how exposure of rats to white noise (90db) induces freezing behavior and bradycardia. The bradycardia is mediated by neuronal activation of the lateral/ventrolateral PAG (l/vlPAG) - an in situ injection of a GABA agonist (blocker) suppresses it. Neuronal tracing shows these neurons send projections directly to the nucleus ambiguus (NA). They “propose that the l/vlPAG-NA monosynaptic pathway transmits fear-driven central signals that induce bradycardia via parasympathetic outflow.”
Wang et al. (2021) describe how a specific projection (dorsal premammilary) to the PAG controls the escape reflex.
Koustikou et al. (2015) show how the PAG orchestrates sensory and motor circuits at multiple levels, including the spinal cord, cerebellum, and spinal motor reflex pathways.
Most authors attribute active defensive patterns to the dorsal PAG (dlPAG) and inactive defensive patterns (immobilization) to the ventral PAG (vlPAG) (Leite-Panissi, 2003). Recent research, mainly based on optogenetic techniques, challenges this view.
Deng et al. (2016) describe how optogenetic activation of dPAG neurons induces defensive behaviors, including running, freezing, and avoidance. Optogenetic stimulation of the pathway between the hypothalamus (VMH) and the dlPAG produces immobility, but not flight or avoidance.
Similarly, Cola et al (2023) show how chemo- and optogenetic activation of specific hypothalamic neurons terminating in the upper dlPAG (dorsolateral) leads to rapid breathing, bradycardia, and immobility.
In summary, while current research demonstrates the complexity of defensive responses, we can see the critical role of the PAG in orchestrating somatomotor (e.g., immobility) and autonomic (e.g., bradycardia) responses to an inescapable threat. Unlike the PVT, the NAext induces bradycardia and not the DVMN (dorsal parasympathetic), which is entirely absent from the image.
2.12.4
Why Immobility and Meditation Are Not ‘Dorsal Shutdown?
PVT and immobility
The PVT makes three assumptions, which he then equates:
1. Immobility, a characteristic of “reptiles,” is a form of dissolution (Jackson) and phylogenetic regression.
2. Immobility implies a metabolic shutdown that can harm the mammalian heart.
3. Immobility is uncomfortable and even frightening.
The PVT combines all three claims into one cliché: immobility is a potentially lethal reptilian stunted behavior induced by the “dorsal”. Porges has long had a problem with immobility. Consistent with the “reptilian dorsal” explanation for immobility, he tried to solve the problem by using oxytocin as a calming factor that allows copulating and nursing females to hold on until males and babies are satisfied. Remember that his wife, Sue Carter, is a top expert in this field. So, Porges found oxytocin in the “family pharmacy.” In Role of Social Engagement in Attachment and Bonding (Porges, in Carter, 2005), Porges explains how mating and nursing females can stay still without fear.
The erroneous equating of immobility = reptilian = fear certainly doesn't help the debate.
In rare interviews, Porges admits that a form of immobility — quiescent immobility — can be experienced without fear. See Love’s Brain: A Conversation with Stephen Porges (Loizzo, 2018). Porges paints a fascinating portrait of the dorsal vagus in this interview — another illusion. The silence of the mind certainly doesn't originate in a visceral complex designed to modulate appetite, digestion, the immune system, and cardio-protection, to name the most basic functions. We must look higher in the brain to the general cortical networks. Meditation practices reducesactivity in the DMN (default mode network), resulting in less mind-wandering and an increased sense of presence.
Let's go back to the three polyvagal statements:
1. As presented elsewhere (a reptilian dystopia), immobilization is not the default response of reptiles. Just observe lizards basking in the sun: when disturbed, they run and hide; they don't freeze. Snakes, like humans, have all three responses: attack, hide, or freeze. Worldwide, 5.4 million people are bitten each year, resulting in 1.8 to 2.7 million cases of poisoning. The WHO reports that 81,000 to 138,0000 people die annually from snakebites.
2. Immobility saves energy, but the sloth, a mammal, is the world's laziest animal. Bradycardia is not a typical reptilian trait: slowing of the heart during diving is seen in numerous aquatic mammals. In humans, the fatal slowing of the heart occurs with pre-existing tissue, molecular, and genetic defects.
3. Immobility from nursing or consensual sex is not freezing. A sexologist knows that the need to be active or passive — as a man or a woman — is very variable. And that's okay! While it is true that, for example, a restless New Yorker landing in Bali or Mykonos may need to adjust before “just doing nothing,” there is nothing pathological or exceptional about this behavior (including the siesta). In our bustling cities, we have forgotten to do it.

Brain networks and meditation

The relationship between meditation and brain networks, particularly the default mode network (DMN), has been the subject of extensive research. Here are some key studies and reviews discussing these interactions:
Brewer, J. A., Worhunsky, P. D., Gray, J. R., Tang, Y. Y., Weber, J., & Kober, H. (2011). This study, titled “Meditation experience is associated with differences in default mode network activity and connectivity,” published in the “Proceedings of the National Academy of Sciences,” provides evidence of altered default mode network connectivity in experienced meditators.
Fox, K. C. R., Nijeboer, S., Dixon, M. L., Floman, J. L., Ellamil, M., Rumak, S. P., Sedlmeier, P., & Christoff, K. (2014). In their meta-analysis titled “Is meditation associated with altered brain structure? A systematic review and meta-analysis of morphometric neuroimaging in meditation practitioners,” published in “Neuroscience & Biobehavioral Reviews,” the authors provide an overview of how meditation might influence brain structures, including those in the default mode network.
Tang, Y. Y., Hölzel, B. K., & Posner, M. I. (2015). This review article titled “The neuroscience of mindfulness meditation” in “Nature Reviews Neuroscience” discusses how mindfulness meditation practice affects brain regions related to the default mode network, attention, and self-regulation.
Garrison, K. A., Zeffiro, T. A., Scheinost, D., Constable, R. T., & Brewer, J. A. (2015). Their study, “Meditation leads to reduced default mode network activity beyond an active task,” in the “Cognitive, Affective, & Behavioral Neuroscience” journal, also contributes to understanding the effects of meditation on the DMN (default mode).
