2.7.0
Sympathetic Nervous System:
What PVT Gets Wrong?
The sympathetic nervous system is a branch of the autonomic nervous system, often associated with the body's “fight or flight” response. It also:
Activates physiological changes such as increased heart rate, dilated pupils, and energy mobilization to prepare the body for rapid action.
Plays a critical role in the response to inflammation (anti-inflammatory reflex) and exposure to cold (thermogenesis). It works through a network of nerves that release neurotransmitters such as norepinephrine and epinephrine.
Counterbalances the parasympathetic system, which promotes “rest and digest” activities.

The sympathetic system triggers pupil dilation, creating this fascinating look: la Bella Donna.
2.7.1
Defining the Sympathetic System
Sympathy comes from a Greek compound word (Sym συμ = together, and Pathos παθοσ = experience, feeling, suffering, and disease) (Bailly, 1950). Like empathy, sympathy expresses the human response to the pain of others and our response to the adversities of life.

When we speak of the “sympathetic system,” we must distinguish three categories:
The term “sympathetic system” describes a specific group of paravertebral nerves that leave the spine at the thoracic and lumbar levels. They have a short preganglionic (cholinergic) and a long postganglionic (adrenergic) part.
Postganglionic sympathetic fibers activate adrenergic receptors on target organs (such as the heart). They are part of a larger group, the “adrenergic system,” which includes peripheral and central adrenergic centers-the locus coeruleus (LC) in the upper medulla (brain activation), the blood pressure centers (RVLM and CVLM), and the higher structures (e.g., hypothalamus) that modulate movement and fight-flight situations.
Ergotropic (Greek: toward work or effort). Not all sympathetic or adrenergic activity is related to fight-flight situations or exercise. The sympathetic system has regulatory functions such as thermoregulation and the anti-inflammatory reflex. The adrenergic system is critical for cognitive work (concentration), social interactions, and exploration.
2.7.2
Anatomy of the Sympathetic Nervous System
“Sympathetic” and “parasympathetic” belong to anatomy and physiology. On the one hand, there are anatomical neural structures (Greek: anatemno ανατεμνω = cut up), nerves that the anatomist patiently dissects with his scalpel. It is dead matter. Conversely, the physiologist observes living processes: the sympathetic system activates, energizes, and engages, while the parasympathetic system calms the body and promotes rest and digestion. However, as we can see, the sympathetic system also has other functions.
The first observations of the autonomic nervous system were made by Galen (129-201) (Nilsson, 1983) while dissecting pigs. He was fascinated by the beaded collars on either side of the spine. Galen, a Greek physician who lived in Pergame (Turkey) and Rome and treated several emperors, described the nerves as hollow tubes through which the animal spirit could circulate from one organ to another, creating what he called “sympathy. The term was then adopted by Winslow (1732) to describe the human paravertebral ganglionic chains. But it was Walter Cannon (1871-1945), an American physiologist, who made the most valuable contribution when he described the sympathetic system as supporting the fight-or-flight response.
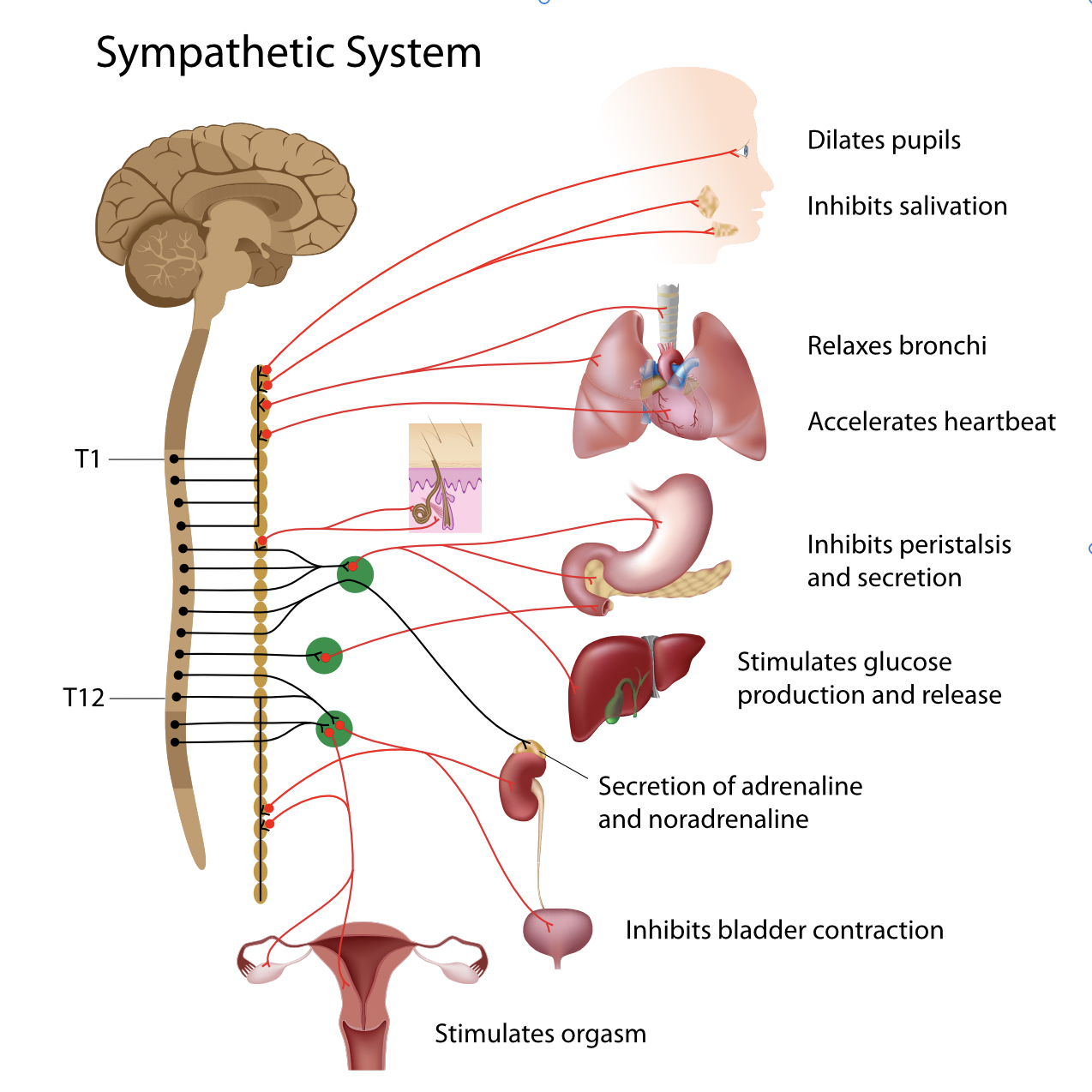
Schematic representation of the sympathetic nervous system. We see one of the two columns formed by the lateral ganglia parallel to the spinal cord, where pre- and postganglionic neurons connect (synapses).
2.7.3
The Curse of Cannon’s «Fight and Flight»
In the 1920s, Cannon proposed the concept of the “fight or flight” response to describe the sympathetic system's responses. He noticed that when animals are faced with a threat, they respond in a way that prepares them to either “fight” (fight the threat) or “flight” (run away from the threat). While Cannon focused primarily on acute physical stressors in developing this concept, it has since been expanded to include responses to chronic physical and psychological stressors.
This response is triggered by releasing hormones from the adrenal medulla, part of the adrenal glands above the kidneys. These hormones, including adrenaline (epinephrine) and norepinephrine (noradrenaline), prepare the body for intense physical activity. This preparation includes increased heart rate, blood pressure, sweating, metabolic rate, dilated pupils, increased blood sugar (for muscle function), and slowed digestion.
While the “fight-flight” concept coined by Cannon helps the reader understand what the sympathetic system stands for, the dramatic description forever puts it in the danger zone. Then, it quickly takes place in Porges' narrative, with the villain posing as the protagonist in a trilogy worthy of Sergio Leone's classic western “The Good, the Bad, and the Ugly” (1966).

2.7.4
Catecholamines from an evolutionary perspective
Over the last 2 billion years of Earth's existence, life developed an important strategy for hacking the sun's energy: chlorophyll. Chlorophyll gives plants the ability to absorb that energy and turn it into grass or leaves—food for herbivores. But soon, herbivores became food for carnivores. Hunting was born. Both had territories to defend, and males competing for social dominance needed fighting. Evolution selected biochemical ways to increase muscle strength, heart pumping, and breathing on demand.
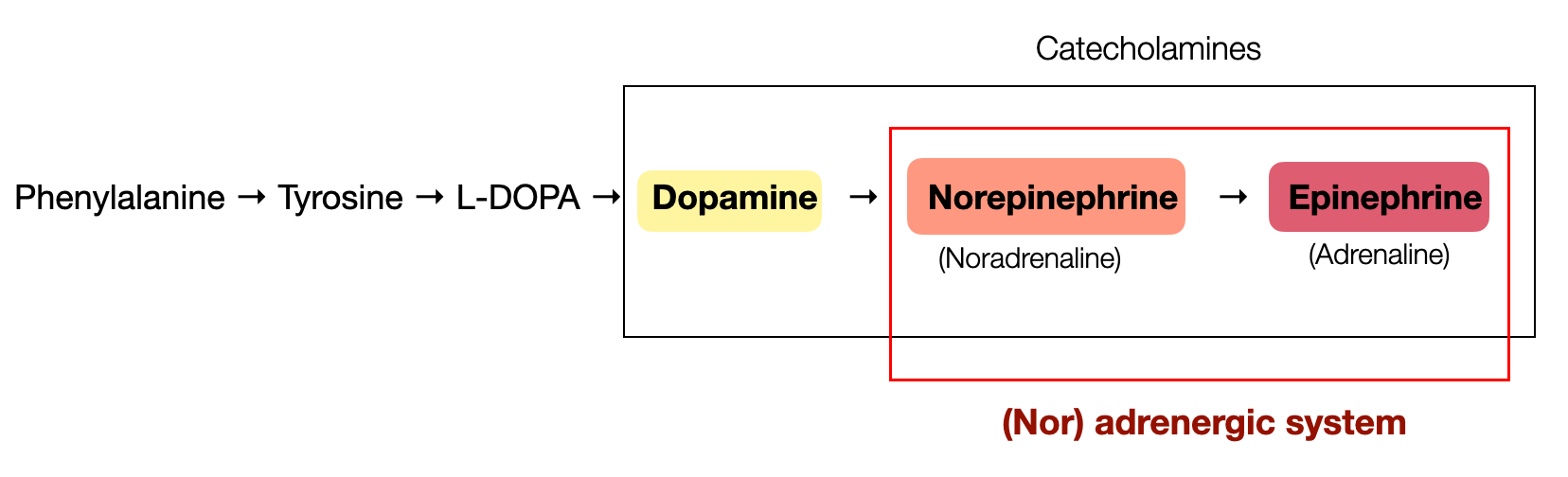
At the basis of this enhancement are catecholamines, a specific class of neurotransmitters and hormones, including:
Dopamine
Norepinephrine (noradrenaline)
Epinephrine (adrenaline)
Dopamine appeared first in evolution, followed by norepinephrine, and finally epinephrine. Biosynthesis follows a similar pattern, starting with amino acids and then hormones (see below).
