2.9.0
Is Heart Rate Variability Unique to Mammals?
Heart rate variability (HRV) is dominant in the polyvagal construct and is the critical factor that makes the mammalian “smart vagal” system unique. Indeed, Porges tends to limit cardio-respiratory synchrony to mammals. Grossman and the Taylor group challenge this view.
In the 1990s, most HRV experts seemed to be on the same page. They even wrote a paper together. This changed in 2007.
Heart Rate Variability: Origins, Methods, and Interpretive Caveats (Berntson, 1997) is an excellent presentation of HRV by Berntson, G. G., Grossman, P., Porges, S. W., and eight other authors. This article is fascinating because it reunites the future opponents in a co-authored publication: Bernston and Grossman vs. Porges. Ten years later, Whither vagal tone (Berntson, Cacioppo, Grossman, 2007) and Toward understanding respiratory sinus arrhythmia (Grossman, Taylor, 2007) set the record straight.
2.9.1
What is Heart Rate Variability (HRV)?
A long time ago, researchers observed that breathing affects the heartbeat—or, more precisely, the interval between two heartbeats. Inspiration accelerates the pulse; exhalation slows it. Because the heart rhythm originates in the sinoatrial node (see picture) in the upper part of the right atrium, this variability is called respiratory sinus arrhythmia (RSA). Including other parameters besides respiratory input, such as sympathetic tone variation, leads to the expanded Heart Rate Variability (HRV) concept.
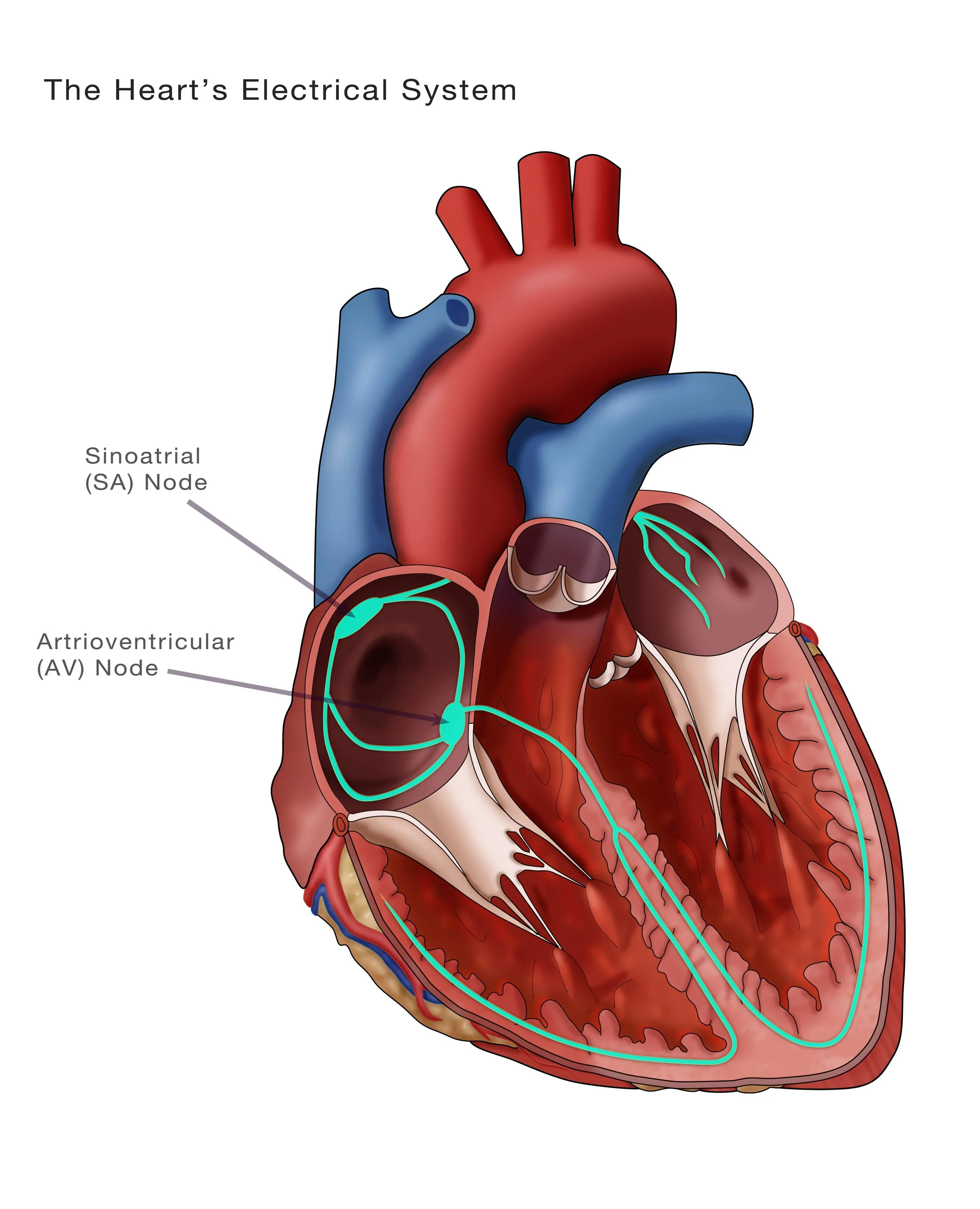
The sinus node (SA node) is the heart's primary pacemaker. A small mass of specialized cells at the top of the right atrium (upper chamber) discharges spontaneously at 90–107 beats per minute (Shaffer, 2014), causing the atrium to contract. Further down, the atrioventricular node (AV node) acts as a secondary pacemaker, discharging at about 40–60 beats per minute. Further down, a third group of pacemaker cells with a rate of 30–40 beats per minute (X, W. 2022). The ventral branch of the parasympathetic nervous system slows the sinus node's natural heartbeat to an average rhythm of about 70 beats per minute (adult human). Reducing vagal input or increasing sympathetic tone both accelerate the heartbeat. Parasympathetic nerves act more quickly (< 1 s) than sympathetic nerves (> 5 s) (Nunan, 2010).
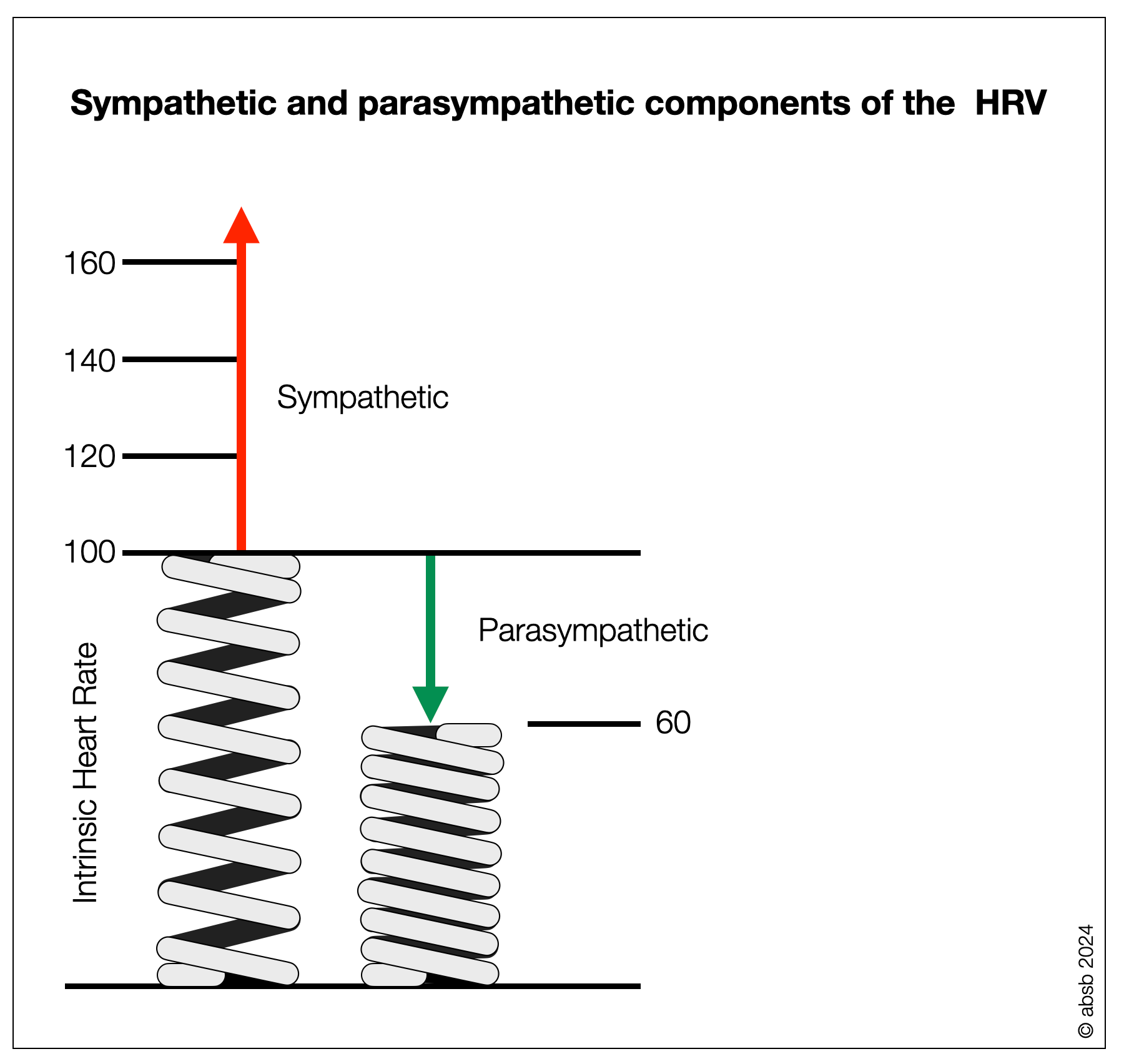
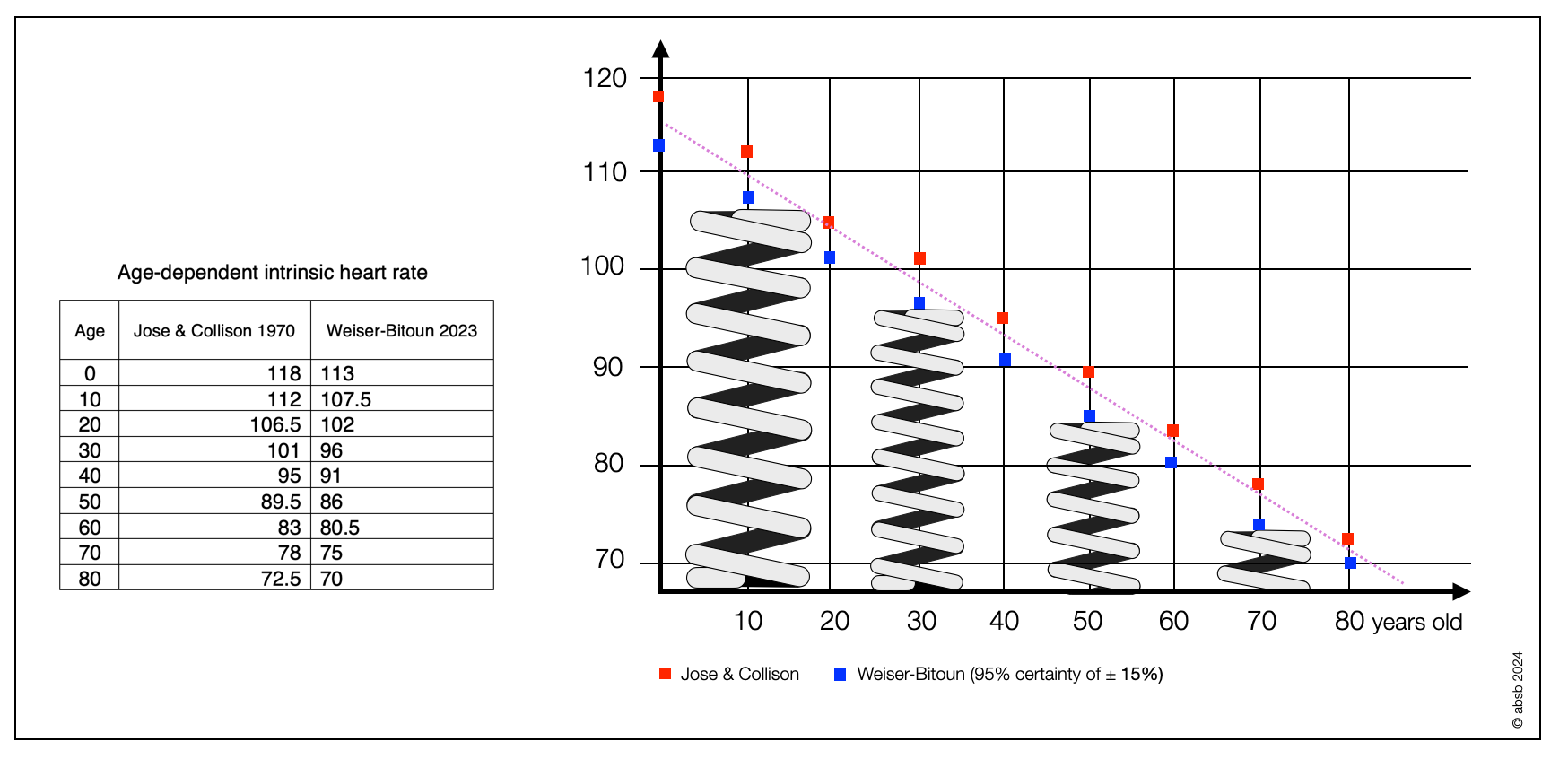
In these diagrams, the feather symbolizes the activity of the intrinsic cardiac system, which has its own rhythm of about 90 bpm. The parasympathetic system squeezes the spring and lowers the heart rate. When the pressure is released, the spring uncoils, and the rhythm increases. The sympathetic system naturally contributes to the acceleration, especially when the effort requires a heart rate above 90 bpm. Intrinsic heart rate decreases with age.
2.9.2
Tonic vs. Phasic Parasympathetic Activity
In his “polyvagal manifest,” Porges (1995) clearly distinguishes between bradycardia, a tonic parasympathetic activity, and heart rate variability, a phasic phenomenon. In physiology, "phasic” and “tonic” refer to different types of responses or activities in cells and tissues, particularly in the context of muscle and nerve cells.

When you write on your laptop, your fingers make short, phasic movements while the rest of your body stays slightly in the same position (tonic muscle tension)-so you don't fall out of your chair! You can be relaxed and write, or tensed and write. Phasic movement and muscle tone are two different aspects of your muscle system.
How do these concepts apply to the heart and circulation? Under the influence of the sympathetic system, the vascular muscles maintain a certain tone, which results in a relatively — but not quite — constant blood pressure. Your blood pressure varies according to the phases of the heart (contraction = systole vs. dilation = diastole). Similarly, the parasympathetic system creates two phenomena through the vagal nerve: bradycardia (tonic) and HRV (phasic).
Tonic may relate to blood pressure, muscle tone, and heartbeat. It shows steadiness. It's static.

Tonic: A sustained slowing of the heartbeat below 60 beats per minute (bpm) results in bradycardia (Greek prefix bradu- βραδυ = slow). The opposite would be tachycardia (Greek prefix tachy- ταχυ = fast), which is an acceleration of the heartbeat above 100 beats per minute (bpm) at rest.