These publications provide a solid foundation for understanding how meditation affects brain networks, particularly the default mode network, and how these changes correlate with the subjective experiences reported by practitioners.
Anatomy of Meditation
Tang (2015) published information about the brain regions involved in mindfulness meditation. These two illustrations are based on Tang's (2015) (anatomical background from Sobotta, 1922, modified). All these regions are cortical and don’t involve the brainstem.
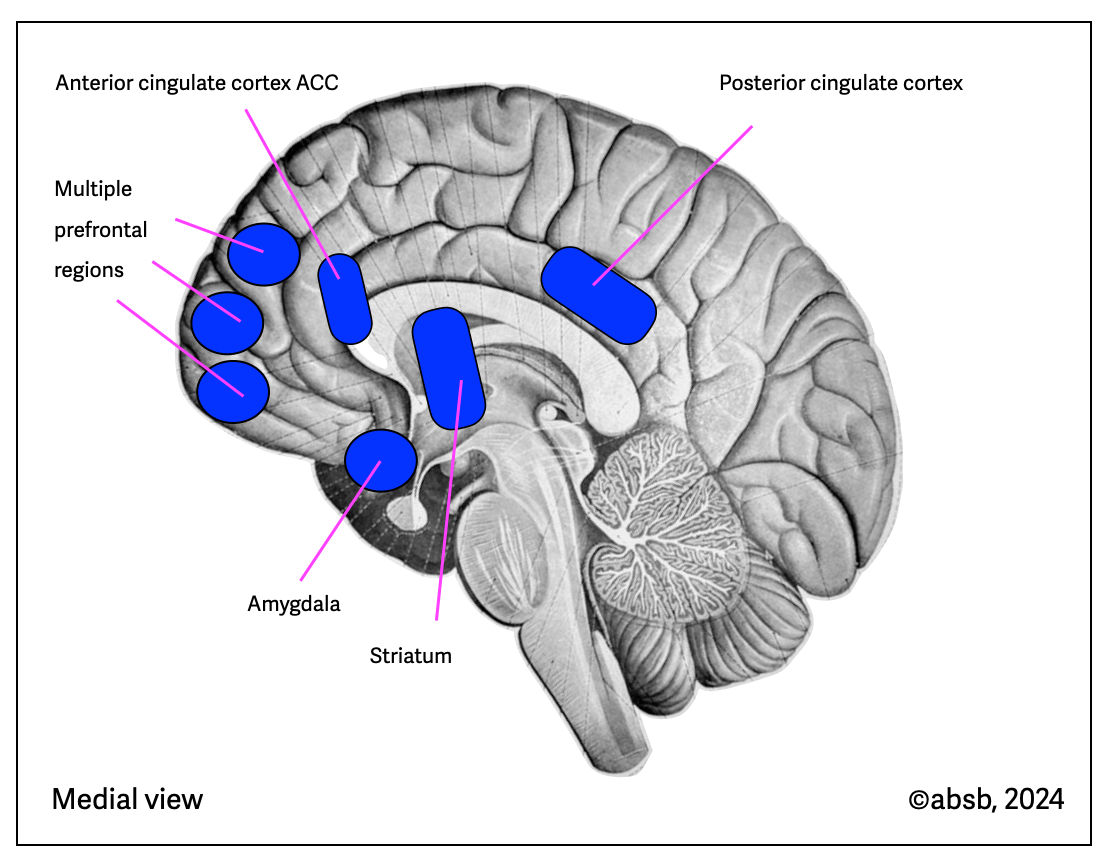
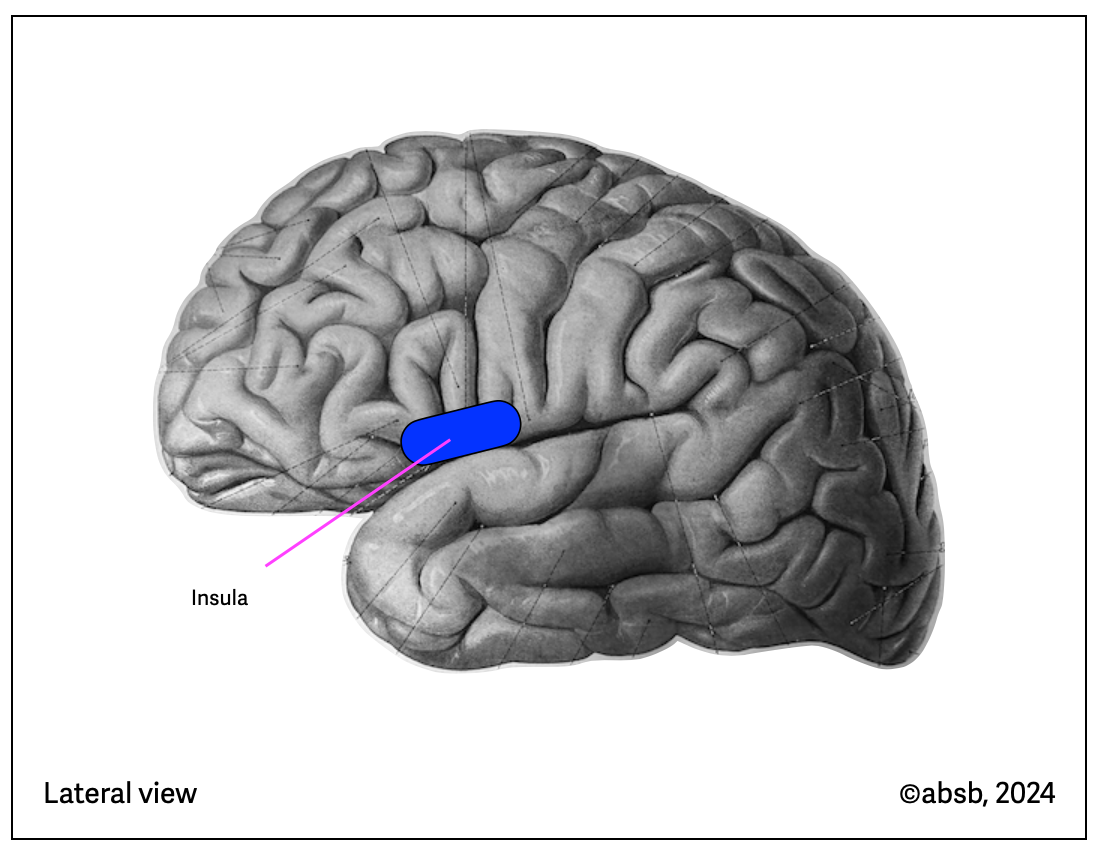
2.12.5
Quiescence: Even Simple Cells go to Sleep
In biology, the term quiescence has another signification. Cellular Mechanisms and Regulation of Quiescence (Marescal, 2020). Quiescence is “a state of reversible growth arrest in which cells have exited the cell cycle but remain capable of renewed division upon stimulation. Entry into quiescence allows cells to persist in a non-dividing state over extended periods of time and enact mechanisms to protect themselves from damage.”