N.B. For trademark reasons, the term adrenaline (Latin: ad-renal = beside the kidneys) has been replaced in the U.S. by the word epinephrine (Greek: epi + nephron = above + kidneys). Adrenaline is mainly produced in the medulla of the adrenal glands.
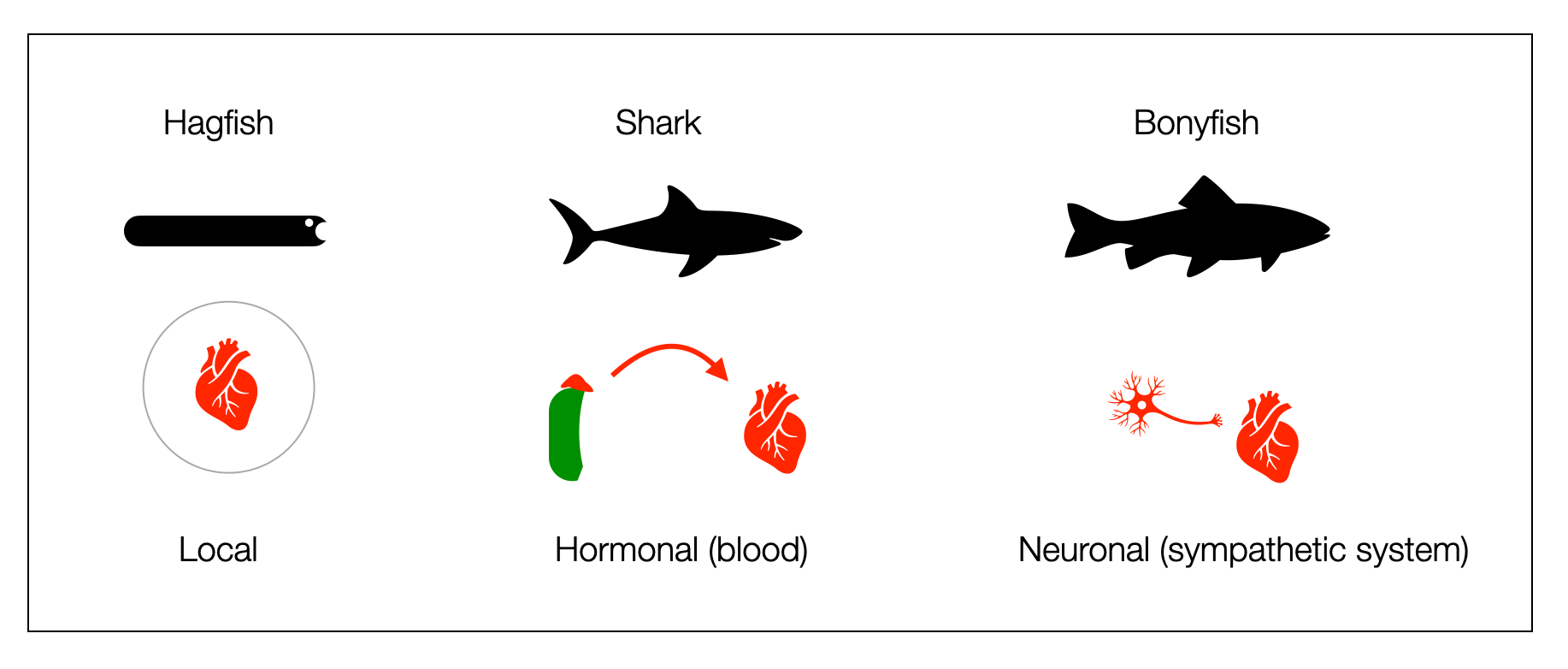
Three forms of production and distribution of adrenaline to the heart exist in marine vertebrates. Does it prove the concept of “ladder?”
Local production. The “earliest vertebrates,” such as hagfish, produce and store adrenaline in the heart (endocrine) to boost cardiac function when needed.
Hormonal distribution. In cartilaginous fish (e.g., sharks), catecholamines produced in the adrenal glands are distributed to the heart through the bloodstream.
Neural distribution. From bony fishes onward, vertebrates have a dual autonomic system (sympathetic + parasympathetic).
This polyvagal hierarchy has a problem: hagfishes, sharks, and bony fish are not ancestors of each other. They appeared about the same time in evolution. Taylor (2014) doesn’t exclude the possibility of a loss of function in the elasmobranchs (e.g., sharks).
2.7.5
How Old is the Sympathetic System?
Porges (2012) postulates a phylogenetic hierarchy of the autonomic system. Dana (2016) even gives numbers for her “ladder” perspective: 500 mya for the dorsal, 400 for the sympathetic, and 200 for the ventral system. This topic needs to be treated with caution, as no direct highway has run through the last 600 billion years-evolution is, as always, tinkering. As we saw in a previous chapter, neither sharks nor hagfish are our ancestors.
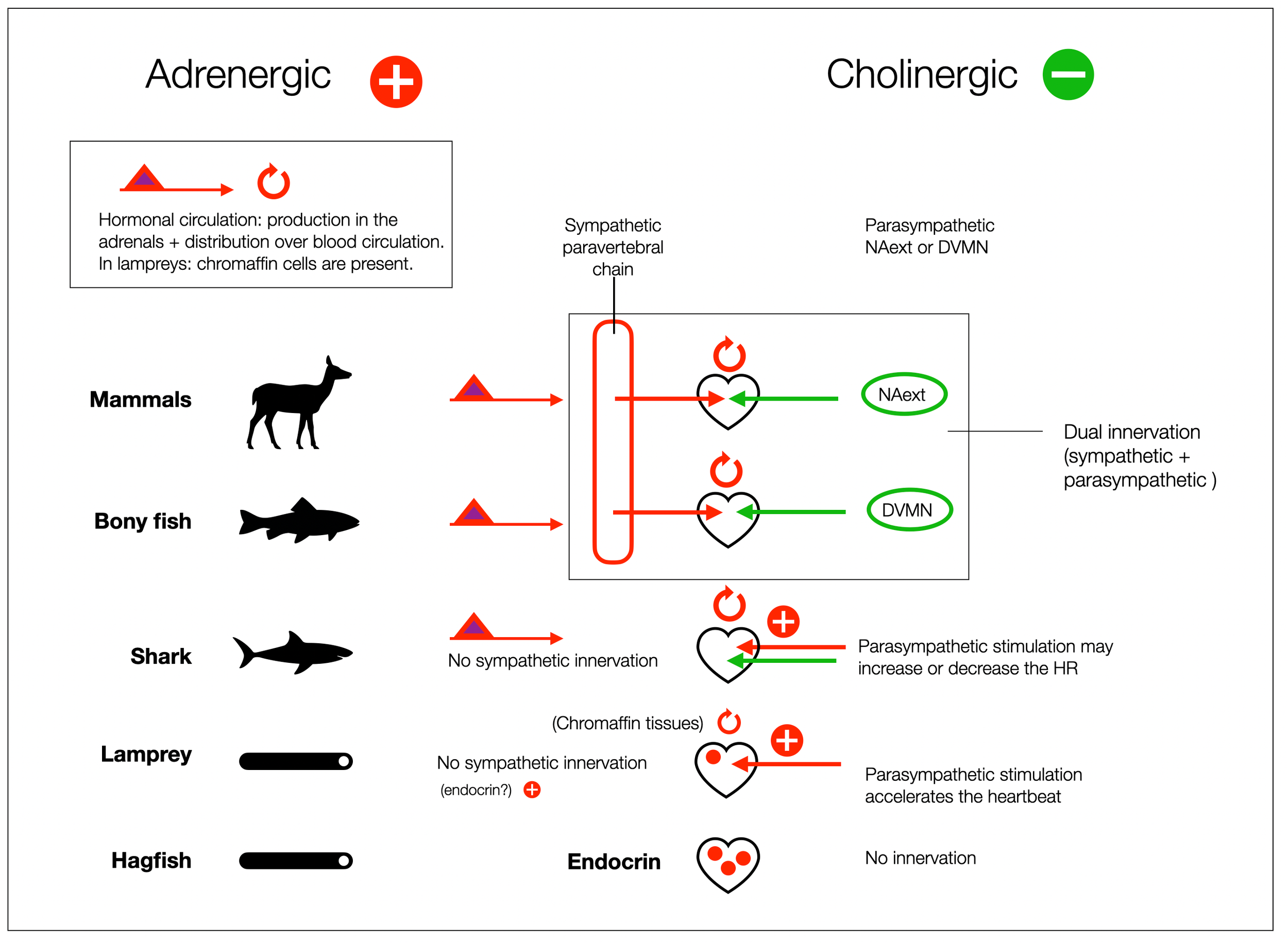
Paradoxical physiological effect. In lampreys, the parasympathetic system accelerates the heart (nicotinic receptors). Vagal stimulation in dogfish “drives both slower and faster heart rates” (Taylor, 2006). (Diagram @absb, 2024).
In Cardiac vagal preganglionic neurones : An update (Gourine, 2016), the authors write: “The parasympathetic nervous system develops first in the evolution of the vertebrates with the sympathetic division appearing relatively late” …”Therefore, the DVMN is probably the most (evolutionary) ancient CNS structure which harbours autonomic neurones in vertebrates.”
The previous diagram (based on Taylor, 2014; Nilsson, 1998; and Haverinen, 2014) offers a more nuanced view. From an anatomical perspective, the vagal innervation of the heart (cranial nerve X) precedes the sympathetic innervation, which in cartilaginous fishes (e.g., sharks) doesn't extend to the heart. “This condition is unique among jawed vertebrates, and may be either an ancestral trait or the result of secondary loss”(Taylor, 2014). However, these fish have adrenergic solid control of the heart through hormones produced by the adrenal glands. In addition, hagfish, which have no cardiac innervation, have abundant storage of adrenaline and noradrenaline in the heart, which allows for vigorous performance (Nilsson, 1998).
According to (Häming, 2011), lampreys are genetically ready to develop a sympathetic system. However, the neural crest cells do not migrate ventrally to form sympathetic ganglia — as they do in jawed vertebrates).
Talking about the adrenergic system instead of the sympathetic system puts the discussion into a different focus. Why would evolution evolve a brake without an accelerator? The fast pace of the intrinsic cardiac system is insufficient for the high energy demands of the heart. Haven't vertebrates already overtaken ancient biological mechanisms — since the bilaterians or even before?