Phasic: Depending on the respiratory phases (inspiration vs. expiration), the heartbeat shows a short phase of acceleration vs. deceleration, reflecting the vagal nerve's motor action.
Phasic comes in waves, cycles, or periods. It shows variability. It’s rhythmic and dynamic.
The sixty-four-dollar question is: Do one or two neural centers control these parameters? Or more? Is our heart “monovagal,” “bivagal,” or genuinely polyvagal?
2.9.3
The Polyvagal Paradox
There are two polyvagal paradoxes.
The first was triggered by the neonatologist's question to Porges: How can vagal tone be beneficial if it causes lethal bradycardia in newborns?
The second polyvagal paradox described by Porges in his inaugural lectures in 1994 relates to the observation of simultaneous bradycardia (e.g., heart rate below 60 BPM) and low heart rate variability (HRV).
However, the vagal tonus should increase HRV. Porges' answer is, like King Alexander, to cut the Gordian knot with the polyvagal sword. Not so elegant, but highly effective. According to the PVT, the ventral “smart vagal” (originating in the nucleus ambiguus) creates phasic variations (HRV). At the same time, the tonic aspects (e.g., bradycardia) are related to the activity of the dorsal “old vagal.”
Before we can confirm or challenge the polyvagal hypothesis, we need to understand what HRV is, why it seems to be good, and what the experts argue about. This will be a rather technical chapter. So don't expect to understand it all.
2.9.4
How to Measure the Heart Rate Variability (HRV)?
There are two main ways to measure HRV: time scales and frequency scales.
1. Temporal scales: This method looks at the time between heartbeats over some time. Imagine counting the time between each heartbeat in milliseconds. Because The heart is not a metronome (Shaffer, 2014), this time (duration) constantly changes. By analyzing these time intervals, we can measure how variable the heart rate is — generally a positive sign of flexibility. A simple example below shows how to calculate the average time difference based on four heartbeats — which is too short. This method is easy to understand because it's directly about time.

2. Frequency scales: This approach gets a little more technical and involves breaking down the heart rhythm into different components based on how fast or slow they are. It is like listening to music and separating the beats into bass and treble. In HRV, we use special mathematics (Fourier transform or similar methods) to identify these rhythms and see how they relate to various health and nervous system activity aspects. These rhythms are categorized into bands, high frequency (HF) and low frequency (LF), each associated with different physiological processes. It is helpful to distinguish between the parasympathetic (vagal) and sympathetic components of HRV.

2.9.5
HRV based on Temporal Scales

SDNN and RMSSD are two HRV measures based on time scales:
SDNN (standard deviation of NN intervals): This is the standard deviation of all normal-to-normal (NN) intervals (effectively the R's of the QRS complexes). SDNN represents the overall heart rate variability and reflects both sympathetic and parasympathetic influences on the heart. A higher SDNN indicates increased variability and is typically considered a healthy heart and nervous system marker.
RMSSD (Root Mean Square of the Successive Differences): This measure calculates the square root of the mean of the squares of the successive differences between adjacent NN intervals. RMSSD focuses on heart rate variability over short periods and is considered a good indicator of parasympathetic (vagal) activity. Higher RMSSD values indicate more significant variability due to parasympathetic activity, which is generally associated with better health and stress resilience.

This ECG example shows the electrical activity at the sinus node. From one peak (the R of the QRS complex) to the next, there is a variable time interval measured in milliseconds (ms). This is not the same as the heartbeat measured by BPM (beats per minute)—diagram by YitzhakNat, Wikipedia. Below, we asked ChatGPT to calculate the SDNN and RMSSD of the ECG example (2.5 seconds of heart beat data).