Consistent with the PVT, Porges proposes the quiescence to be a positive property of the “dorsal.” As he sees the whole world parted between the three realms (ventral, sympathetic, and dorsal), he has not much choice– he places quiescence aside from intimacy. Just as positive sexual immobility is only possible thanks to the intervention of oxytocin, so quiescence, and intimacy need the security of the “ventral.” In Love’s Brain: A Conversation with Stephen Porges (Loizzo, 2018), Porges declares, “In the early 90’s, I went to India to study yogis and learned some interesting things from them. Many of their practices are designed to trigger the older vagal shutdown response that drives the faint or freeze reflex.” “ In my own model, the older evolutionary part of the vagus (…) is a very powerful system of calming that enables the deepest forms of connectedness and intimacy. But it can only be consciously recruited for social life when our bodies are in safe states. That’s where the newer vagus literally is your shield.” In this conversation, Porges suddenly makes the “dorsal” the place of “self-intimacy.” “Yogis who hold their breath, like deep-sea divers, are in a sense saying, “If I can master this biological shutdown reflex by controlling my breath, I can now visit these powerful physiological states.”
This impressive description reminds us of a flyer for a workshop, “The Dorsal Vagal, Door to Spirituality,” by Sônia Gomes (2012). Unfortunately, the “dorsal” has little to do with spirituality. What we have here is just another polyvagal story. The nucleus ambiguus (ventral) and the sympathetic system create the diving reflex. The DVC has other functions. Also, “self-intimacy” mostly happens when we are socially disconnected: in silence, with a closed door, maybe with a “please do not disturb” sign. Meditation is a solitary activity. As the Gospel of Matthew 6:6 says: “When you pray, go into your room, close the door, and pray to your Father.” In Rapid Hypnosis (a variation of hypnotic induction), the hypnotist doesn't need progressive relaxation to create immobility; the shock induction and immediate immobility induce a blissful trance. Hypnosis uses an innate circuit just waiting to be reactivated!

2.12.6
Do we need sleep?
Everybody needs sleep! – even worms and parakeets.
Although animals have different sleep behaviors, the phenomenon is ubiquitous. In most animals (e.g., zebrafish, tegu lizard, birds, or mammals), we observe two sleep modes of brain activation. These phases are paradoxical (rapid eye movement = REM) and orthodox (NREM) sleep in humans. In dolphins, one hemisphere of the brain may be asleep while the other is asleep. The common swift (martinet) can sleep while flying at 75 mph (ca. 121 km/h). Typically, this extraordinary bird manifests high-intensity behavior (see article). Lions can sleep more than 14 hours a day; a horse or giraffe can survive on 3-4 hours of sleep. Elephants may need as little as 2 hours of sleep, with REM sleep every 3 or 4 days when lying down. Koalas and bats sleep about 20 hours a day. (See Louis, 2023, pp. 141, 152).
In 1964, Randy Gardner, an American student, deprived himself of sleep for 11 days and 24 minutes. Although this record has been broken, it still shows the same effects: reduced concentration, memory and reflexes, mood swings, and disorientation. Bees display similar behavior after sleep deprivation.


Another form of immobility is sleep. This section shows why this activity, which takes up nearly a third of our time, is so important — perhaps as old as life itself. Sleep is a state of reduced environmental awareness in animals, from worms to humans. It carries some risks. Yet most animals sleep. Of course, we need security to sleep. Giraffes, for example, usually sleep only three hours a night because they are easily attacked. So, does the ventral parasympathetic nervous system create the necessary state of safety while we sleep? Some people require “company” to sleep; others prefer to be alone. Sooner or later, babies and children learn to sleep alone: it means concentration and effective self-regulation.
Sleep-Active Neurons: Conserved Motors of Sleep (Rodent, Zebra, Drosophila) (Bringmann, 2018) shows how sleep is crucial for survival and well-being. This state has been studied in most genetically accessible animal models, including rodents, fish, flies, and worms. Genetic and optogenetic studies have identified several neurons that control sleep. We find a group of neurons that generate sleep across species by directly inhibiting wakefulness. These sleep-active neurons and their regulation by upstream pathways in both vertebrate and invertebrate species suggest common and ancient components that evolved early in the history of nervous systems. However, paradoxical sleep (rapid eye movement, or REM, dreaming), a typical need for many animals, appears to emerge in amniotes.
Penguins snatch seconds-long microsleeps (Harding, 2023). Since penguins can't sleep while incubating eggs, they “steal” seconds of sleep to rest.
Neurophysiology of sleep and wakefulness (Harris, 2005). In this comprehensive presentation, the author describes how the autonomic system shapes the three distinct states: wakefulness, NREM sleep, and REM sleep. Various factors influence the need and timing of sleep onset. These factors affect the NTS, causing its noradrenergic projections to midbrain and forebrain structures to inhibit activity in the ARAS (ascending reticular activating system), resulting in activation of inhibitory GABAergic thalamocortical projections to the cortex.