Reading the literature on this subject shows how difficult it is to establish a phylogenetic order.
Thompson et al. (1997) write, “These findings strongly suggest that acetylcholine mediates a localized release of a catecholamine via muscarinic cholinoceptors in shark heart. A cholinergically controlled catecholamine store has been proposed (cholinergic-adreno complex), implying that elasmobranchs may be capable of finer control of cardiac output than has previously been suspected. This complex may represent a transitional adrenergic state between humoral and neuronal regulation.”
Haverinen et al. (2014) describe how hagfish and lampreys modulate cardiac contraction by varying the length of the action potential.
2.7.6
Adrenergic is more than Sympathetic

Adrenergic and cholinergic receptors, which support sympathetic and parasympathetic activity, are ancient, perhaps predating the appearance of the bilaterians. While “sympathetic” initially relates to a peripheral anatomical structure, “adrenergic” addresses a molecular process– how some synapses use adrenergic receptors to transmit information. Therefore, “adrenergic” is more than “sympathetic.”
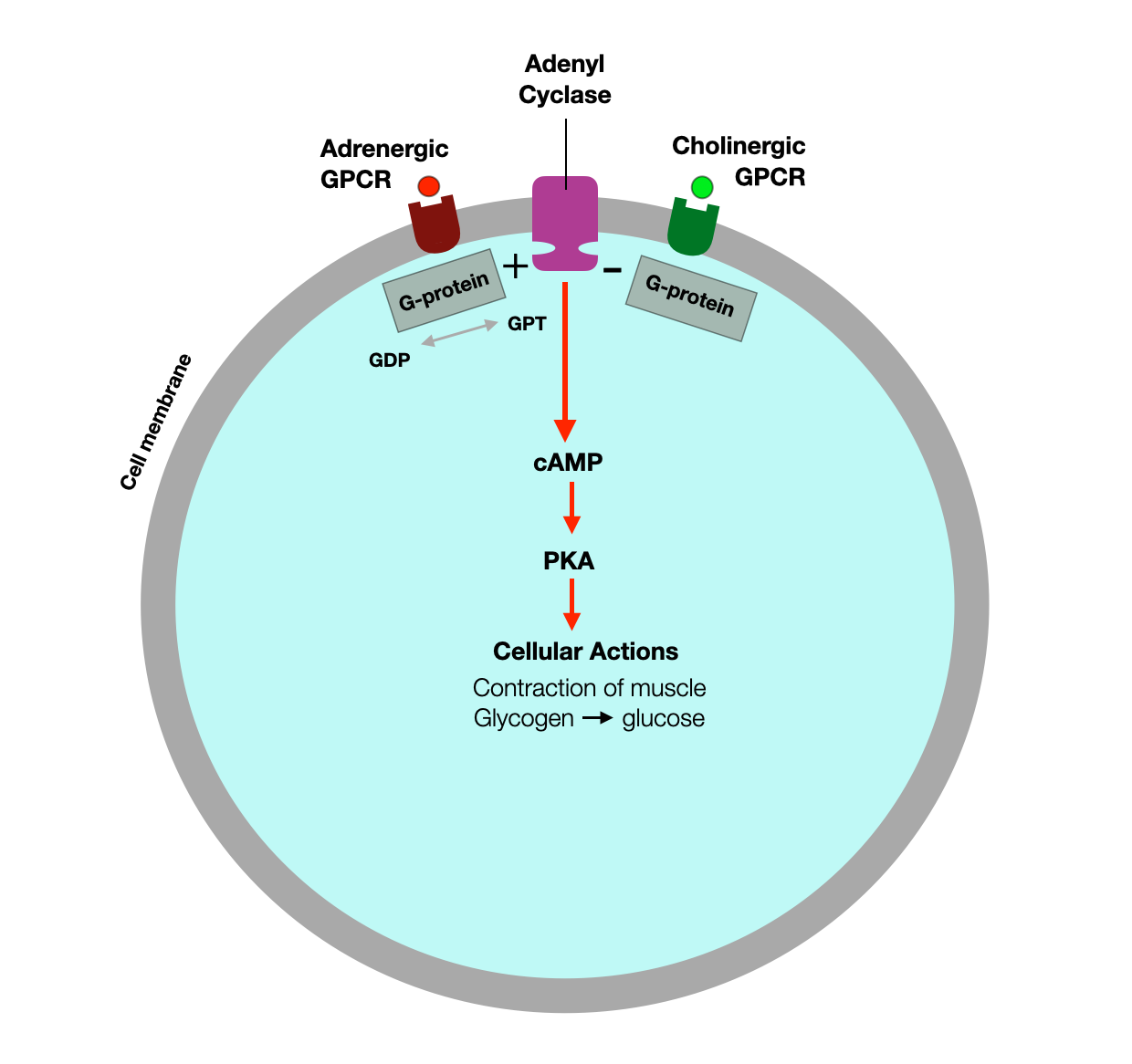
The following diagram shows how specific receptors nested in the cell membrane respond to molecules (ligands) and initiate (or block) a signaling cascade.
Adrenaline (epinephrine) activates the G-coupled adrenergic receptors, leading to the activation of adenyl cyclase. This enhances the conversion of ATP to cAMP, which then activates PKA. This signaling cascade has several downstream effects, including regulation of carbohydrate metabolism and muscle contraction (e.g., heart).
The cell also has regulatory systems. For example, in the heart, vagal fibers activate cholinergic (muscarinic) G protein-coupled receptors (GPCRs), reducing cAMP amplification and PKA activation.

In his phylogenetic description, Porges places the SNS in an intermediate position between the “old” and “new” vagal systems. As seen above, comparative anatomy agrees with Porges. Parasympathetic innervation appears in elasmobranchs (cartilaginous fish such as sharks and rays), and sympathetic innervation in bony fish.
However, the adrenergic system is much more than the sympathetic system. Any situation that requires alertness, concentration, or large amounts of energy activates a wide range of adrenergic circuits:
The central adrenergic system originates in the pons (locus coeruleus); it delivers noradrenaline to specific destinations in the brain and small brain. It is essential for concentration and focus.
The sympathetic system in the dorsal and lumbar regions (noradrenaline) is primarily responsible for cardiovascular and pulmonary functions.
The adrenal glands act as a large sympathetic ganglion, flooding the circulation with epinephrine and norepinephrine.
The adrenergic system refers to the network of cells, receptors, and signaling mechanisms that use catecholamines (epinephrine and norepinephrine). These receptors, classified as alpha (α1, α2) and beta (β1, β2, β3) receptors, are located in various tissues and organs and mediate the effects of catecholamines.
Adrenergic acceleration is as old as cholinergic deceleration. Adrenaline (epinephrine) and acetylcholine are ancient molecules with an evolutionary history dating back to single-celled organisms (see below).
Monoaminergic (dopamine, serotonin, and norepinephrine) neurotransmission is essential for neocortical functions such as perception, cognition, and motivation. The locus coeruleus is connected to the executive control network (ECN) (Bär, 2016).
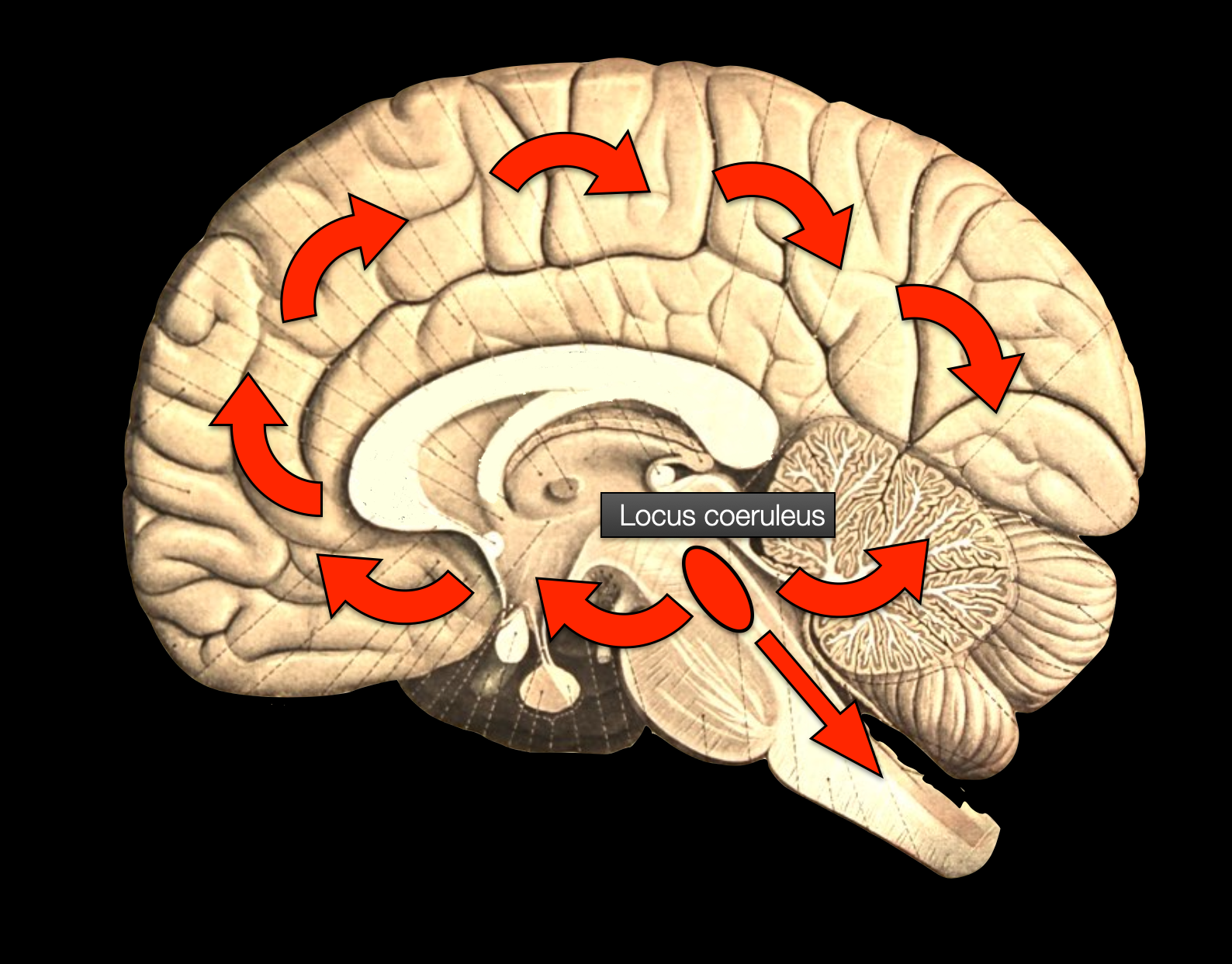
According to Neuroscience: Exploring the Brain, the central adrenergic network originates in the locus coeruleus and distributes throughout the brain and cerebellum. (Bear, 2016) (Diagram ©absb, 2014, with Brain background from Sobotta, 1922).