-
This is how you calculate the SDNN (Standard Deviation of NN intervals):
1. Calculate the mean interval: 859 + 793 + 726 = 2378.
2. Divide by 3 = 792.67
3. The variance is the average of the squared differences. Calculate and square the differences (interval – mean interval).
• (859 – 792.67)² = (66.33)² = 4399.67
• (793 – 792.67)² = (0.33)² = 0.11 (rounded)
• (726 – 792.67)² = (-66.67)² = 4444.89
4. Add the results and divide by n-1 (Bessel’s correction), which means here by 3-1=2. 8’844.67 divided by 2 = 4’422.33
5. The standard deviation is equal to the square root of the variance: ≈ √4422.33 ≈ 66.50 ms.
N.B. The Bessel correction better reflects a population standard deviation based on a specific sample of data (sample standard deviation).
-
RMSSD is the root mean square of the successive differences between normal heartbeats. It's another time-domain method of assessing heart rate variability (HRV). Here is how we calculate it:
1. Calculate the successive differences: In this example, we calculate the difference between the first and second intervals and the second and third intervals.
2. Square the differences: We square each of these successive differences.
3. Calculate the mean of the squared differences: We find the average (mean) of these squared differences.
4. Take the square root: Finally, we calculate the square root of the mean from step 3 to obtain the RMSSD.
Let's calculate the RMSSD using the RR intervals from the figure above:
1. First RR interval (R1): 859 ms.
2. Second RR interval (R2): 793 ms.
3. Third RR interval (R3): 726 ms.
We do the following calculations:
1. Compute the successive differences:
- 793-859=-66 ms
- 726-793=-67 ms
2. Square the differences:
- (-66)2=4356 ms².
- (-67)2=4489 ms².
3. Calculate the mean of the squared differences:
- Mean of the squared differences=4356+44892=4422.5
- Mean of the squared differences=24356+4489 =4422.5 ms².
4. Take the square root of the mean of the squared differences to find the RMSSD:
- RMSSD = 4422.5≈66.5RMSSD = 4422.5 ≈ 66.5 ms
2.9.6
HRV Based on Frequency Scales

Frequency-domain measurements estimate absolute or relative “power” distribution in four frequency bands. The Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology (1996) divided heart rate (HR) oscillations into ultra-low frequency (ULF), very-low frequency (VLF), low frequency (LF), and high frequency (HF). For various reasons, the vagal efferent fibers create more variability. Compare this to the many gentle taps on the brakes as you drive through a city. These taps are frequent and create high-frequency variation. In contrast, leaving the city for the highway is a single “full-throttle” acceleration — e.g., from 35 mph (ca. 56 km/h) (about 56 km/h) to 75 mph (ca. 121 km/h) (about 121 km/h). Similarly, the sympathetic system is the heart's primary agent for acceleration and dominates the low-frequency band.
-
The LF/HF ratio is calculated by dividing the LF component's power by the HF component's. This ratio is a marker of the balance between the sympathetic and parasympathetic branches of the autonomic nervous system. A higher LF/HF ratio indicates a dominance of sympathetic activity (or reduced vagal tone). Conversely, a lower LF/HF ratio suggests a dominance of parasympathetic activity, indicating relaxation or recovery.
In HRV spectral analysis, the power spectrum is typically divided into different frequency bands, mainly:
Low Frequency (LF): Typically in the 0.04 to 0.15 Hz range. The LF component reflects sympathetic and parasympathetic activity, but it's often considered an indicator of sympathetic modulation.
High Frequency (HF): Typically, it is in the range of 0.15 to 0.4 Hz. The HF component is associated with respiratory rhythm and is considered an indicator of parasympathetic (vagal) activity.
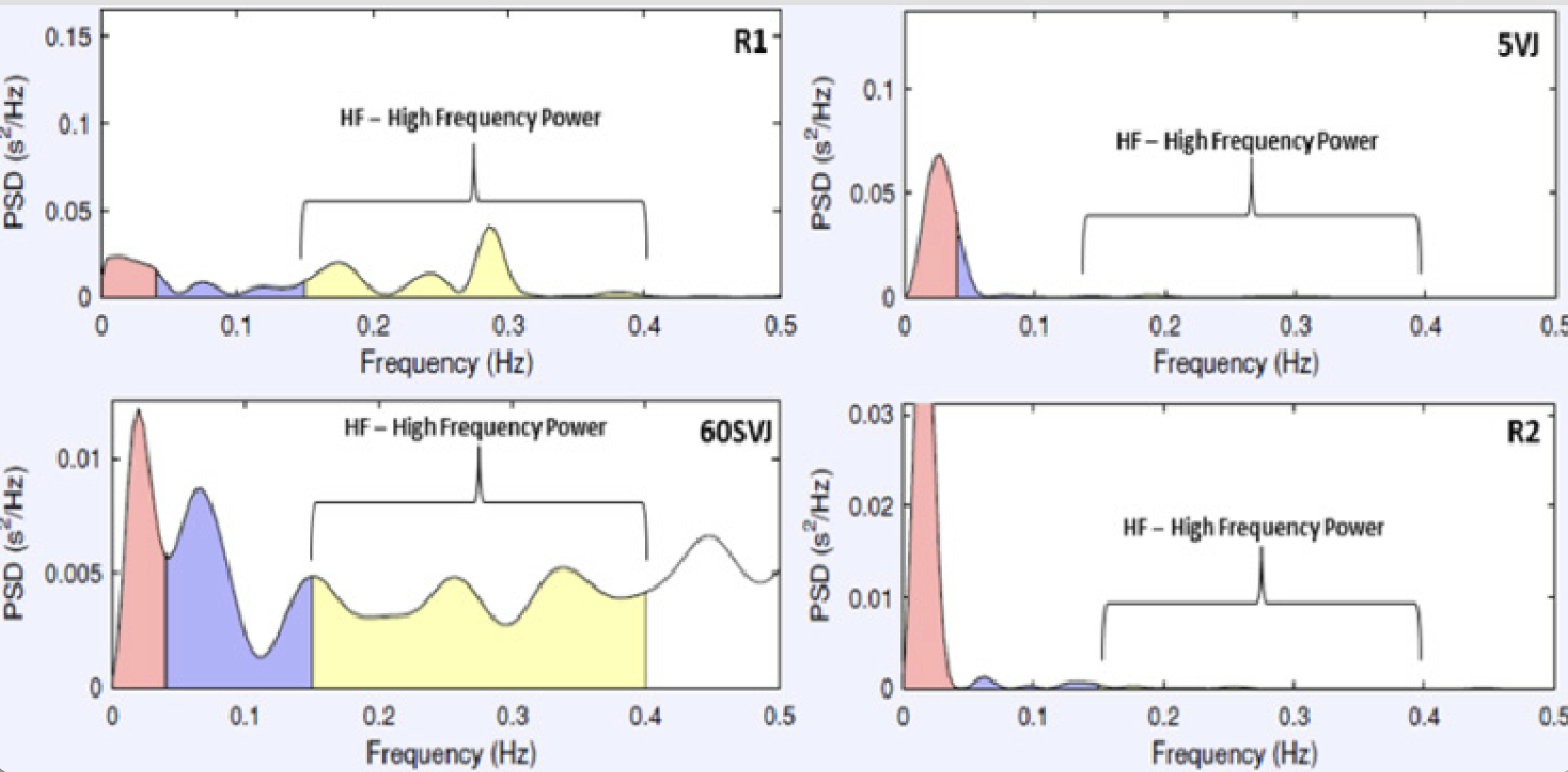
Heart Rate Variability Responses in Vertical Jump Performance of Basketball Players — Scientific Figure on ResearchGate. (Morales, 2014).
2.9.7
What is the Source of the Heart Rate Variability (HRV)?
There is no doubt that HRV exists. However, PVT does not provide much explanation as to its origin other than that when the “ventral” or “social engagement” predominates, HRV is high. When the “dorsal” predominates, it leads to a significant decrease in heart rate and HRV. This is not enough.
The previous sections show that HRV is a phasic phenomenon that a simple homeostatic loop, including the NAext, the baroreceptors, and the NTS, can't explain. There is too much variability, and we must include the respiratory centers to account for the respiratory rhythm.
In Should heart rate variability be “corrected” for heart rate? Biological, quantitative, and interpretive considerations (De Geus, 2019), the five authors, including Gary G. Berntson, provide an update on where and how HRV originated–without citing the polyvagal theory. Berntson co-authored Cacioppo and Grossman's first review of the PVT in 2007: Whither Vagal Tone.
The following section presents various models for the source of HRV.