Sleep in Invertebrates (van Swinderen, 2007, 1.28 in Kaas, 2007. p.451-456), describes phenomena such as quiescence (preparation for sleep), specific postures, micro-behaviors during sleep, and electrophysiology (e.g., crayfish, honeybee, and fruit lies). Sleep is ultimately described as an uncoupling of the brain from external stimuli.

Further Reading
Why sleep is essential for health: a psychoneuroimmunology perspective (Irwin, 2015).
Sleep Health: Reciprocal Regulation of Sleep and Innate Immunity (Irwin, 2017).
Sleep research goes wild: new methods and approaches to investigate the ecology, evolution and functions of sleep (Rattenborg, 2017).
Call it Worm Sleep (Trojanowski, 2016). In this remarkable article, the author tells us how the nematode Caenorhabditis elegans stops eating and moving during a larval transition stage called lethargus and after exposure to cellular stressors. This sleep is similar to circadian-timed sleep in insects and mammals, and sleep in response to cellular stress is similar to disease-induced sleep in other animals. Mammalian and Drosophila sleep share molecular and functional conservation with C. elegans sleep.
Inactivity Is Nycthemeral, Endogenously Generated, Homeostatically Regulated, and Melatonin Modulated in a Free-Living Platyhelminth Flatworm (Osmond, 2017). Sleep evolved early in the evolutionary history of animals. Despite their simplicity, inactive flatworms appear to sleep. Specifically, rest occurs mainly during the day. This basic circadian rhythm persists in constant darkness and is probably endogenously generated.

2.12.7
Is the Dive Reflex the Ultimate Explanation for Sudden Death?
The cessation of breathing (apnea) associated with bradycardia (slow heart rate), the “dive reflex,” deprives the brain and heart of oxygen. According to the PVT, this could be fatal for mammals, as only reptiles with a low metabolism can survive such stress. Is the dorsal vagus responsible for the reflex? Here, we show how the PVT is wrong about the DR (dive reflex).
The DR is a highly conserved behavior in numerous respiratory animals. The best divers among respiratory animals — in depth and duration — are mammals.
The DR is primarily respiratory — not autonomic — and is therefore controlled by specific respiratory centers in the spinal cord.
DR is accompanied by sympathetic and parasympathetic co-activation. In mammals, vagal activation originates in the nucleus ambiguus. The mammalian dorsal vagal motor nucleus (DVMN) doesn't participate.
Maximum diving capacities of the Weddell seal (Kooyman, 1966). This study provides a detailed description of the diving behavior of Weddell seals. Weddell seals' probable maximum diving capacities were determined by observing 959 dives and measuring the depths of 381 dives. The deepest dive was 600 meters; the longest was 43 minutes 20 seconds.
Heart rate regulation in diving sea lions: the vagus nerve rules (Ponganis, 2017). Some marine mammals have morbid arrhythmias secondary to autonomic conflict. The authors describe how:
Sympathetic cardiac accelerator and peripheral vasoconstrictor fibers are activated during diving.
Parasympathetic cardiac tone via the vagus nerve dominates over sympathetic cardiac tone during all phases of diving, resulting in bradycardia.
Heartbeat variations (benign arrhythmias) are common in marine mammals.
he Mammalian Diving Response: Inroads to Its Neural Control (Panneton, 2020). The diving response (DR) in mammals generally consists of cessation of breathing (apnea), a dramatic slowing of the heart rate (bradycardia), and an increase in peripheral vasoconstriction. First observed by Irving and Scholander in the late 1930s, DR is found not only in marine mammals (e.g., seals, whales, and dolphins), but in all mammals studied. The DR allows for conserving vital oxygen stores and directs perfusion to the heart and brain. This article describes the centers and mechanisms involved in this powerful reflex.
Diving physiology of marine mammals and birds: the development of biologging techniques (Williams, 2021). This review describes a brief history of the beginning of field diving physiology investigations, including the invention of the time-depth recorder. It then traces biological studies in four key areas of diving physiology: heart rate, blood flow, body temperature, and oxygen storage management. Investigations of diving heart rate in whales and O2 storage management in diving emperor penguins are highlighted.

Molecularly defined circuits for cardiovascular and cardiopulmonary control (Veerakumar, 2022). Using a mix of technologies including retrograde neuronal tracing, single-cell RNA sequencing, and optogenetics to dissect the cardiac parasympathetic organization of the nucleus ambiguus, the authors identify two parallel circuits. One that selectively controls cardiac function (ACV circuit). Another that coordinates cardiac and pulmonary function (ACP circuit).
The first (ACV) circuit mediates the baroreceptor reflex, slowing heart rate and atrioventricular conduction in response to increased blood pressure.
The second (ACP) circuit is not only related to cardiac neurons (intermingled with ACV neurons), but also mediates the diving reflex, i.e., the simultaneous bradycardia and bronchoconstriction that follows water immersion.
This brilliant article finally ended the suggestion that the mammalian diving reflex could originate in the “dorsal.” Three other articles explore the same questions.
Heart rate at rest and during the diving reflex is set primarily by cardiovagal neurons (CVNs) in the nucleus ambiguus (nAmb). In this article, the authors describe how they identified the molecular underpinnings of this powerful and highly conserved mammalian autonomic reflex.
In Molecular Disambiguation of Heart Rate Control by the Nucleus Ambiguus (Jalil, 2023), the authors come to similar conclusions regarding the production of the diving reflex in the nucleus ambiguus.
Molecular cell types as functional units of the efferent vagus nerve (Coverdell, 2023) completes this molecular disambiguation.
2.12.8
Can humans hibernate? The facts and the story
The phenomenon of hibernation, like the diving reflex, shows that Porges' view of hypometabolism as a reptilian trait is incorrect. Mammals, such as wild bears or marmots, practice hibernation to survive the winter. Animals in zoos don't hibernate; they have plenty of food throughout the winter. As for human hibernation, some anecdotes suggest that peasants in Russia and other Slavic countries practiced a form of hibernation known as "lotska" during the harsh winter months. They are said to have significantly reduced their daily activities and spent much time sleeping, while remaining active and keeping the fire going. Two online articles (see below) express clear doubts.