Adrenergic neurons can also directly modulate the activity of vagal neurons at the presynaptic level (Fedel, 2020).
The two populations of neurons also appear to share a common ancestor (idem, more at the end of this chapter).
More info about adrenergic receptor and G-protein signaling

These YouTube videos explain how adrenergic receptors work.
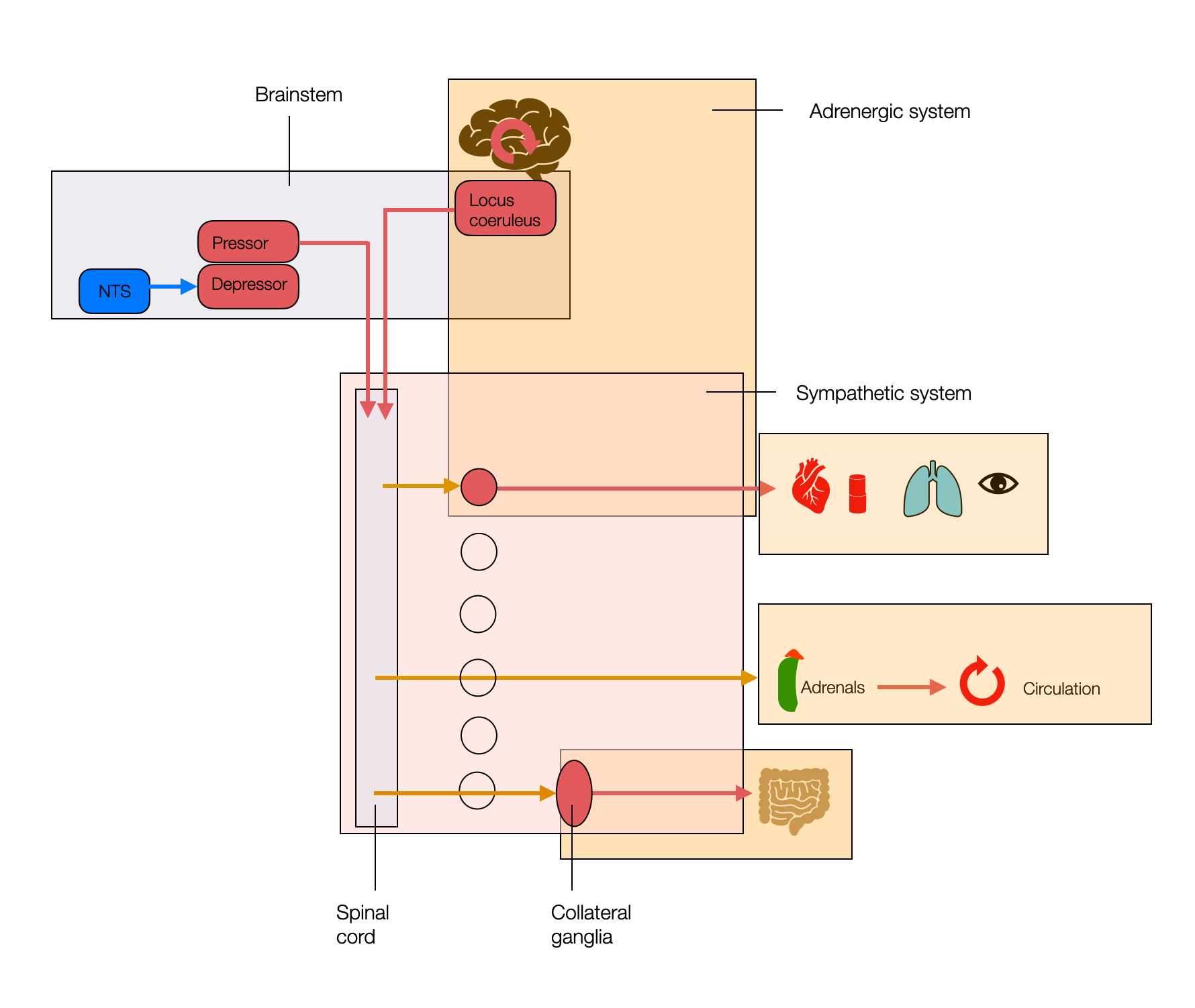
This simplified diagram shows the ambiguity of the sympathetic system. The adrenergic and sympathetic (SNS) systems don't overlap. This may be important as we discuss the phylogeny of the SNS. “Sympathetic” is an anatomical concept, while “adrenergic” is a functional, biochemical concept. The sympathetic system performs half its functions through cholinergic (nicotinic) receptors.
The locus coeruleus in the brainstem involves many cognitive functions (e.g., memory, attention, or alertness) that aren't directly related to stress and threat situations. Unfortunately, this is not part of the polyvagal reflection because the PVT mostly ignores higher centers.
This schematic representation of the sympathetic system (@absb, 2024) also shows one of the two columns (sympathetic chain) formed by the lateral ganglia parallel to the spinal cord, where pre- and postganglionic neurons connect (synapses).
2.7.7
What Was First? Challenging the Hierarchical Polyvagal View
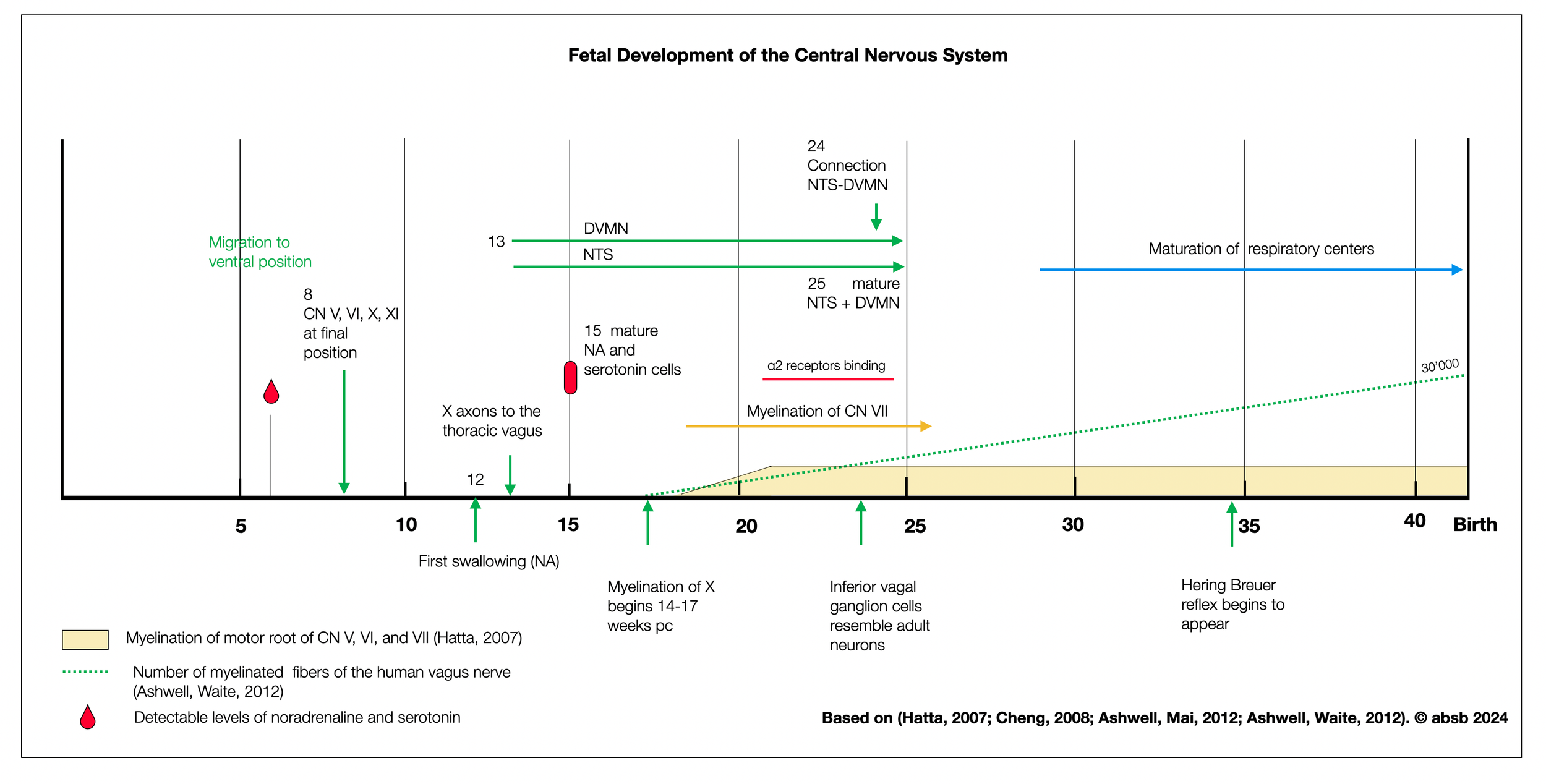
The diagram below shows the fetal development of the central nervous system. The thoracic vagus, NTS, and DVMN begin to develop somewhat simultaneously (13th week). There is no clear hierarchical appearance as postulated by the PVT, except for myelination, which starts around 17 weeks for all cranial nerves (except the dorsal vagal).