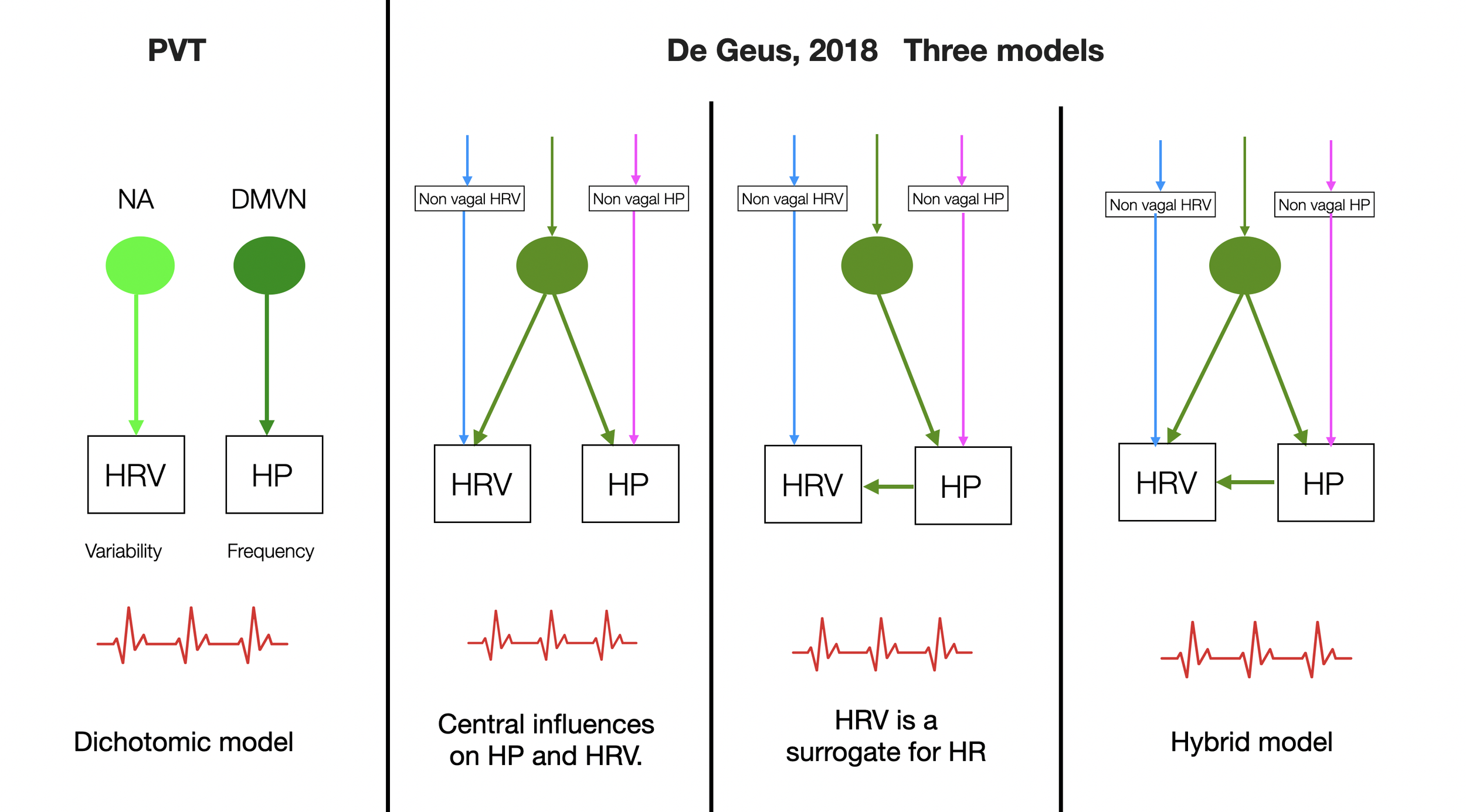
Here, we have added PVT to a simplified version of Figure 4, as seen in the article (De Geus, 2018). This illustrates the controversy over the origins of HRV and HP (heart period). The polyvagal theory radicalizes the debate by presenting the DMVN as the sole cause of bradycardia. A circular loop connects the Ventral Vagal Complex (VVC), HRV, and the sense of safety to the facial interface.
In De Geus' first model, the heart period is hardwired to its variability. A central variable influences both the heart period and HRV.
In the second model, the heart period is the sole driver of HRV, which acts only as a nonlinear surrogate for HR. This model doesn't explain the decoupling of HR and HRV induced by respiratory manipulations.
The third hybrid model raises the question: Is the cardiac period a driver of HRV independent of vagal activity?
2.9.8
The Role of the Central Respiratory Oscillator in Heart Rate Variability
We long thought that Porges ignored the role of respiratory centers until we came across a paragraph entitled Fallacy of Equivalence in Polyvagal Safety (Porges, 2021, p. 39). “First, in mammals there is a well-defined common central respiratory oscillator that sends a respiratory rhythm from the brain-stem to both the heart and the bronchi.” Porges continues:
This information, originating in the nucleus ambiguus, flows through the vagal nerve.
This oscillator is an emergent property of the NA-larynx-pharynx interaction. (VVC).
This is not consistent with the primitive nucleus ambiguus seen in vertebrates that preceded mammals (accusing Taylor and his group of missing some points).
A “common central oscillator” is functionally unique to mammals.
There are some errors here.
The pacemaker cells in the inspiratory and expiratory groups (two groups) give the rhythm, which may not flow directly to the NA, but first to the NTS (which includes a respiratory nucleus of its own). The NTS integrates and synthesizes the afferent information before passing it on.
The central respiratory oscillator is purely respiratory and is not an emergent property of the NA.
There is no “primitive nucleus ambiguus” but a homologous center. Therefore, as the small list of references in the next paragraph shows, mammals didn't invent the respiratory oscillator.
Mammals don't have one “common” oscillator. The respiratory system is very heterogeneous and consists of two central oscillators. The NTS, not the NA, plays the most significant role in integrating and modulating.
Wilson, R. J., Vasilakos, K., & Remmers, J. E. (2006). Phylogeny of vertebrate respiratory rhythm generators: the Oscillator Homology Hypothesis. Respir Physiol Neurobiol, 154(1-2), 47-60. https://doi.org/10.1016/j.resp.2006.04.007
Kinkead, R. (2009). Phylogenetic trends in respiratory rhythmogenesis: insights from ectothermic vertebrates. Respir Physiol Neurobiol, 168(1-2), 39-48. https://doi.org/10.1016/j.resp.2009.05.011
Vasilakos, K., Wilson, R. J., Kimura, N., & Remmers, J. E. (2005). Ancient gill and lung oscillators may generate the respiratory rhythm of frogs and rats. J Neurobiol, 62(3), 369-385. https://doi.org/10.1002/neu.20102
Hedrick, M. S. (2005). Development of respiratory rhythm generation in ectothermic vertebrates. Respir Physiol Neurobiol, 149(1-3), 29-41. https://doi.org/10.1016/j.resp.2005.03.019
Janes, T. A., Rousseau, J. P., Fournier, S., Kiernan, E. A., Harris, M. B., Taylor, B. E., & Kinkead, R. (2019). Development of central respiratory control in anurans: The role of neurochemicals in the emergence of air-breathing and the hypoxic response. Respir Physiol Neurobiol, 270, 103266. https://doi.org/10.1016/j.resp.2019.103266
Baghdadwala, M. I., Duchcherer, M., Paramonov, J., & Wilson, R. J. (2015). Three brainstem areas involved in respiratory rhythm generation in bullfrogs. J Physiol, 593(13), 2941-2954. https://doi.org/10.1113/jp270380
Johnson, S. M., Hedrick, M. S., Krause, B. M., Nilles, J. P., & Chapman, M. A. (2016). Respiratory neuron characterization reveals intrinsic bursting properties in isolated adult turtle brainstems (Trachemys scripta). Respir Physiol Neurobiol, 224, 52-61. https://doi.org/10.1016/j.resp.2014.11.004
Baghdadwala, M. I., & Wilson, R. J. (2014). Lamprey breathing when feeding sucks: the respiratory rhythm generator of a parasitic fish. J Physiol, 592(8), 1725-1726. https://doi.org/10.1113/jphysiol.2014.272732
2.9.9
Pacemakers, Central Pattern Generators, and Heart Rate Variability
Central pattern generators (CPGs) are self-organizing biological neural circuits found in invertebrates and virtually all vertebrates, including humans. They play a crucial role in locomotion, respiratory rhythms, or swallowing. These networks are driven by a pacemaker formed by one or more neurons that act as a central oscillator. Inhibitory neurons are also involved.
In The Human Respiratory Gate, Eckberg (2003) describes how respiratory activity gates the timing of autonomic motoneuron firing without affecting their tonic level (heart period) — in fact, respiration gates the responsiveness of both vagal and sympathetic motoneurons to changes in baroreceptor input. Breathing is necessary for R-R (peak-to-peak) respiratory frequency variation. This variation is absent during apnea. Eckberg (2009) shows that even without lung movement (e.g., inspiration), minimal activity in respiratory motoneurons is sufficient to modify HRV.