Can Humans Ever Harness the Power of Hibernation?
Human Hibernation Doesn’t Exist — Yet
Nevertheless, ancestors of us — 500,000 years ago — may have practiced some forms of hibernation, as the following article proposes: Hibernation in hominins from Atapuerca, Spain half a million years ago (Bartsiokas, 2020).

Further reading
Mammalian Hibernation: Cellular and Molecular Responses to Depressed Metabolism and Low Temperature (Carey, 2003). The ability to hibernate is found throughout the class Mammalia and appears to involve the differential expression of genes common to all mammals rather than the induction of novel genes. Hibernation is characterized by prolonged periods of torpor, during which the minimum body temperature can drop as low as 2.9°C and metabolism can be reduced to 1% of euthermic rates. Many global processes use low temperatures to minimize reaction rates, ready to resume total activity upon rewarming. Other critical functions must continue at physiologically relevant levels during torpor, even at body temperatures near 0°C.
Survivable hypothermia or torpor in a wild‐living rat: rare insights broaden our understanding of endothermic physiology (Nowack, 2022). Maintaining a high and stable body temperature is energetically costly for endothermic mammals and birds. Heterothermic species can reduce their energy requirements during energetic shortages through torpor. Observation of a native Australian bush rat shows a 5.5 hour hypothermic bout with a decreased body temperature of 12°C. Heterothermy, the ability to withstand and overcome periods of reduced body temperature, appears to be an evolutionary ancestral trait.
Mammalian Hibernation: Cellular and Molecular Responses to Depressed Metabolism and Low Temperature (Carey, 2003). The ability to hibernate is found throughout the class Mammalia and appears to involve a differential expression of genes common to all mammals rather than the induction of novel gene products unique to the hibernating state. The hibernation season is characterized by extended bouts of torpor, during which minimal body temperature can fall as low as −2.9°C and metabolism can be reduced to 1% of euthermic rates. Many global biochemical and physiological processes exploit low temperatures to lower reaction rates but can resume full activity upon rewarming. Hibernators survive repeated cycles of torpor and arousal during the hibernation season.
Natural hypometabolism during hibernation and daily torpor in mammals (Heldmaier, 2004). Daily torpor and hibernation are drastic measures taken by endotherms to conserve energy. During these states, metabolic rate is suppressed to a fraction of normal metabolism, accompanied by reduced respiration and heart rate. Body temperature gradually decreases to the level of the ambient temperature. Torpid states are interrupted by arousal, where metabolic rate returns to normal levels and a burst of heat production establishes euthermic body temperature. Several cellular mechanisms that contribute to hypometabolism have recently been identified, including cessation of protein synthesis, transcription, and translation. ATP production from glucose and lipids rapidly returns to normal upon arousal. Hibernation and diurnal torpor are also found in small mammals in temperate and tropical climates. This suggests that this behavior is a measure to cope with temporary or seasonal depletion of energy supplies.
The role of energy availability in Mammalian hibernation: a cost-benefit approach (Humphries, 2003). Expression of torpor provides substantial energy conservation, increasing the chance of surviving a food shortage and emerging with residual energy for early spring reproduction. However, all hibernating mammals periodically awaken to euthermic body temperatures during hibernation, presumably to avoid oxidative stress, reduced immune competence, and neuronal tissue damage.
Hibernation and gas exchange (Milsom, 2011). Hibernation in endotherms and ectotherms is characterized by an energy-conserving metabolic depression due to low body temperatures. Gas exchange rates are correspondingly reduced. In hibernating mammals, ventilation decreases even more than metabolic rate, resulting in a relative respiratory acidosis that may contribute to metabolic depression. Breathing becomes episodic in some mammals, and in some small mammals, significant apneic gas exchange may occur by passive diffusion through the airways or skin. Arousal in hibernating mammals results in restoration of euthermic temperature, metabolic rate, and gas exchange. This happens periodically even when ambient temperatures remain low, whereas body temperature, metabolic rate, and gas exchange in hibernating ectotherms are closely linked to ambient temperature.
Regulation of cardiac rhythm in hibernating mammals (Milsom, 1999). The dramatic decrease in heart rate exhibited by hibernating mammals begins before body temperature drops. The decrease in heart rate due to cyclic parasympathetic activation induces skipped beats and regular systoles and slows the steady heartbeat. As body temperature drops, the parasympathetic influence is progressively withdrawn and periods of parasympathetic and sympathetic dominance alternate, resulting in regular periods of arrhythmia (tachycardia followed by bradycardia) and occasional long asystoles or periods of highly irregular cardiac activity. The rise in body temperature is preceded by a dramatic increase in sympathetic activation, followed by a slow withdrawal of sympathetic influence. Arrhythmias are common during natural arousals or shifts from lower to warmer hibernation temperatures.
Human torpor: translating insights from nature into manned deep space expedition (Shi, 2020).In the 1960s, several scientists proposed that an induced state of suppressed metabolism in humans that mimics "hibernation" could solve many spaceflight problems. In recent years, with the introduction of specific methods, it has become increasingly feasible to induce an artificial hibernation-like state (synthetic torpor) in non-hibernating species. Although progress has been made, the precise mechanisms underlying the active regulation of the torpor-arousal transition remain poorly understood. Reduced thermoregulation, relatively low body temperatures, and basal metabolic rates have been documented in marsupials, monotremes, xenarthrans, subterranean rodents, and afrotherians, as well as in some mammals undergoing various forms of heterothermy (e.g., long-term hibernation or daily torpor).
2.12.9
Do We Learn Helplessness, or Did We Fail to Learn Hope?
American psychologist Martin Seligman developed the concept of “learned helplessness” in the late 1960s. Seligman's work built on earlier experiments by Ivan Pavlov, known for his work on classical conditioning, and John B. Watson and B. F. Skinner, key figures in behaviorism.