2.7.8
Metabolism and Digestion: Role of the Sympathetic
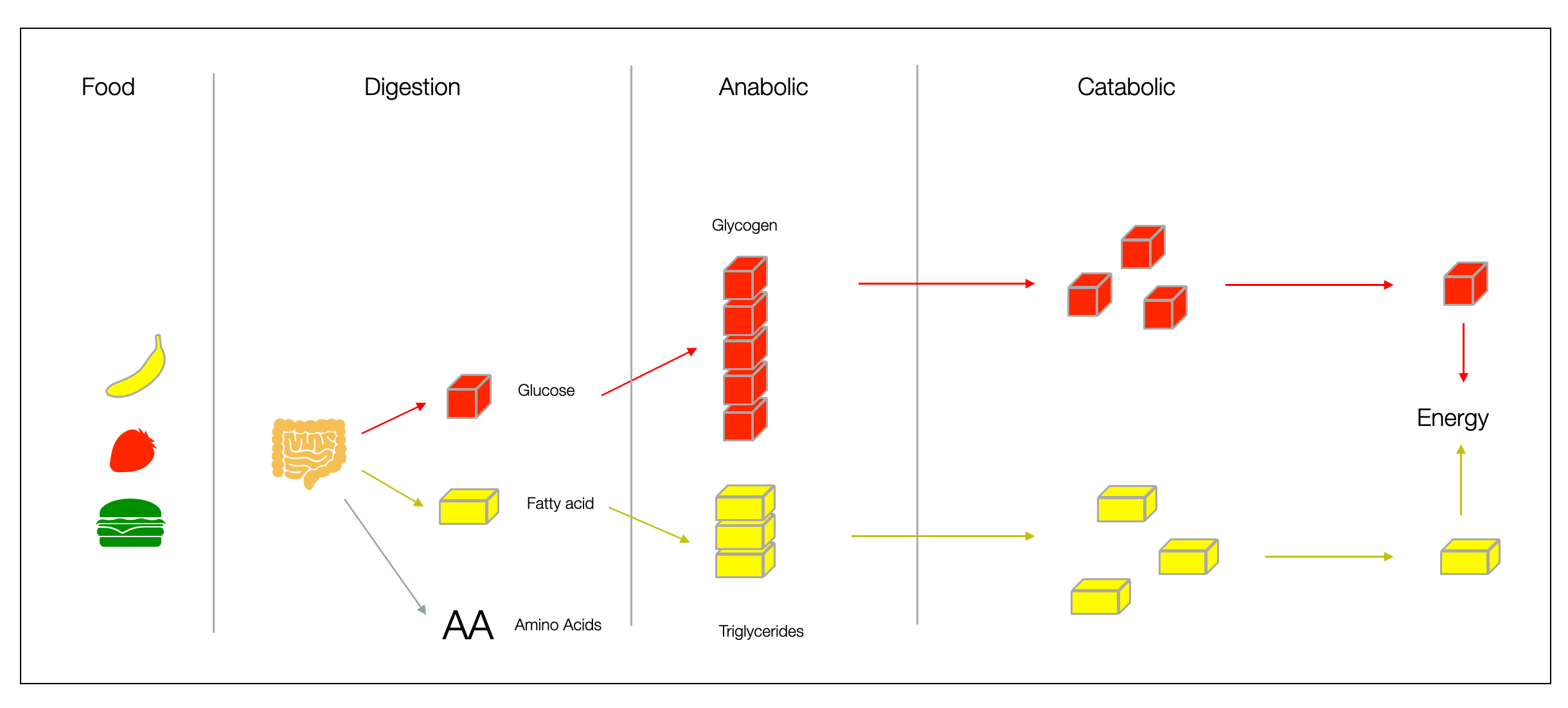
When we eat food, our digestive system breaks it into molecules (such as glucose and fatty acids) that can be easily absorbed and converted into biochemical energy (ATP). Cells also have mechanisms to store these molecules in chains, such as glucose in glycogen in the liver or fatty acids in triglycerides (body fat tissue). Norepinephrine and epinephrine can rapidly break down energy stores into small molecules that mitochondria convert into ATP, the primary energy currency. More in (Vasudevan, 2018).
The sympathetic system helps regulate food intake and increases “fat burning.” It interacts with leptin and insulin — two critical hormones for weight regulation.
In general, adrenaline and noradrenaline are catabolic: they break down and “burn.” The opposite effect is “anabolic,” a term you may recognize from muscle-building product advertisements.
2.7.9
What Polyvagal Theory Gets Wrong About the Sympathetic Nervous System?
PVT gives the sympathetic nervous system a bad name. Is it that bad? An examination of PVT's postulations about the SNS revealed ten major errors, which we will describe in the following sections.
a. Why Maintaining the Negative Reputation of the Sympathetic System?
Following Cannon's description (“fight-flight”), Porges maintains the shift from a generalized “energetic” or “effort-based” perspective to a “threat-centered” or “defensive” perspective. This conveys that the body isn't just a system that prepares for any form of effort (e.g., standing up, climbing stairs, carrying a heavy bag), but is specifically tuned to respond to perceived threats. This dysphoric negative approach is unrealistic because it excludes the SNS from everyday life. It is “stress-centered” or “alarm-based. In reality, the SNS is present in many pleasurable activities, such as dancing, sex, watching movies, and all kinds of sports.
In his comprehensive book Adrenaline, Hoffman (2013) recounts how acetylcholine and adrenaline (epinephrine) were discovered. He spends several pages praising adrenaline.
In the chapter The Significance of Stillness, in Polyvagal Safety (Porges, 2021. p.232), Porges explains: «Most people conceptualize the sympathetic nervous system as a fight-and-flight system. However, (…) it has other more prosocial functions and supports energetic activities. Without a well-functioning sympathetic nervous system, we would be lethargic. Our exuberance and upbeat movements and feelings depend on the sympathetic nervous system as an energy source, whereas, in general, the Parasympathetic nervous system is about repose and support of health, growth, and restoration.»
Such an explanation could make us happy. But these are just casual lines in an interview, not part of an article. Nevertheless, the PVT remains on the rails of a hierarchical conceptualization.
b. "The Benefits of Exercise and Movement: How the Sympathetic Nervous System Helps
Positive spiritual value: To support his epic trilogy, Porges limits the role of the sympathetic to emergencies, suggesting a perfect world without sympathetic participation. This is unfortunate since the sympathetic, besides causing harm, has healing qualities. This is all thanks to Peter Levine, who, in Waking the Tiger (Levine, 1997), correctly showed how a vigorous mobilization can heal the trauma. Various psychiatrists have since recognized that exertion, such as running, can significantly help depressed patients.

Exercise training preserves vagal preganglionic neurones and restores parasympathetic tonus in heart failure (Ichige, 2016). Treadmill training increases physical performance and normalizes autonomic tonus, and IHR restores the number of DMV and NA neurons and corrects ChATir without affecting ventricular function. There are strong positive correlations between parasympathetic tonus and ChATir in NA and DMV. RVLM DBHir is also normalized by training, but there was no change in neuron number and no correlation with sympathetic tonus. Training-induced preservation of preganglionic vagal neurons is critical for normalizing parasympathetic activity and restoring autonomic balance to the heart in persistent cardiac dysfunction.
Untapping the Health Enhancing Potential of Vigorous Intermittent Lifestyle Physical Activity (VILPA) (Stamatakis, 2021). Recently revised public health guidelines recognize the benefits of regular bouts of Vigorous Intermittent Lifestyle Physical Activity (VILPA), such as carrying groceries, walking uphill, and climbing stairs. This article reviews current research to understand better and promote feasible VILPA interventions.
Hormesis
In Browning of the white adipose tissue regulation (Machado, 2022), the authors describe how our white adipose tissue (WAT) can transform into beige adipose tissue (halfway to brown tissue), enhancing its non-shivering thermogenesis and metabolic capacity. This “WAT browning” is a highly dynamic process that is influenced by several factors, including temperature (cold and hot), physical activity, thyroid hormones, circadian rhythms, food components, and dietary regimens (e.g., intermittent fasting).
Neurocognitive and Somatic Components of Temperature Increases during g-Tummo Meditation: Legend and Reality (Kozhevnikov, 2013).
Untapping the Health Enhancing Potential of Vigorous Intermittent Lifestyle Physical Activity (Stamatakis, 2021)
Vagal determinants of exercise capacity (Machhada, 2017).
Effects of Physical Exercise on Executive Function in Adults with Depression: A Systematic Review and Meta-Analysis (Contreras-Osorio, 2022).
Can physical activity improve the mental health of older adults? (Lautenschlager, 2004).

Vigorous Intermittent Lifestyle Physical Activity and Cancer Incidence Among Nonexercising Adults (Stamatakis, 2023). Previous studies have shown that small doses of intermittent VPA (Vigorous Physical Activity), such as fast stair climbing, rapidly improve cardiorespiratory fitness. Researchers using wearable devices (accelerometers) to measure subjects' daily efforts have found that more VPA is also explicitly associated with a reduced risk of breast, endometrial, and colon cancer. The primary biological factors in cancer incidence are inflammation, insulin resistance, and endogenous sex hormones. Four to five minutes of daily exercise significantly reduces cancer risk. At least 3.4-3.6 minutes and 4.5 minutes per day are associated with a 17-18% and 31-32% reduction in total cancer risk, respectively.
Even in the Wild, Mice Run on Wheels reports a study by Dutch searcher Meijer (+video). In Wheel Running in the Wild (2014), Meijer shows that wild mice were attracted to running on wheels, even without a reward.
This study is exciting because it shows that mobilization is vital for some mammals, regardless of the threat.
c. Hormesis and the Cold: How does the Sympathetic help?
The Polyvagal Theory is soft. It's about security. Is that a good purpose for your life? What about risk, challenge, and exposure?
Wim Hof, the Ice Man (Hof, 2017, 2020) (Carney, 2017) introduces an interesting concept: hormesis. From the same Greek root as a hormone, hormone ορμαο means to excite or whip. We will see below how the sympathetic system positively activates the immune system. Another effect is seen on a more general level, helping brave swimmers in the cold become more resilient, brave, and strong. Hormesis has definite psychological benefits.
Browning of white adipose tissue regulation (Machado, 2022) describes how our white adipose tissue (WAT) can transform into beige adipose tissue (halfway to brown tissue), increasing its non-shivering thermogenesis and metabolic capacity. This “WAT browning” is a highly dynamic process that is influenced by several factors, including temperature (cold and hot), exercise, thyroid hormones, circadian rhythms, food components, and dietary regimens (e.g., intermittent fasting).
Neurocognitive and Somatic Components of Temperature Increases during g-Tummo Meditation: Legend and Reality (Kozhevnikov, 2013).

d. The Critical Role of the Sympathetic System in Immune Responses
Parallel to the autonomic system, the immune system is a mighty task force. The modulation of immune processes (e.g., inflammation) is highly complex and delicate. In cooperation with the dorsal vagal complex (NTS and DVMN), the sympathetic system controls inflammatory processes (e.g., autoimmune diseases). The healing power of “cold” (e.g., cold baths) has been proven in scientifically controlled studies by Wim Hof (Hof, 2017, 2020).

Further reading
The Carotid Body Detects Circulating Tumor Necrosis Factor-Alpha to Activate a Sympathetic Anti-Inflammatory Reflex (Katayama, 2021). Recent evidence suggests that the carotid bodies act as immunological sensors, detecting pro-inflammatory mediators (TNF-α) and signaling through the NTS to the central nervous system, which in turn activates the rostral ventrolateral medulla (RVLM), where most pre-sympathetic neurons are located. This triggers a counteracting sympathetic anti-inflammatory mechanism.
Adrenal sympathetic nerve mediated the anti-inflammatory effect of electroacupuncture at ST25 acupoint on a rat model of sepsis (Zhang, 2022). The adrenal medulla is a major source of catecholamines, including norepinephrine (NE), epinephrine (E), and dopamine (DA). There is considerable evidence that NE acts as a neurotransmitter and is involved in the modulation of the inflammatory response in a concentration-dependent manner.