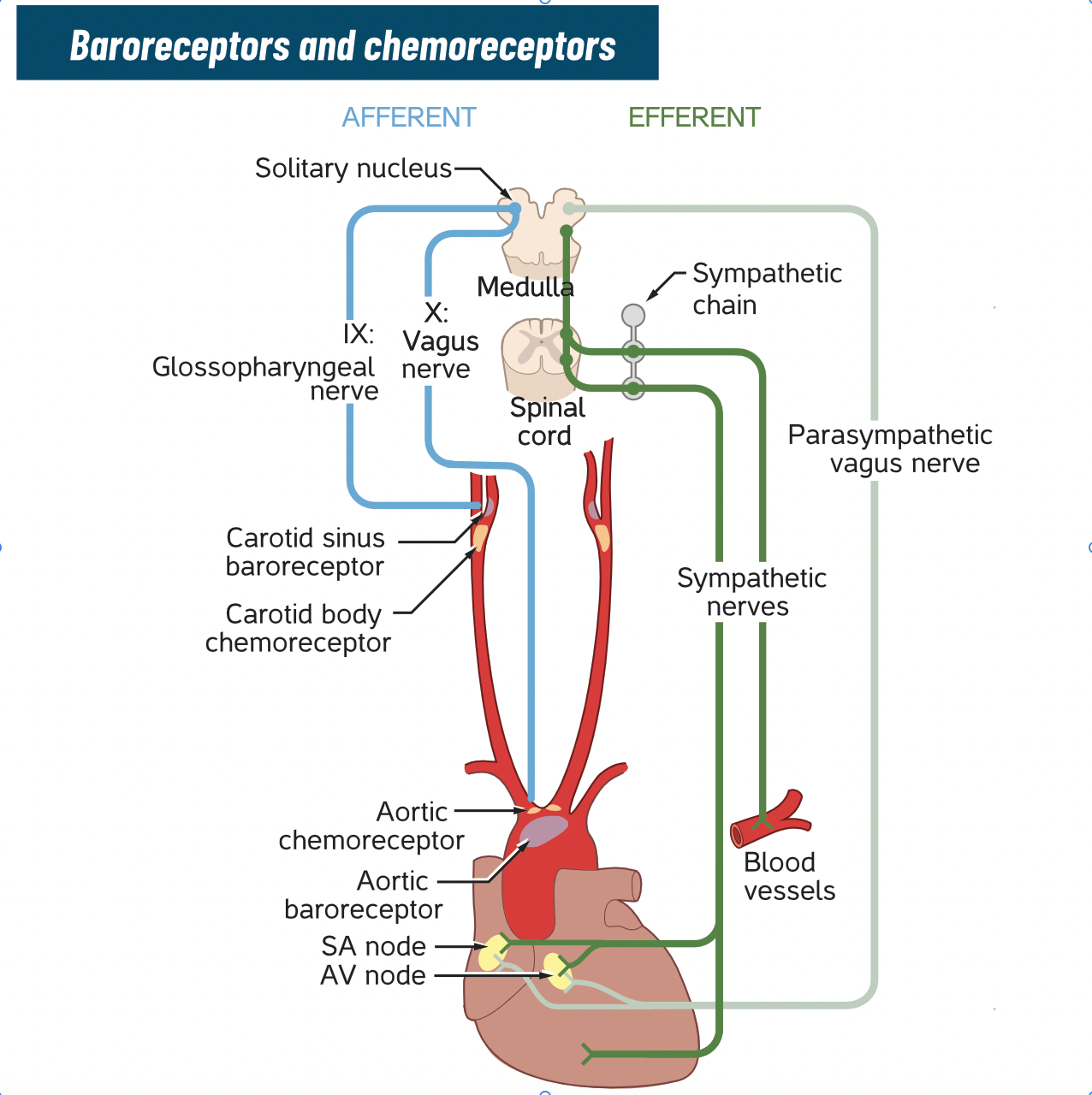

©absb, 2024
Parasympathetic Chain of Command
The myelinated ventral vagal branch provides a rapid braking action on the heart. As the vagal input decreases, the more rapid beat of the pacemaker (SA node) pushes the rhythm toward 80 BPM or more (depending on age). The sympathetic nervous system maintains its accelerator input for more intense, energetic demands. HRV is created by the constant change in distance between two heartbeats (R-R). A healthy ventral vagal input from the ventral part of the nucleus ambiguus (NAext) is required for this variability to exist. But is that all?
Neurophysiologists have discovered other sources. The NA does not have pacemaker properties like the sinus node and is inactive without input from other brainstem centers.
We now know that the NTS (the integrating center of all visceral information) is the primary hub for visceral inputs and central sources. It relays, filters, and organizes information from the baroreceptors and higher brain regions (second and third order neurons).
The respiratory medullary centers are another source (e.g., the pre-Bötzinger center, a respiratory center with pacemaker properties, located just below the ventral NAext.
Recent neuroimaging studies (Mulcahy, 2019) have shown that HRV is generated by an even broader network of brain regions (central autonomic network), including areas of the prefrontal cortex, anterior cingulate cortex, insula, amygdala, periaqueductal gray, pons, and medulla. The NAext is thus the lowest link, just before the heart, in a complex chain of command.
The Dashboard is not the Engine
HRV is not a solid or a substance that the NA produces like a gland. The dashboard of a car displays readings; it's not the engine. Similarly, HRV is an index of the balance between sympathetic acceleration and parasympathetic deceleration. It gives a greater or lesser ratio (e.g., low frequency to high frequency LF/HF). The variability we found in the signal produced by the NA reflects the variability of the neurovegetative system, including respiratory, visceral, subcortical, and cortical influences. It provides the weather report, but it doesn't make it rain or shine.
Variability and Entropy
HRV is not the only form of brain variability. When we have feedback circuits, variability (i.e., entropy) represents fitness. The sway amplitude of a standing person decreases after a concussion (minimal traumatic brain injury, or mTBI). Aging has the same effect. Neural connectivity (e.g., cortex to brainstem) is crucial. A certain amount of entropy (unpredictable) accompanies health and youth.
2.9.10
The Fetal Heart Rate
HRV and Prevention of Fetal Mortality
Porges describes HRV as a valuable parameter for detecting possible threats to the fetus. He interprets that the ventral parasympathetic branch improves HRV while the dorsal branch decreases the heart rate, sometimes to lethal levels. This view is flawed in two ways:
As we present in this document, the dorsal has minimal influence on the heartbeat (chronotropic) — the ventral has this function.
Our extensive literature review on electronic fetal monitoring has revealed many disappointing and critical comments.
In Fetal Heart Monitoring (Lyndon, 2015, pp. 132-135), a handbook dedicated to monitoring, only four of 346 pages address the topic of fetal heart variability (fHRV). Minimal variability (less than 5 bpm) without associated deceleration is almost always unrelated to fetal acidemia (blood acidity due to hypoxia). A decrease in variability is usually induced by administering analgesics or sedatives. Minimal variability is also seen during fetal sleep cycles. The absence of variability is generally alarming only in cardiac decelerations or bradycardia. However, it has little predictive value. In addition, “marked variability” (greater than 25 bpm) is thought to result from increased alpha-adrenergic activity.
How valuable is Electronic Fetal Monitoring?
Fetal heart rate monitoring has long been considered essential for assessing a baby's well-being during labor. However, electronic fetal monitoring (EFM) has not been successful as a public health screening program. Nevertheless, it continues to be used for most of the four million low-risk women who give birth in the United States each year (Grimes, 2010). Although introduced 40 years ago as a promising method to detect fetal hypoxia and potentially reduce perinatal mortality and morbidity, randomized controlled trials have not shown significant benefits. Instead, FHR monitoring has increased cesarean delivery rates and high disagreement among practitioners (Kwon, 2016). Over the past five decades, EFM has been used extensively to prevent neonatal encephalopathy and cerebral palsy, but even opinion leaders acknowledge its failure to achieve these goals (Evans, 2022; Knupp, 2020).
According to Kwon (2016), many obstetricians use CTG (cardiotocogram) to keep a hard copy of evidence that the baby was safe while in their care. This can protect them in the event of severe criticism or legal action. Even so, it has been reported that if four obstetricians examine 50 CTGs, they will only agree on 22% of the cases (Belfort, 2015).
2.9.11
The Fetal Heart Rate Variability (fHRV)
Fetal Heart Rate Variability (fHRV)fHRV reflects the heart's autonomic regulation and the maturation of the nervous system. The ANS continuously adjusts heart rate to maintain homeostasis. Fetal respiratory movements improve the reliability of heart rate variability and suggest a coupling between fetal respiratory arrhythmias and vagal activity (Zizzo, 2022) despite the fetus's lack of respiratory (breathing) function.
Breathing begins long before birth. In 1970, Dawes et al. showed that the fetal sheep makes regular respiratory movements during rapid eye movement (REM) sleep. Human fetuses also breathe (Rigatto, 2004) (Alvaro, 2011) (Hansen, 2005) as fetal breathing is readily detectable in the fetus at 10 weeks of gestation (Guttentag, 2005). Sustained periods of fetal breathing occur during REM sleep and are absent during quiet sleep. Fetal breathing promotes proper lung development (Reynolds, 2013). The increasing synchronization of fetal movement and HRV reflects the growth of autonomic nervous system (ANS) control (Brändle, 2015). Fetal heart rate is influenced by the ANS, which matures during pregnancy. In addition, several HRV parameters express the maturing influence of both ANS branches (sympathetic and parasympathetic).
Zwanenburg et al. (2021) describe the appearance of HRV (SDNN and RMSSD) between 20 and 30 weeks of gestation. This suggests that the parasympathetic branch matures rapidly at this stage of development, resulting in short-term heart rate variability. From 30 to 32 weeks, the sympathetic branch dominates, as evidenced by the appearance of long-term accelerations, mainly activated by fetal movements. After 35 weeks, a second phase of parasympathetic development, provoked by respiratory sinus arrhythmia (RSA), has been proposed. Zwanenburg et al. (2021) “could not confirm this,” and Schneider et al. (2018), based on Cheng (2003) and Porges (2011), “could only partially” confirm it.
The development of the parasympathetic system (PVT) is a complex process. It might lead the reader to believe that different populations of neurons (dorsal first and ventral later) follow each other to generate HRV. However, “ventral” vagal neurons (cardiac and branchiomotor) are active before fully myelinated and reaching the ventral position. At different stages of development, the same “ventral” population is responsible for the high-frequency portion of HRV: first unmyelinated and dorsal, later myelinated and ventral. As the ventral branch exits the ambiguus nucleus, it makes a long upward curve before leaving the brainstem. The facial nerve does the same.
Cardiac interaction between mother and infant: enhancement of heart rate variability (Suga, 2019). This study investigated whether the HRV of infants aged 3-8 months could be enhanced by influencing the HRV of their mothers (through slow breathing). The results showed an age-dependent effect of the low frequency (LF) component of maternal HRV on the LF of the infant.