While conducting experiments on dogs, Seligman and his colleagues first observed that after repeated exposure to an unavoidable negative event (such as an electric shock), the dogs later stopped trying to avoid the shock, even when escape was possible. The researchers suggested that these animals had learned to be helpless, a concept he later extended to human behavior. He proposed that humans could also learn to feel helpless and become passive in the face of traumatic or uncontrollable events, leading to feelings of depression and anxiety. This led to significant developments in cognitive therapy and our understanding of mental illness.
Learned Helplessness: A Theory for the Age of Personal Control (a book by Peterson, Maier, and Seligman, 1993) presents their research on helplessness.
Learned Helplessness at Fifty: Insights from Neuroscience (Maier, Seligman, Martin, 2016). In this article, the authors turn the concept of “learned helplessness” on its head. They explain how the animals didn't learn anything, but on the contrary, they did NOT learn to escape. They describe a neural circuit that allows the ventromedial prefrontal cortex (vMPFC) to control the activity of the dorsal raphe nucleus (in the brainstem), a serotoninergic structure responsible for helplessness.
“In conclusion, the neural circuitry underlying the phenomenon of learned helplessness strongly suggests that helplessness was not learned in the original experiments. Rather, passivity and heightened anxiety are the default mammalian responses to prolonged bad events. What can be learned is cortical–that bad events will be controllable in the future. The top-down circuitry that descends from the ventromedial prefrontal cortex to the dorsal raphe nucleus and other structures acts to inhibit this default”.
The Hope Circuit: A Psychologist's Journey from Helplessness to Optimism (Seligman, 2018). In this autobiography, Seligman recounts how the concept of “learned helplessness” evolved into “the hope circuit.” Seligman developed positive psychology, which promotes a positive approach to mental health.

Other mechanisms: the Habenula.
Seligman and Maier are not alone in suggesting a neurological explanation. Researchers using the zebrafish as a model have discovered another way animals manifest helplessness. It involves the habenula, a pair of small nuclei above the thalamus (Hikosaka, 2008). Together with the pineal gland (so dear to Descartes), it is part of the epiphysis.
Habenula: A Role in Brain State Transitions during Coping Behavior (Fore, 2019) Fore and Yaski.
Zebrafish Tools for Deciphering Habenular Network-Linked Mental Disorders (Bühler, 2021) Bühler and Carl.
Prefrontal cortical control of a brainstem social behavior circuit (Franklin, 2017). The prefrontal cortex helps adapt an organism's behavior to its environment. In behavioral adaptation to social defeat in mice, this study finds neural projections from the medial prefrontal cortex (MPFC) to the dorsal PAG, an area critical for defensive responses. Social defeat caused a weakening of functional connectivity between these two areas, and selective inhibition of these projections mimicked the behavioral effects of social defeat. (It is interesting to compare this study on helplessness with that of Maier, 2016).
2.12.10
The Truth About Depression and the Dorsal Vagus

The Myth: The internet is flooded with testimonials from people feeling trapped in "dorsal shutdown", unable to move forward, make decisions, or reconnect with themselves. Time and again, the dorsal vagus is blamed for this state of despair.
The Reality: Few seem to realize that the dorsal vagus nerve doesn’t generate emotions—it has no feelings at all. Its role is simple: protecting the heart, modulating the immune system, and regulating appetite and digestion.
So, let’s be frank: what does this biological function have to do with the agonizing experience of the human soul?
The Animal Model for Depression
Peter Levine's work is primarily based on observing similar immobilization behaviors in traumatized clients and animals (prey).
The animal model became popular with the rise of psychotraumatology in the 90s.
We have the same problem with dissociation, which the PVT confuses with immobilization after a traumatic shock.
Following Porges, polyvagal authors describe a group of “dorsal” labeled reactions — including depression. For us, this is a true “fleabag.”
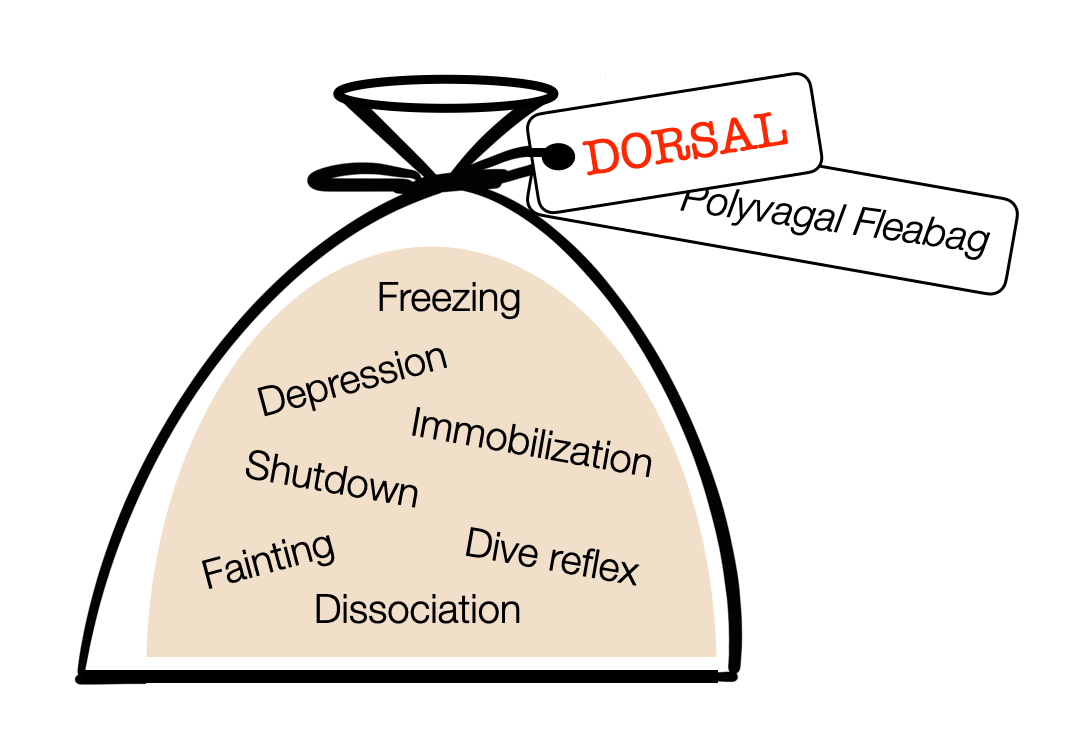

In Animal Models of Major Depression: Drawbacks and Challenges (Planchez, 2019), The authors question the use of animal models to study depression. Although still extremely useful (e.g., in pharmaceutical research), observing the reactions of mice or rats in a maze can't capture the subjectivity of depressed people. That is why psychiatry always makes a double assessment: objective and subjective symptoms.