Voluntary activation of the sympathetic nervous system and attenuation of the innate immune response in humans (Kox, 2014).
Vagal afferent activation suppresses systemic inflammation via the splanchnic anti-inflammatory pathway (Komegae, 2018).
Reflex control of inflammation by sympathetic nerves, not the vagus (Martelli, 2014).
Cutting Edge: Sympathetic Nervous System Increases Proinflammatory Cytokines and Exacerbates Influenza A Virus Pathogenesis (Grebe, 2010).
The sympathetic nervous response in inflammation (Pongratz, 2014). The SNS not only plays a role in emergencies, but with other important players such as the hypothalamic-pituitary-adrenal (HPA) axis and the sensory and vagal nervous systems (SNV), it keeps the body in balance. Its role is very complex, with more pro-inflammatory effects in the early phase, whereas catecholamines have anti-inflammatory effects in the late phase of autoimmune arthritis.
The role of the sympathetic nervous system in intestinal inflammation (Straub, 2006). The SNS has pro- and anti-inflammatory functions. Neurotransmitters such as norepinephrine, adenosine, and others can have different opposing effects depending on their concentration. There are striking similarities between rheumatoid arthritis and inflammatory bowel disease.
e. Sympathetic System Helps Survive
As Spencer put it, “The fittest will survive. Sexiness and communicative qualities may confer certain reproductive advantages, but adrenal hormones and a fighting spirit are even more critical. Beyond mating and birthing, males and females need the best fight-and-flight capabilities to protect their offspring. Mother love requires claws!
Reducing the role of compassion to the danger zone is to forget that predators must fight to survive. Hunting and killing prey doesn't come without effort. The polyvagal vision of a peaceful mammalian world is a utopia. Any documentary about natural life will challenge this cliché within minutes. The sympathetic system is not for emergencies. It is part of normal life for most animals and, sadly, for most inhabitants of this planet. We are not in Utopia.

f. How does the sympathetic system regulate metabolism?

The sympathetic nervous system (SNS) plays an important role in regulating energy expenditure and body weight. The SNS is involved in several metabolic processes that can affect weight. Here's how:
Lipolysis: Norepinephrine (noradrenaline) stimulates lipolysis in white adipose tissue, which breaks down adipose tissue into free fatty acids and glycerol, releasing stored energy.
Thermogenesis: The SNS regulates thermogenesis, particularly in brown adipose tissue (BAT) — which is crucial in small mammals and human infants.
Resting metabolic rate: Chronic activation of the SNS can increase the resting metabolic rate. The body then burns more calories at rest, which impacts weight.
Appetite regulation: While the primary centers for appetite regulation are in the brain (e.g., hypothalamus), stress responses that activate the SNS lead to changes in appetite and eating behavior.
Insulin: The SNS influences glucose metabolism by modulating insulin secretion from the pancreas and insulin sensitivity in target tissues. However, chronic activation of the SNS can reduce insulin sensitivity, affecting weight and overall metabolic health.
g. Sympathetic System and Evolution: A Polyvagal Fallacy
In his phylogenetic description, Porges places the SNS in an intermediate position between the “old” and “new” vagal systems. At first glance, comparative anatomy seems to agree with Porges. Parasympathetic innervation already appeared in elasmobranchs (cartilaginous fish such as sharks and rays), while cardiac sympathetic innervation appeared in bony fish. This is the hard-wiring. However, in hagfish, considered the oldest vertebrate (Nilsson, 1998), the non-innervated heart is full of catecholamines, allowing for strong adrenergic activation. Adrenergic acceleration is as old as cholinergic deceleration. Adrenaline (epinephrine) and acetylcholine are ancient molecules with an evolutionary history dating back to single-celled organisms (see below). Sharks don't have a sympathetic heart, but their hormonal circulation of catecholamines allows them to power their hearts and give them impressive strength and speed.
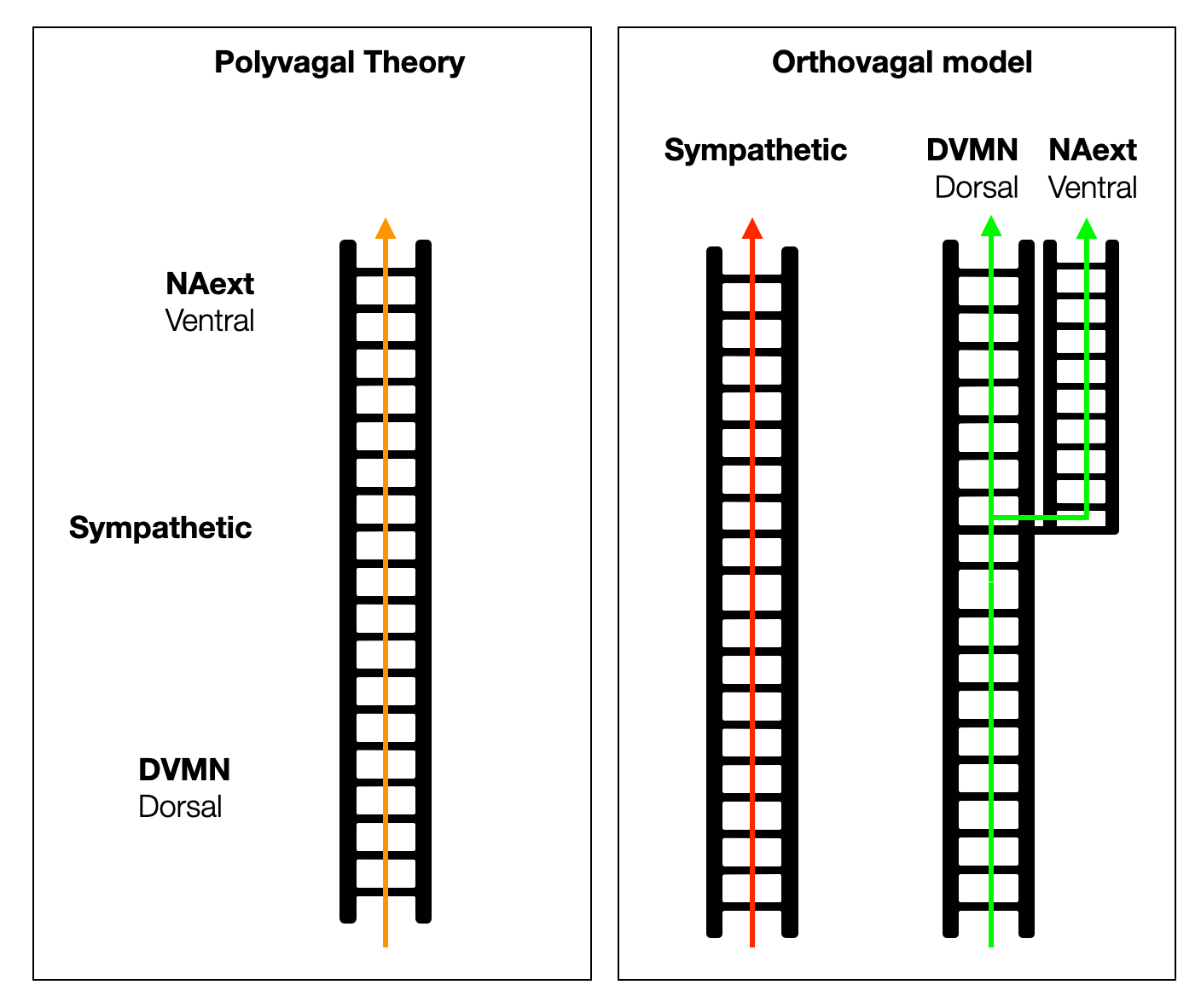
The PVT is arranged on a unique vertical ladder, with the dorsal, sympathetic, and ventral systems arranged as if they had evolved in a straight line. In reality, each system evolved independently, from an ancient organization to a high level of complexity. The adrenergic system first relied on intrinsic cardiac catecholamines, then on circulating (hormonal) catecholamines, and finally took on a physical form as an anatomical system.
From hagfish to birds, the parasympathetic system controls all parasympathetic functions with one dorsal branch. In mammals, the slowing of the heartbeat was moved to a ventral position in the NAext, creating a new branch. However, a small fraction of cardiac vagal neurons (CVPNs) in the DVMN protect and support the left ventricle.
h. How is the Sympathetic System Relevant for Cognitive & Social Tasks?
As suggested above, the adrenergic drive is more than the anatomical neural system along the spine. The neurotransmitter noradrenaline (norepinephrine) is critical for all mental effort, with or without physical consequences — no creativity, concentration, or sustained mental effort without noradrenaline. Noradrenergic systems are also involved in attention processes relevant to social play behavior. Dopamine and norepinephrine are critical neuromodulators that control brain states, vigilance, action, reward, learning, and memory processes. The ventral tegmental area (VTA) in the midbrain and the locus coeruleus (LC) in the upper medulla are canonically described as the primary sources of dopamine (DA) and norepinephrine (NA) with dissociated functions. Comparison of different studies shows that these neuromodulators overlap to a large extent (Ranjbar-Slamloo, 2020).
-
Attention and Vigilance
Norepinephrine is critical for modulating attention and increasing vigilance. It is critical to the brain's salience network, which helps determine relevant stimuli that require attention. By modulating attention, norepinephrine can influence social interactions. For example, paying attention to social cues can enhance bonding and understanding in social contexts.
Social memory
Some studies suggest that norepinephrine may influence social memory, which is the recognition and recollection of other individuals. This is critical for forming and maintaining social bonds in many species.
Bonding
While oxytocin is the molecule most commonly associated with bonding (especially maternal bonding), norepinephrine may also play a role, especially under stress. For example, social animals may seek out and bond with others for mutual support during stressful events.
Emotional contagion and empathy Noradrenaline enhances emotional responses, making individuals more attuned to the emotions of others — a fundamental aspect of empathy.
Response to social exclusion
Elevated levels of norepinephrine increase sensitivity to social exclusion. Norepinephrine may be part of the brain's mechanism for promoting social bonding and cohesion by making social exclusion feel distressing.

Further reading
Noradrenaline might enhance assertive human social behaviours (Tse, 2006) presents preliminary evidence that a two-week increase in norepinephrine transmission induced by reboxetine makes healthy volunteers more confident and assertive. Reboxetine is a type of antidepressant known as a selective norepinephrine reuptake inhibitor (NARI).
Monoaminergic Receptors as Modulators of the Perivascular Sympathetic and Sensory CGRPergic Outflow (Marichal-Cancino, 2020). Together, monoamines are involved in many physiological and mental processes. Norepinephrine prepares us to respond to stressful situations. Norepinephrine and epinephrine are also involved in alertness, wakefulness, attention, and memory.
Emotional enhancement of memory: how norepinephrine enables synaptic plasticity (Tully, 2010).