Furthermore, why does Schneider limit RSA (Respiratory Sinus Arrhythmia) to the late stage, whereas the respiratory centers are already (weakly) active at 10 weeks of gestation?
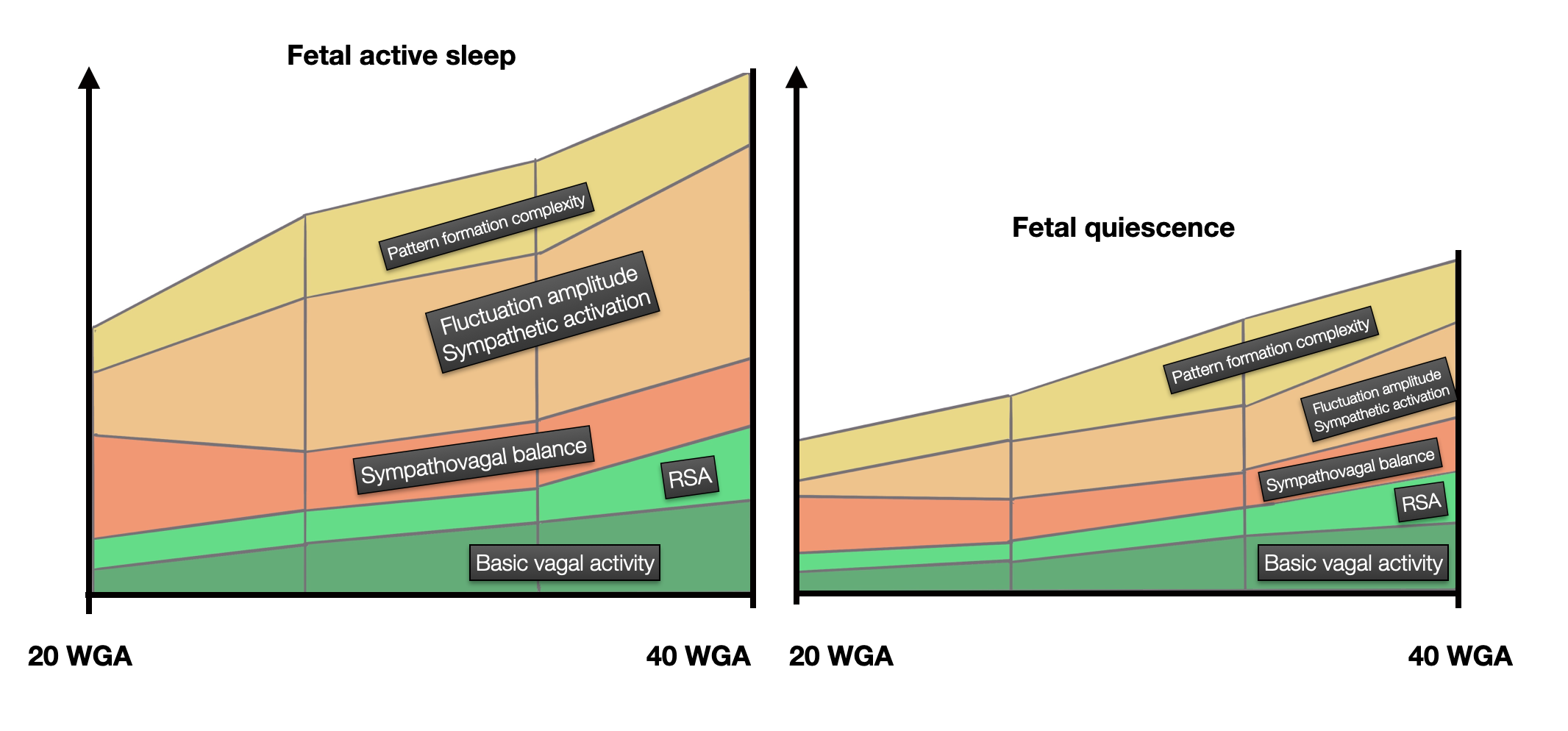
Development of the Autonomic Nervous System and Fetal HRV. (©absb 2024).
Autonomic maturation as reflected by heart rate regulation. The axis is heart rate regulation. The diagram above is adapted from Schneider (2018); the color choice is ours. Please use the link for the content and the exact diagram! As we see here, the increase from week 32 is modest.
Developmental milestones of the autonomic nervous system revealed via longitudinal monitoring of fetal heart rate variability (Schneider, 2018) states indeed that “The previous theory of ‘polyvagal development’ which sees a sequential development of vagal, followed by sympathetic and again vagal maturation, can only in parts be confirmed.“ There is no reason to postulate a second parasympathetic phase, which would “nicely fit the two-step parasympathetic development.” The authors concluded that “an increase in sympathetic activation is accompanied by a higher ability of parasympathetic modulation, and this maturational milestone is reached during the transitional period for the late second into the early third trimester.” (page 9/13). This is not the polyvagal view.
2.9.12
Challenging the Polyvagal Perspective on Heart Rate Variability
According to PVT, three systems compete to regulate the heart: “the old” dorsal vagal, the sympathetic, and the new ventral vagal systems. While the dorsal vagal has a continuous (tonic) influence and can slow the heart to death, the ventral vagal has a phasic role. It uses rapidly myelinated B-fibers to maintain vagal tone (in contrast to sympathetic tone) and generate heart rate variability (HRV), which correlates with positive health outcomes. HRV has psychological implications, as it predicts behavior (e.g., decreased HRV in antisocial groups). In A whither vagal tone (Berntson, Cacioppo, Grossman, 2007, p. 298), the authors strongly oppose the “inference”: “if φ (physiological), then ψ (psychological),” calling it a logical fallacy. “A singular measure of RSA cannot be used to infer psychological states or processes.” The polyvagal theory (...) “has several ingredients that make it attractive to researchers seeking a conceptual model for their work(...) Short of that, perhaps a more neutral designation would be something like phasic vagal cardiac control, rather than vagal tone.”
Other arguments that challenge the PVT arguments
Cardiorespiratory interactions (CRI) are not specific to mammals. On the contrary, they appear very early in evolution. According to The phylogeny and ontogeny of autonomic control of the heart and cardiorespiratory interactions in vertebrates (Taylor, 2014), coordination of cardiac and respiratory activities is already observed. In mammals and non-mammals, the feedback loop runs from the baroreceptors through the NTS (part of the DVC) before an output through one of the two vagal efferent branches. The heart rate variability signal in rainbow trout (Vera, 1991) is another example of HRV in a teleost (bony) fish.
The purpose of HVR is not psychological but energetic.
In Respiratory Sinus Arrhythmia: Why does the heartbeat synchronize with the respiratory rhythm? Yasuma (2004) describes how RSA allows respiration and circulation to synchronize, saving energy for two reasons: optimizing exchange at the level of the lungs and sparing the energetic expansion of the heart.
In Evaluating the physiological significance of respiratory sinus arrhythmia: looking beyond ventilation-perfusion efficiency (Ben-Tal, 2012), the authors discuss the hypothesis that RSA allows more efficient gas exchange between the lungs and the blood. As a new hypothesis, they propose that RSA helps the heart work less while maintaining healthy blood gas levels.
Mental health. While HRV is a good indicator of health on a physical and psychological level, it doesn't accurately predict the asocial behavior of a population (e.g., delinquency among young men). In two different articles, two independent groups of researchers (Zijlmans, 2021)(Prätzlich, 2019) found no correlation between low HRV and antisocial behavior. Another author (Fanti, 2019) found co-inhibition of the two systems (sympathetic and parasympathetic) in children and adolescents with behavioral problems rather than low HRV. Beffara et al. (see below) show that the association between HRV and “mental reading” is related to cortical functions.
Circular argument. The PVT created a circular argument: the ventral generates HVR — HVR is a vagal index — HVR is a prosocial index — the pharyngeal apparatus creates communication — communication creates safety — safety relaxes and brings up the parasympathetic tone — back to HVR. Placing facial interactions in the same functional space as the cardio-respiratory space is a mistake.
7. Respiratory centers. According to the PVT, the “ventral vagus” is the source of RSA or HRV. However, authors have recently described the interactions between the NAext, the NA, and the respiratory centers (e.g., preBötzinger).
In Heart Rate Variability — More than Heart Beats? Ernst (2017) presents an extensive list of functions in which HRV is involved: in addition to the ANS and the SA node, the respiratory, endocrinological, and immunological systems, as well as metabolic parameters. In Brainstem Sources of Cardiac Vagal Tone and Respiratory Sinus Arrhythmia (Farmer, 2016), the authors observed that blocking the respiratory center Kolliker-Fuse in the pons eliminated 88% of heart rate variability, but only 52% of chronotropic vagal tone. Blocking the NTS (part of the DVC) further reduced vagal tone. The remaining sources were not detected. It appears that cardiac vagal tone depends on neurons in at least three brainstem sites, and much of it arises independently of RSA (respiratory sinus arrhythmia).
8. HRV, Connectivity and Complexity. While HRV can be considered a positive health index, it is not a substance or a product of the VVC, like a hormone or a signaling substance! HRV measured in milliseconds is an index. Improving HRV means improving the variability of a system (brainstem/hypothalamus, cortical activity, endocrine system, or balance). Variability as an expression of vitality and complexity is not limited to the heart. Brain injury has been shown to affect the variability of spontaneous eye movements. In Concussion history is negatively associated with visual-motor force complexity: evidence for persistent effects on visual-motor integration (Raikes, 2018) and Loss of“complexity” and aging (Lipsitz, 1992); the authors describe multiple causes of reduced HRV. White matter lesions may impair connectivity at the central level. Loss of dynamic range in the EEG or hormonal deficits (anterior pituitary) are also described. Other authors have observed a decreased amplitude of sway (whole body oscillation while standing) in elderly people, which is another example of loss of complexity.