Labeling these reactions as “dorsal” is a powerful attribution fallacy. Immobilization and freezing are motor patterns commanded by the PAG (midbrain). The sympathetic system shuts down the digestive process. Fainting reaction results from a sudden decrease in sympathetic tone or a ventral vagal response. Dissociation refers to higher (cortical) regions. The diving reflex combines vagal (NAext) and sympathetic activity. The DVMN doesn't participate in any of the responses packaged in the “polyvagal fleabag.”
Serotoninergic hypothesis
Antidepressants block the reuptake of neurotransmitters (serotonin and norepinephrine), thereby increasing the ongoing activity of the synapse. They also support the formation of receptors on the second neuron (neuroplasticity).
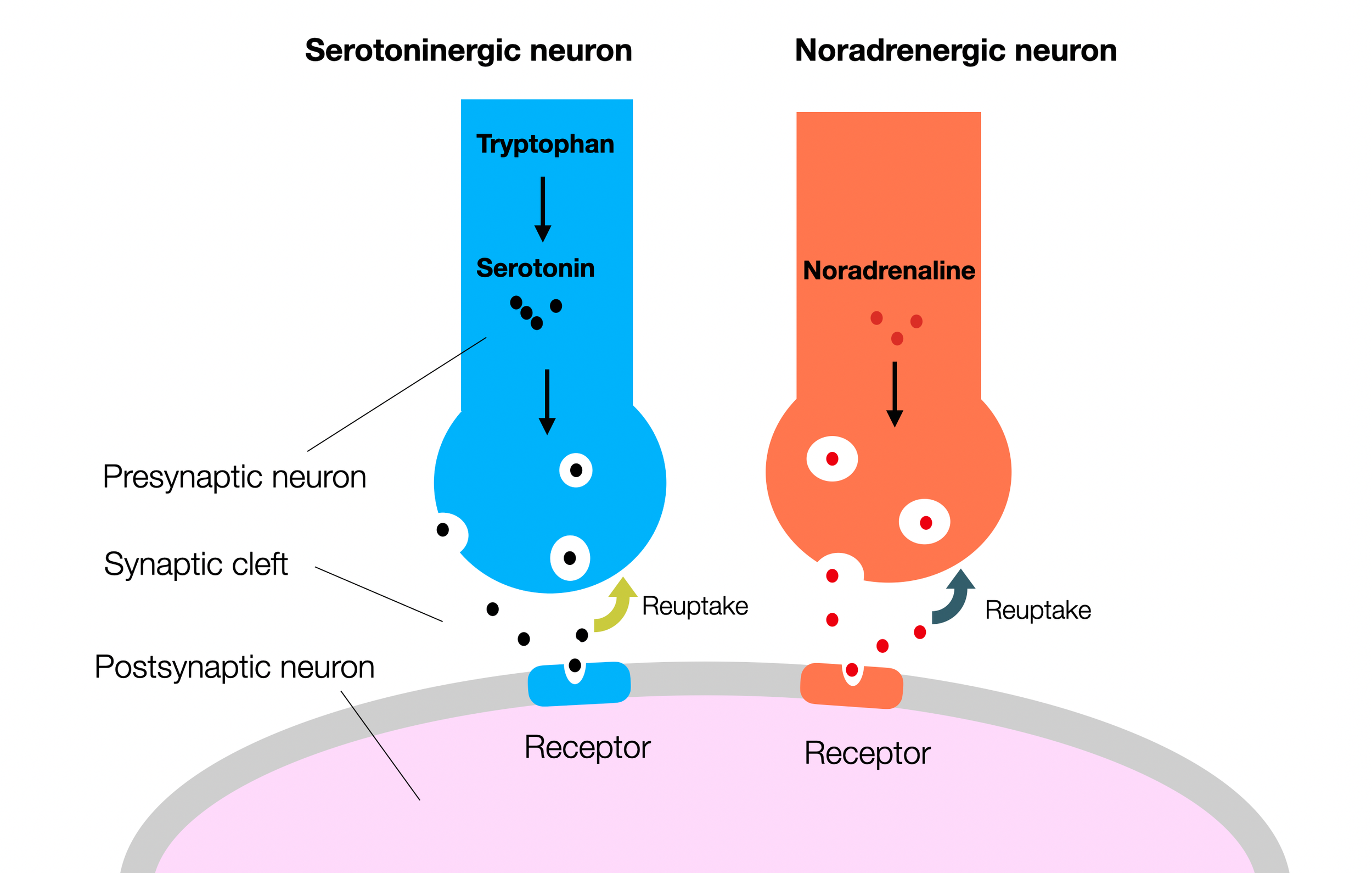
Despite the general distrust of “drugs” (e.g., Servan-Schreiber, 2004), the primary treatment of depression, besides psychotherapy, has been based for decades on the serotonin (Jacobsen, 2012) and norepinephrine hypothesis. Medication blocks the reuptake of these molecules, leaving them available and active in the synaptic cleft (Boschloo, 2023; Moncrieff, 2023; Smith 2023). Recently, researchers have described the relationship between the cortical default mode network (DMN) and rumination, a predominant symptom of depression (Cooney, 2010; Zhou, 2020). Arguing against the polyvagal view is the fact that many depressed patients suffer from sympathetic overdrive, which leads to hypertension (Jacobsen, 2012; Meng, 2012,; Ginty, 2013; Rubio-Guerra, 2013; Ho, 2015; Alhawari, 2022). This overdrive may also explain why HRV is lower in depressed patients, who are indeed under chronic stress (Hage, 2017; Gerber, 2023). However, HRV is not always moderated by a history of depression (Ding, 2023). Moreover, in addition to lower HRV, bipolar patients show inflammatory scores (Hage, 2019). Reduced cortical connectivity — as in dissociation — has been described (Smith, 2015; Huang, 2024). There are also genetic and epigenetic factors (Lin, 2019).