Noradrenaline effects on social beaviour, intergroupe relations, and moral decisions (Terbeck, 2016). Contrasting Roles of Dopamine and Noradrenaline in the Motivational Properties of Social Play Behavior in Rats (Achterberg, 2016).
The neurobiology of social play behavior in rats, (Vanderschuren, 1997)shows how social play in animals may serve to establish social organization within groups, inhibit aggression, and increase group stability. Social play is one of the earliest forms of non-maternal social behavior to appear in mammalian ontogeny. In addition to opioids and dopaminergic receptors, cholinergic and adrenergic systems are involved.
i. Sympathetic System and Love
An anecdote associated with Avicenna, the famous physician of the Islamic Golden Age, says that he diagnosed a prince's illness by taking his pulse. Each time the name of a woman the prince secretly loved was mentioned, his pulse would jump, leading Avicenna to conclude that his patient was suffering from lovesickness. This consultation was the historic birth of psychosomatic medicine.


Porges doesn't share this romantic view of the sympathetic nervous system that makes our hearts leap at the thought of the beloved. He draws a caricature of the sympathetic nervous system by fully embracing the reductionist “fight-flight” cliché. He also tries to convince his audience that the sympathetic is old-not as old as the “dorsal”-and associated with threat and negative arousal. In this chapter, we will correct this dystopian portrait. We will discover another view of the anatomy, physiology, and embryology of the SNS that goes beyond threat. We will challenge and correct the polyvagal paradigm, which places the sympathetic system in a middle position between the “dorsal” and the “vagal”.
The PVT postulates that the sympathetic system is
Correlates with a state of danger, resulting in a “fight-flight” response.
Appeared in evolution (phylogeny) after the “dorsal” parasympathetic system and before the “intelligent vagal” system.
Implies negative social and medical consequences
j. Mitochondria and Sympathetic System
Promoting a lifestyle that stimulates and protects the mitochondrion is challenging for psychosomatic medicine. In An Energetic View of Stress: Focus on Mitochondria (Picard, 2018), the author presents research on hard-wired mechanisms that link psychosomatic processes to the current biomedical framework. More specifically, he examines how the multifunctional mitochondria respond to stress. Mitochondria are more than cellular energy plants (ATP) we know. They react to allostatic load through signaling and remodeling (fission). There is a growing interest in mitochondria in medical research, and many diseases are now recognized as being caused by mitochondrial defects. Mitochondrial DNA, telomere length and DNA methylation may represent the biological “aging clock”. Because mitochondria are present at all metabolic levels of the brain, their good condition is critical for mental health.
In Exerkines in health, resilience and disease (Chow, 2022), the authors review the molecular mechanisms underlying the beneficial effects of exercise. Since the discovery in 2000 that muscle contraction releases IL-6, many exercise-associated signaling molecules have been identified. Exerkines are signaling components (= parts of molecules) released by various organs and tissues in response to acute and chronic exercise. They improve cardiovascular, metabolic, immune, and neurological health. They have been implicated in treating cardiovascular disease, type 2 diabetes mellitus, and obesity, and may facilitate healthy aging.
In Dietary Restriction and AMPK Increase Lifespan via Mitochondrial Network and Peroxisome Remodeling (Weir, 2017), the authors show how the cellular mitochondrial network (fusion vs. fission) is a health index that predicts lifespan. Intermittent fasting, like any form of calorie reduction, has a positive effect on mitochondria.
k. Sympathetic System and Vagal Nerve
Adrenergic agonist induces rhythmic firing in quiescent cardiac preganglionic neurons in nucleus ambiguous via activation of intrinsic membrane excitability (Aiba, Noebels, 2019).
Cholinergic vagal nerves projecting from neurons in the nucleus ambiguus (NA) play a predominant role in cardiac parasympathetic pacemaker control. Central adrenergic signaling modulates the tone of this vagal output. This study shows that central adrenergic signaling increases the intrinsic excitability of NA neurons. This central compensatory mechanism may counteract excessive peripheral cardiac excitation during increased sympathetic tone.
Correlation of sympathetic and parasympathetic nervous system activity during rest and acute stress tasks (Weissman, 2019).
The seemingly opposing roles of the SNS and PNS may lead one to assume that their activity functions along a uniaxial continuum. However, the autonomic space model (Berntson et al., 1991) suggests that SNS and PNS activity is not antagonistic. Instead, depending on the situation and individual differences, the two branches can shape peripheral responses through coordinated and uncoordinated activity (e.g., heartbeat).
2.7.10
Literature on the Sympathetic Nervous System
Here, we present several scientific articles showing how the sympathetic nervous system, which relies on catecholamines (e.g., adrenaline or epinephrine), was installed very early in evolution. Authors describe systems in plants, jawless fishes (agnathans), and jawed fishes (gnathostomes) such as sharks and rays (elasmobranchs) or teleosts (bony fishes). Furthermore, in early vertebrates, the parasympathetic system and acetylcholine strengthen and accelerate the heart (Taylor 1992, 2014). Therefore, the phylogenetic perspective of the PVT (polyvagal ladder) is incorrect.
Catecholamines are active compounds in plants (Kulma, 2007). Their involvement in plant growth, stress responses, and sugar metabolism suggests they have essential regulatory functions. Plant leaves subjected to wounding and water stress show a rapid, transient increase in catecholamines.
Neuromodulation by Monoamines is a Bilaterian Innovation (Goulty, 2022). Monoamines like serotonin, dopamine, and adrenaline/noradrenaline (epinephrine/ norepinephrine) act as neuromodulators, contributing significantly to ‘higher’ cognitive functions such as learning and memory formation and modulating fundamental homeostatic needs like sleep or feeding. The authors propose that most of the genes of the monoaminergic system originated in the common ancestor of bilaterians. Kimberella, 555 mya, is the first accepted Bilateral fossil.
Expression of Sympathetic Nervous System Genes in Lamprey Suggests Their Recruitment for Specification of a New Vertebrate Feature (Häming, 2011). The sea lamprey, a basal, jawless vertebrate, lacks jaws and sympathetic ganglia, but has many neural crest derivatives. This suggests that the factors involved in sympathetic neuron differentiation were either innovations by the gnathostomes (fish with jaw) or already present in lamprey but serving different purposes. The presence of transcription factors that mediate sympathetic neuron differentiation in the lamprey genome and their expression in various sites in the embryo, albeit not overlapping in ganglionic structures, suggests their potential recruitment during gnathostomes’ evolution to a new function in a neural crest derivative.
According to Evolution of the autonomic innervation of visceral and cardiovascular systems in vertebrates (Burnstock, 1969), the primitive parasympathetic innervation of the heart is cholinergic and inhibitory. It has been consistently maintained throughout vertebrate evolution. Similarly, adrenergic cardiac acceleration fibers appear early in evolution and are maintained throughout.
Primitively, sympathetic and vagal fibers converge in the vagosympathetic trunks. In reptiles, they reach the heart through separate nerve trunks. We may find some adrenergic fibers in the mammalian vagal trunks. Special catecholamine-containing cells are a constant feature of the vertebrate heart. The morphologically defined sympathetic nervous system of the nerve trunks of fish and amphibians consists primarily of excitatory cholinergic fibers. In reptiles and birds, there are varying proportions of adrenergic and cholinergic fibers in sympathetic trunks, whereas in mammals, most, and sometimes all, of the fibers in sympathetic trunks are adrenergic. There is a general trend from cholinergic to adrenergic sympathetic control of the visceral and vascular systems.
The influence of circulating catecholamines released from diffusely distributed chromaffin cells appears to play a much more important role in lower vertebrates than in higher vertebrates, where direct nervous control has evolved to a greater extent. Chromaffin tissue is widespread in cyclostomes and closely associated with sympathetic ganglia in elasmobranchs and, to a large extent, in teleosts and amphibians.
Stem cells, evolutionary aspects and pathology of the adrenal medulla: A new developmental paradigm (Kastriti, 2020). The study of the “live fossil” agnathans, the lamprey, reveals the presence of extra- and intra-cardiac chromaffin cells(i.e., adrenergic) in larval and adult animals.
On the Vagal Cardiac Nerves, with Special Reference to the Early Evolution of the Head–Trunk Interface (Higashiyama, 2016). In the lamprey, no vagus branch was found in the venous pole throughout development, whereas a branch from the branchial portion innervated the arterial pole.
Electrical Excitation of the Heart in a Basal Vertebrate, the European River Lamprey (Lampetra fluviatilis) (Haverinen, 2014) describes two distinct modes of cardiac excitability, allowing for fast and slow activity, respectively.
The Unusual Adrenergic-Like Excitatory Action of Acetylcholine on the Ventricular Cardiac Muscle of the Horned Shark, Heterodontus portusjacksoni (Thompson, 1997), describes a massive increase in the ventricle’s contraction force in response to exogenously applied acetylcholine. This is similar to that produced by applied adrenaline and blocked by the antagonist atropine. Acetylcholine mediates a catecholamine release through a cholinergic-adrenocomplex, representing a transitional adrenergic state between humoral and neuronal regulation. On the contrary, the spontaneously beating atrium shows an atropine-sensitive slowing, a response typical of other vertebrates.
Comment: Elasmobranchs (sharks) show a parallel evolution of sympathetic and parasympathetic functions related to the heart.
Physiological Responses to Stress in Sharks (Skomal, 2010). The primary response to stress in fish is neuroendocrine, with rapid increases in circulating levels of stress hormones (primarily catecholamines and corticosteroids). Circulating catecholamines increase significantly in elasmobranchs immediately after the perception of a stressor and peak within a few minutes after the stress event has ended. Some authors have found that catecholamine levels increase from 100 to 1600 times above routine swimming levels.
The Unusual Adrenergic-Like Excitatory Action of Acetylcholine on the Ventricular Cardiac Muscle of the Horned Shark (Thompson, 1997) describes a complex that may represent a transitional adrenergic state between humoral and neuronal regulation. The spontaneously beating atrium showed no evidence of such an excitatory response to applied acetylcholine but produced an atropine-sensitive slowing, a response typical of other vertebrates.
Adrenergic tone benefits cardiac performance and warming tolerance in two teleost fishes that lack a coronary circulation (Ekström, 2021). Role of the temperature on the adrenergic tone.
Catecholamines in a cavefish brain (Gallman, 2019). The teleost fish Astyanax mexicanus has two forms: an ancestral riverine form (surface) and a derived blind cave form (cavefish). Adaptation to a nutrient-poor cave life without predation has led to marked behavioral differences. The cavefish has lost the defensive responses of the ancestral surface fish, such as stimulus aversion, and instead displays enhanced food-seeking behavior. This is reflected by increased catecholamine immunoreactivity in regions associated with non-visual sensory perception, motor control pathways, attention, and endocrine release. The increased size and number of catecholaminergic neurons in the cavefish brain have evolved from aversive defensive responses to attractive exploratory behaviors.