9. HRV doesn't create social engagement or security. It is not even entirely representative of the parasympathetic tone. There is no support for a direct link between the branchiomotor muscles — involved in facial movements, chewing, swallowing, and vocalization — and HRV, an index of vitality. The PVT postulates a link between HRV and the social engagement system formed by the branchiomotor nerves (V, VII, IX, X) and CN XI. The PVT doesn't consider that these nerves have been selected since the first vertebrates were mainly for catching prey, chewing, tasting, swallowing, later vocalizing, and moving the upper airway. Placing facial interactions in the same functional space as cardio-respiratory is wrong. The PVT links resting HRV to pro-social behavior. If we follow this logic, our pro-social potential goes red when we start moving, dancing, running, or doing anything silly. The sympathetic makes us aggressive and antisocial. Stop the games — all of them!
10. Anthropocentrism
In this discussion of HVR, Porges uses an anthropocentric perspective and presents a connection between HVR and the face. Furthermore, the PVT postulates an association of HRV with a group formed by the branchiomotor nerves (V, VII, IX, X) and CN XI, which he calls the “social engagement system.” These nerves and muscles have been present in identical or homologous forms since the first vertebrates. Evolution has conserved them because their ability to catch prey, chew, taste, swallow, later vocalize, and move the upper airway isn't given much consideration by the PVT. Porges claims the “pharyngeal apparatus” (branchial arches) for mammals — more precisely, for communication. Can we doubt that the furry snout of a bear or an opossum contributes massively to its social communication? The primary purpose of the face is to catch, bite, chew, and tear apart prey — plant or animal — not to smile. Somehow, the PVT twists the evolution temporal dimension. If evolution were a day, the smile would appear in the last seconds–and not all humans smile.
Further Reading and Thoughts
2.9.13
Phylogeny and Comparative Anatomy
While the PVT describes HRV as a mammalian property, various physiologists have shown this is not the case. We should remember that Porges specializes in human behavior and doesn't have first-hand knowledge of animal anatomy and physiology like Edwin Taylor and associates.
The Heart Rate Variability Signal in Rainbow Trout (Vera, 1991) describes HRV in a teleost (bony) fish, the rainbow trout, which has dual cardiac innervation with adrenergic and cholinergic elements. The cholinergic inhibitory pathways show feedback about twice as fast as the adrenergic stimulatory pathways. The two pathways vary in importance with temperature, with the cholinergic pathway (high frequency) dominating at high temperatures. Priede (1974) showed that a section of the vagus nerves abolished heart rate variation in this species.

The Edwin Taylor Group
Interested in evolutionary biology and phylogenetic considerations? This is a list of particularly relevant articles authored or co-authored by Edwin Taylor:
Central Control of the Cardiovascular and Respiratory Systems and Their Interactions in Vertebrates (Taylor, 1999).
Toward understanding respiratory sinus arrhythmia: Relations to cardiac vagal tone, evolution and biobehavioral functions (Grossman, Taylor, 2007).
Autonomic control of cardiorespiratory interactions in fish, amphibians and reptiles (Taylor, 2010).
The phylogeny and ontogeny of autonomic control of the heart and cardiorespiratory interactions in vertebrates (Taylor, 2014).
Cardiorespiratory interactions previously identified as mammalian are present in the primitive lungfish (Monteiro, 2018).
Respiratory sinus arrhythmia is a major component of heart rate variability in undisturbed, remotely monitored rattlesnakes, Crotalus durissus (Sanches, 2019)
An overview of the phylogeny of cardiorespiratory control in vertebrates with some reflections on the ‘Polyvagal Theory’ (Taylor, Wang, Leite, 2022)
2.9.14
Beauchaine: Is Heart Rate Variability a Social Biomarker?
In five articles published between 2001 and 2019, Beauchaine had long tried to prove a correlation between low HRV and antisocial traits. Although initially confident, his tone became much more cautious in the last publication (2019). He concludes that “associations between RSA reactivity and psychopathology are complex and require better standardized RSA assessments to increase validity and decrease errors.”
Vagal tone, development, and Gray’s motivational theory: Toward an integrated model of autonomic nervous system functioning in psychopathology (Beauchaine, 2001)
Polyvagal Theory and Developmental Psychopathology: Emotion Dysregulation and Conduct Problems from Preschool to Adolescence (Beauchaine, 2007)
Heart rate variability as a transdiagnostic biomarker of psychopathology (Beauchaine, 2015)
Motivation, emotion regulation, and the latent structure of psychopathology: An integrative and convergent historical perspective (Beauchaine, 2017)
Respiratory sinus arrhythmia reactivity across empirically based structural dimensions of psychopathology: A meta‐analysis (Beauchaine, 2019)
2.9.15
Does Low Heart Rate Variability Make You Asocial?
Resting high frequency heart rate variability selectively predicts cooperative behavior (Beffara, 2016) is an interesting critical study that questions the claim that higher vagal tone (high frequency or HF-HRV) is associated with better emotion recognition skills. The conclusion is ambiguous, acknowledging that in certain cases an association between HF-HRV and pro-social behavior could confirm the claims of the Polyvagal Theory for healthy human adults. However, an article published a few months later suggests a different conclusion.
Resting high-frequency heart rate variability is not associated with recognition of emotional facial expressions in healthy human adults (Beffara, BiorXiv, Sept. 27, 2016). This study examines whether the vagal connection between the heart and brain is involved in prosocial behavior. Polyvagal theory postulates that vagal activity underlies prosocial tendencies. Although several findings may show that vagal activity is associated with prosocial behavior, none have used behavioral measures of prosociality to establish this relationship. As it seems, the association between HF-HRV and performance on the “mental states” reading (Quintana et al., 2012) cannot be explained by better emotion recognition skills. The more plausible explanation should consider attention, working memory, and executive functions. Recent studies show that individuals with higher HF-HRV perform better on cognitive tasks depending on executive and attentional functioning. Thayer's neurovisceral integration model (Thayer & Lane, 2000) provides a theoretical framework for understanding the relationship between HF-HRV and attention. Neural control of the heart is highly dependent on cortical inputs, particularly from the prefrontal cortex (PFC), insula, and anterior cingulate cortex (ACC). The observed heart rate variability is mainly influenced by attentional shifts, conflict monitoring and inhibition. The authors conclude:: the current study does not allow to conclude that resting HF-HRV predict emotion recognition, even taking emotion type into account.“
Heart Rate Variability in Schizophrenia and Autism (Haigh, 2021)shows a more significant decrease in HRV in schizophrenic patients than in autistic patients.
Impact of maternal emotional state during pregnancy on fetal heart rate variability (Semeia, 2023). This study evaluates how depression, anxiety, and stress during pregnancy are related to fetal heart rate variability (fHRV).
Can we test the influence of prosociality on high frequency heart rate variability? A double-blind sham-controlled approach (Beffara, 2016) posted on Biorxiv on Sept. 28, 2016, concludes: “We aimed to test a possible causal pathway of the polyvagal theory (Porges, 2007) according to which prosocial behaviors can positively impact heart-brain dynamics. Our data does not support the theory in this direction. We suggest that further studies attempting to test the theory should focus on rigorous methodological features such as double-blind design protocols.”
Brain correlates of autonomic modulation: Combining heart rate variability with fMRI (Napadow, 2008)
Heart rate variability as a biomarker in health and affective disorders: A perspective on neuroimaging studies (Mulcahy, 2019) shows an association between health outcomes, behavioral characteristics, psychological functioning, and heart rate variability. HRV measures the allostatic regulation of homeostasis within the cardiovascular system. A network of brain regions (“central autonomic network”) maps the internal state and controls autonomic responses. This network includes the prefrontal cortex, anterior cingulate cortex, insula, amygdala, and PAG. Human neuroimaging studies of neural activation and functional connectivity broadly support this architecture and its association with cardiac regulation at rest and dysregulation in clinical states, including affective disorders. In this review, neuroimaging research and evidence for HRV are informative markers of autonomic integration with affect and cognition.
Pitfalls of assessment of autonomic function by heart rate variability (Hayano, 2019)
Heart-Rate variability—More than Heart Beats? (Ernst, 2017) compares different models of integration.
Considerations in the assessment of heart rate variability in biobehavioral research (Quintana, 2014)
The Current and Future Role of Heart Rate Variability for Assessing and Training Compassion (Kirby, 2017) explores the possibility of developing compassion through, among other things, vagal stimulation.
2.9.16
Heart Rate Variability and the Sympathetic Nervous System
Two articles describe the role of local adrenergic (sympathetic) centers in rhythm generation
Brainstem sources of cardiac vagal tone and respiratory sinus arrhythmia (Farmer, 2016, p. 7263). Intrinsic rhythmicity of sympathetic central oscillatory circuits. The cardiovascular region of the NTS generates spontaneous pacemaker-like spike activity with projections to the NA.
Acquirement of the autonomic nervous system modulation evaluated by heart rate variability in medaka (Oryzias latipes) (Watanabe-Asaka, 2022). Sympathetic and parasympathetic are responsible for the HRV.