Numerous authors have proposed vagus nerve stimulation for depression (Lv, 2019; Kamel, 2022). Despite encouraging results, the efficacy is still unclear. The mechanism of action is also difficult to understand as the target of this electrical stimulation is still not specific enough.
Touch Medicine (Müller-Oerlinghausern, 2022) shows the importance of skin-to-skin contact in treating depression.
Recognizing the role of the vagus nerve in depression through the microbiota-gut-brain axis (Tan, 2022) describes the importance of a healthy gut microbiome that allows for brain-gut collaboration through the vagal nerve. The use of vagal stimulation is discussed.
Finally, several publications have highlighted the benefits of exercise, which activates the sympathetic system (Contreras, 2022), with caveats (Forys, 2022).
2.12.11
Further Reading about Freezing, Behavioral Arrest, and Quiescence

FREEZING
Threat induces cardiac and metabolic changes that negatively impact survival in flies (Barrios, 2021).
How do fruit flies (Drosophila melanogaster) respond to an unavoidable threat? The authors find cardiac acceleration during running, deceleration during freezing, and increased cardiac pumping, suggesting a mobilization of energy resources. Consistent with this, freezing reduces sugar levels in the hemolymph and makes flies less resistant to starvation. Cardiac responses are absent during spontaneous immobility, reflecting the dynamic nature of the freezing response. This study shows a similarity to vertebrate cardiac responses, contradicting the widely held belief that cardiac slowing during freezing first evolved in vertebrates and is energy-conserving.
Updating freeze: aligning animal and human research (Hagenaars, 2014). Freezing is widely used as a first outcome measure of fear in animal studies. It is also receiving increasing attention in human stress research because of its essential role in the development of psychopathology. Human models of defensive behavior are primarily based on animal models.
Facing freeze: social threat induces bodily freeze in humans (Roelofs, 2010). Freezing is a common defensive response in animals threatened by predators. It is characterized by reduced body motion and decreased heart rate (bradycardia). This study investigates body sway and heart rate in 50 female participants standing on a stabilometric force platform and viewing different faces. Results are similar to those in animal models.
Freeze for action: neurobiological mechanisms in animal and human freezing (Roelofs, 2017). As threat levels increase, animals activate qualitatively different defense modes, including freezing and active fight-or-flight responses. Freezing is a form of behavioral inhibition accompanied by a parasympathetically dominated heart rate slowing (nucleus ambiguus). Fight-or-flight responses are associated with sympathetically driven heart rate acceleration. In rodents, and probably in humans, freezing depends on amygdala projections to the PAG. The author brings confusion by saying, “Freezing is not passive, but a parasympathetic brake on the motor system, relevant to perception and action preparation.” Quoting Porges (2011), he makes the NA responsible for slowing the heart in freezing.
Freezing revisited: coordinated autonomic and central optimization of threat coping. (Roelofs, 2022). Animals have diverse mechanisms for coping with danger. Freezing allows information to be gathered, response possibilities to be previsioned, and preparations for worst-case fight or flight to be made. Rather than a passive fear state, freezing helps to optimize sensory processing and action preparation. Noradrenaline and acetylcholine are critical for these functions.
BEHAVIORAL ARREST
Neural circuitry for behavioural arrest (Roseberry, 2017). This article examines a fundamental requirement for animal survival: the ability to stop movement. This behavioral arrest involves multiple forebrain pathways that ultimately lead to brainstem motor centers.
Not moving: the fundamental but neglected motor function (Noorani, 2017). The motor system's function in preventing rather than initiating movement is often overlooked. These periods of immobility are not just a matter of inhibition and relaxation; they require almost as much energy as when we are moving and place just as many demands on the central nervous system to control their performance. There are different types of standstill and rest.
Inhibition, Not Excitation, Drives Rhythmic Whisking (Deschênes, 2016).
Watch also on YouTube: Rat Whisking Behavior.SWIM STRESS AND ANALGESIA
Stress-induced muscle and cutaneous hyperalgesia: Differential effect of milnacipran (Suarez-Roca, 2006)
Role of -opioid and NMDA receptors in the development and maintenance of repeated swim stress-induced thermal hyperalgesia (Suarez-Roca, 2006)
Neural circuitry for behavioural arrest (Roseberry, 2017). An animal will stop an ongoing movement for various reasons, including reaching a goal, freezing during a perceived threat, and stopping to evaluate a surprising stimulus. Each time, the animal stops the current action, such as locomotion or grooming, and enters postural control. This requires coordination between the neural signals that prevent the current action and those that enable and maintain postural control without movement. No single circuit but several neural systems in the brain are responsible for movement termination.
Cellular Mechanisms and Regulation of Quiescence (Marescal, 2020) describes quiescence as reversible proliferative arrest in which cells are not actively dividing. Yet, they can reenter the cell cycle upon receiving an appropriate stimulus. Quiescent cells are remarkably diverse—they reside in different locations throughout the body, serve distinct roles, and are activated by various signals.
Inactivity Is Nycthemeral, Endogenously Generated, Homeostatically Regulated, and Melatonin Modulated in a Free-Living Platyhelminth Flatworm (Omond, 2017). Despite this simplicity, inactive flatworms appear to be sleeping. Specifically, quiescence is organized in a circadian manner, occurring mainly during the daytime. This basic rhythm persists under constant darkness, suggesting it is endogenously generated. This study reinforces the idea that sleep evolved early in the evolutionary history of animals.
>> next chapter A Polyvagal Theory of Emotions