Comment: This is consistent with Panksepp's concept of the role of catecholamines in exploratory function.
The Autonomic Nervous System and Chromaffin Tissue in Hagfishes (Nilsson, 1998) is a chapter of The Biology of Hagfishes (Jorgensen, 1998). The hagfish heart does not receive extrinsic innervation, so heart rate control depends on mechanisms other than the autonomic nervous system. Their hearts store large amounts of catecholamines, but are relatively insensitive to external transmitters or hormones. Blockade of beta-adrenergic receptors causes severe bradycardia, leading to the idea that the heart needs adrenergic tone to function well. The authors conclude that the hagfish ANS is poorly developed and should be considered a rudimentary version rather than a template.
In The Phylogeny and Ontogeny of Autonomic Control of the Heart and Cardiorespiratory Interactions in Vertebrates (Taylor, 2014), the authors provide a comprehensive review of the development of the autonomic nervous system. Hagfish, the oldest vertebrates, have no cardiac innervation. In lampreys, vagal stimulation accelerates the heart. Elasmobranchs (cartilaginous fishes) have inhibitory vagal innervation of the heart but no direct sympathetic innervation. Circulating catecholamines directly affect heart rate and modulate vagal control.
In summary, there is no direct ladder from hagfish to Homo sapiens with a neat hierarchical order. The sympathetic and parasympathetic systems don't appear in a “one, two, three” order, as the PVT postulates. On the contrary, there is evolutionary tinkering as the two systems slowly evolve from intracardiac production of catecholamines to hormonal circulation to vagal innervation of the heart.
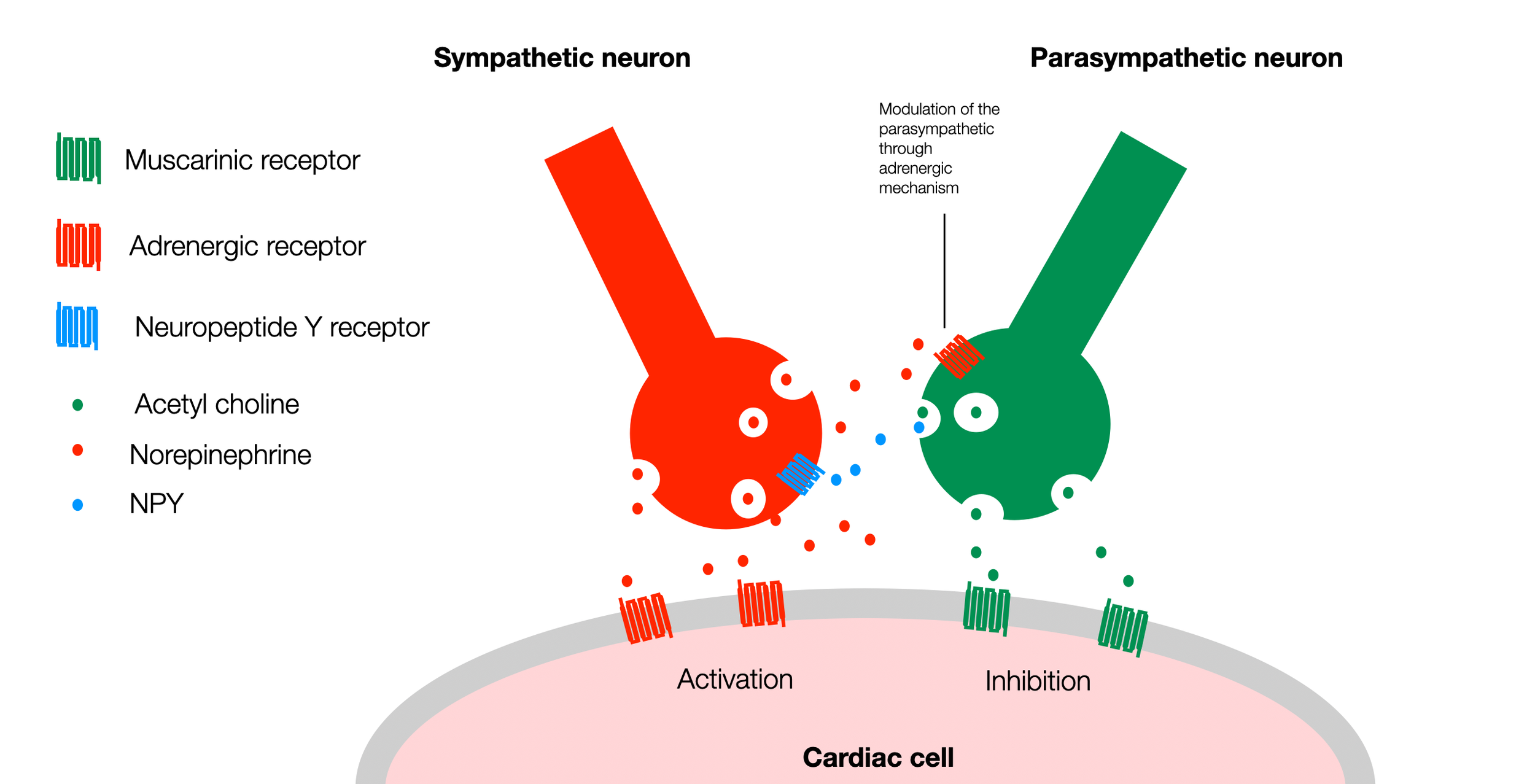
According to The Intrinsic Cardiac Nervous System and Its Role in Cardiac Pacemaking and Conduction (Fedele, 2020), sympathetic and parasympathetic neurons appear to share a common progenitor, with a possible role for bone morphogenetic protein (BMP)2 in their specification. Sympathetic neurons can transdifferentiate into parasympathetic neurons in vitro when stimulated with low levels of BMP2. High levels of BMP2 promote sympathetic differentiation, while low levels induce parasympathetic identity (White, 2001), (Vegh, 2016). Fedele also presents an exciting diagram (Figure 5.) illustrating the crosstalk of the sympathetic and parasympathetic varicosities (terminals) at the level of a sinoatrial node myocyte (heart muscle cell involved in pacemaker activity).
For all these reasons, the polyvagal description of sympathetic activation as a Jacksonian dissolution doesn't make sense.
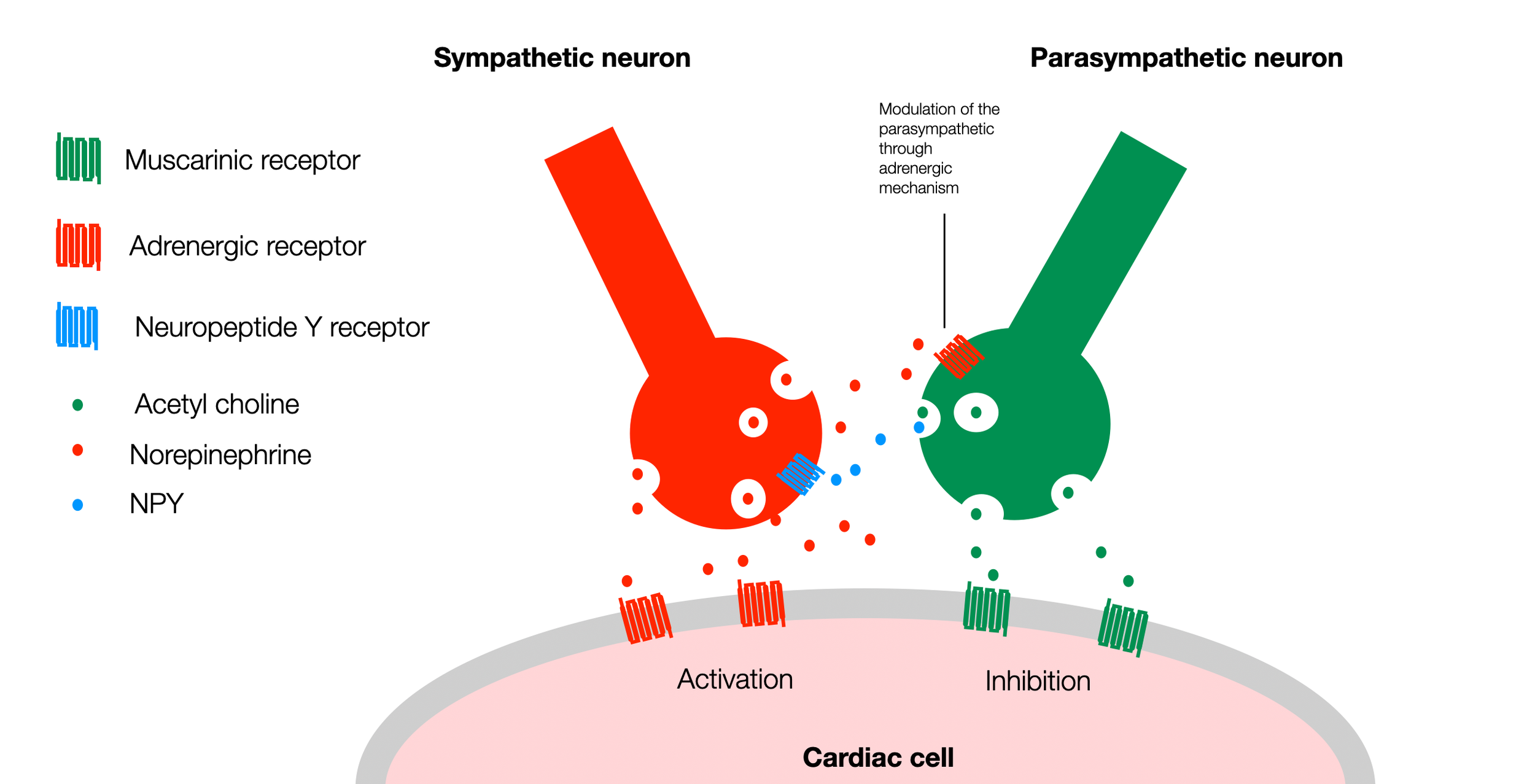
In this simplified version of the diagram (Fedele, 2020), we can see that both sympathetic and parasympathetic neurons have receptors on the surface of the heart cell (pacemaker cell). They up or down regulate the activity of the cardiac cell. However, parasympathetic and sympathetic terminals (varicosities) establish a crosstalk through specific signaling factors. A parasympathetic (muscarinic) receptor on the surface of the sympathetic neuron can block the release of norepinephrine (NE). Similarly, the sympathetic neuron can use neuropeptide Y to block the release of acetylcholine (ACh) from the parasympathetic neuron. For simplicity, this diagram doesn't show other participants in this regulation (ATP, nitric oxide, and VIP) (©absb, 2024).
>> to the next chapter Social Engagement