2.9.17
Complexity and Variability In Biological Systems

Altered functional connectivity between medial prefrontal cortex and the inferior brainstem in major depression during appraisal of subjective emotional responses: A preliminary study (Smith, 2015). Reduced rostral anterior cingulate cortex (rACC)-subcortical functional connectivity to a region of the inferior pons in depressed subjects may account for their autonomic dysregulation. Within-subject differences in rACC-pons connectivity were also significantly correlated with measures of heart rate variability and depression severity. These findings suggest that autonomic dysregulation in depression may be associated with a functional disconnect between the rACC (cingulate cortex) and autonomic brainstem nuclei.
Metabolic flexibility. Eliminating carbohydrates from the diet or fasting forces the body to adapt and find new metabolic pathways. In searching for the best way to optimize the body's metabolism (e.g., for athletic performance, diabetes management, or weight loss), researchers have introduced the concept of metabolic flexibility (Goodpaster, 2017) (Palmer, 2022). Metabolic flexibility is the ability of the human body to switch between fat and carbohydrates based on their availability. Twin sisters Merav and Michal Mor have developed a handheld metabolic tracker that measures carbon dioxide levels in the breath. Under the name Lumen www.lumen.me, they propose a diet and exercise program to improve metabolic flexibility, or the ability to switch between burning carbohydrates (carbs) or lipids (fats). This flexibility, which was critical to the survival of prehistoric hunter-gatherer tribes, helps with weight loss (Roberts, 2023). Interval fasting is a way to replicate this survival adaptation — on a small scale — and serves hormesis (see “What else?”).
Concussion history is negatively associated with visual-motor force complexity: evidence for persistent effects on visual-motor integration (Raikes, 2018). Asymptomatic individuals with a history of concussion show subtle, persistent increases in postural sway regularity compared to pre-injury values or healthy controls. Regularity reflects the degree to which future system states are predictable, i.e., have lower entropy than complex signals. In this study, the authors observed that individuals with multiple previous concussions had greater regularity than individuals with a single or no previous concussion during isometric visual-motor force tracking.
Metabolic complexity increases adaptability (Fritzemeier, 2023). However, it is unclear what determines the ability of individual strains or species to adapt to novel environments. Here, we use pan-genomic modeling to investigate the ability of 102 different single-celled organisms to adapt to each of 5000+ different nutrient environments. While the small metabolic systems of specialized endosymbionts typically require 50+ additional metabolic reactions to adapt to new environments, various strains of the generalist E. coli require, on average, less than 5 new reactions. Thus, there is positive feedback between metabolic complexity and adaptability, contrary to speculation that complex systems are generally less adaptable.
2.9.18
White Matter and Connectivity
Memories of attachment hamper EEG cortical connectivity in dissociative patients (Farina, 2014). However, dissociative patients did not show increased EEG connectivity after the attachment interview (AI) compared to controls. Conclusions: These findings shed light on the neurophysiology of the disintegrative effect of retrieving traumatic attachment memories in dissociative patients.
Decreased EEG connectivity is related to dissociative absorption (Soffer-Dudek, 2019). This study examines whether trait dissociation relates to lower EEG signal connectivity. Dissociative absorption manifests in segregated cortical activity.
General psychopathology factor and unresolved-disorganized attachment uniquely correlated to white matter integrity using diffusion tensor imaging (Riem, 2018) shows processes of demyelination in the genu and corpus callosum correlating with general psychopathology (GPF).
White matter changes in children and adolescents with reactive attachment disorder: A diffusion tensor imaging study (Makita, 2020). Alterations of the white matter in the brain correlates with attachment disorders.
Neurophysiological correlates of dissociative symptoms (van der Kruijs, 2014). HRV, EEG, and (functional) MRI are sensitive methods for detecting physiological changes related to dissociation and dissociative disorders.
Functional reorganization of neural networks involved in emotion regulation following trauma therapy for complex trauma disorders (Schlumpf, Nijenhuis, 2019).
Before treatment and compared to controls, patients showed hypoconnectivity within neural networks involved in emotional downregulation. After treatment, connectivity normalized in these networks, which include regions associated with cognitive control and memory.
2.9.19
Heart Rate Variability and Aging
Which role do age and disease play in complexity?
Loss of Complexity and Aging (Lipsitz, 1992). Healthy physiological function is characterized by a complex interaction of multiple control mechanisms that enable an individual to adapt to the demands of daily life and unpredictable changes. The aging process is characterized by progressive impairment of these mechanisms, resulting in a loss of the dynamic range of physiological function and, consequently, a reduced capacity to adapt to stress. Physiological aging is characterized by a generalized loss of complexity in the dynamics of healthy organ function. Authors have observed a decreased amplitude of sway (oscillation of the whole body while standing) in the elderly.
Changing complexity in human behavior and physiology through aging and disease (Vaillancourt, 2002).
Accelerated Changes in White Matter Microstructure during Aging: A Longitudinal Diffusion Tensor Imaging Study (Sexton, 2014)
BOLD and EEG signal variability at rest differently relate to aging in the human brain (Kumral, 2020)
Complexity analysis of heartbeat-related signals in Brain MRI time series as a potential biomarker for ageing and cognitive performance (López Pérez, 2020) Heartbeat-evoked signals (HES) exist in the brain and their complexity decreases with age. Consistent with the idea that higher complexity is associated with healthier dynamics, the authors show quantitatively that the decrease in these fluctuations is associated with a decrease in the complexity of the signal time series with age. In addition, some aspects of higher complexity were associated with better cognitive performance that was not age-related. Thus, HES can discriminate between brain functions that decline with age and those that do not. Overall, the complex properties of the HES provide a potential mechanism for understanding brain dynamics.
Signatures of white-matter microstructure degradation during aging and its association with cognitive status (Coelho, 2021)
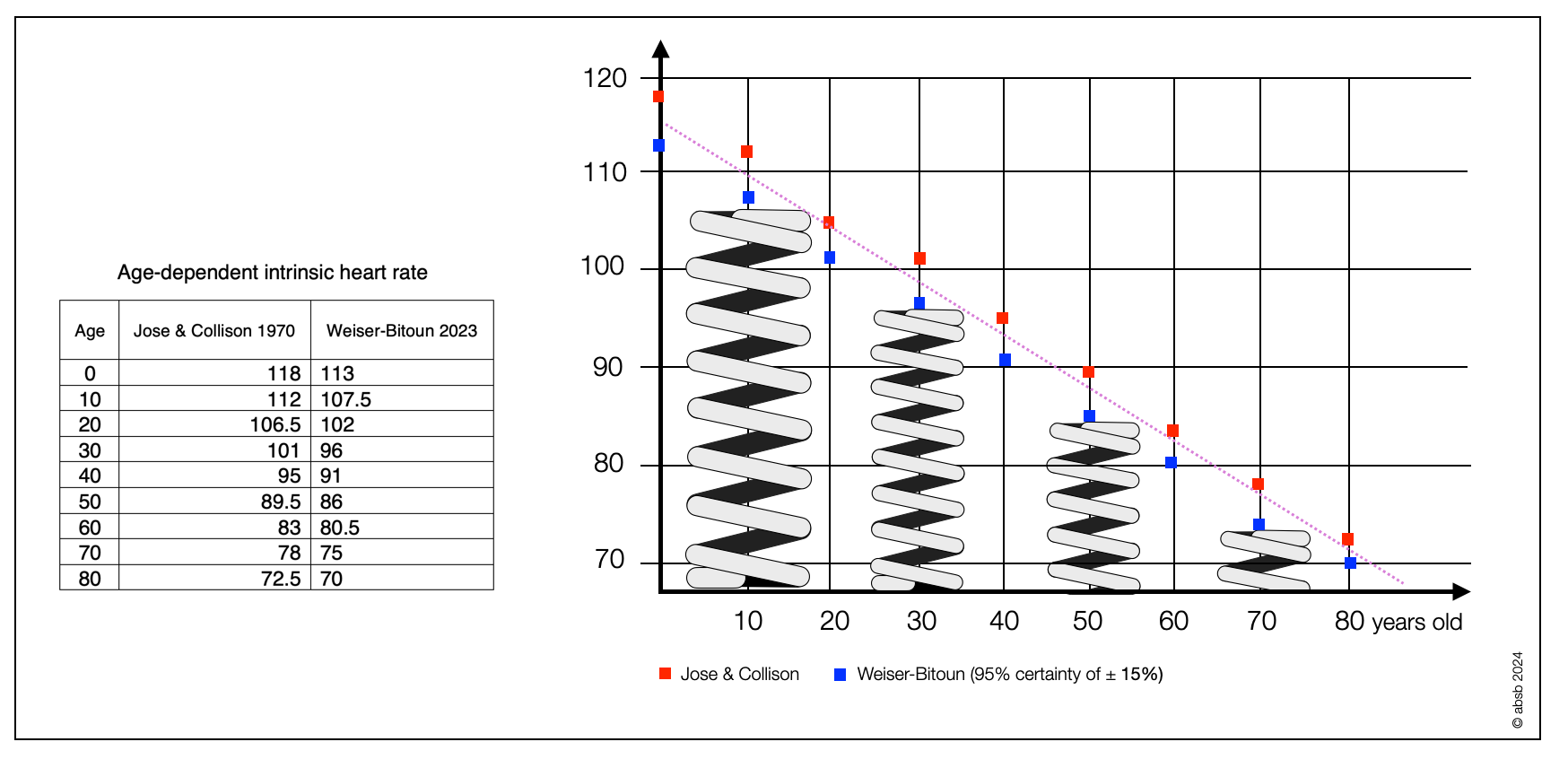
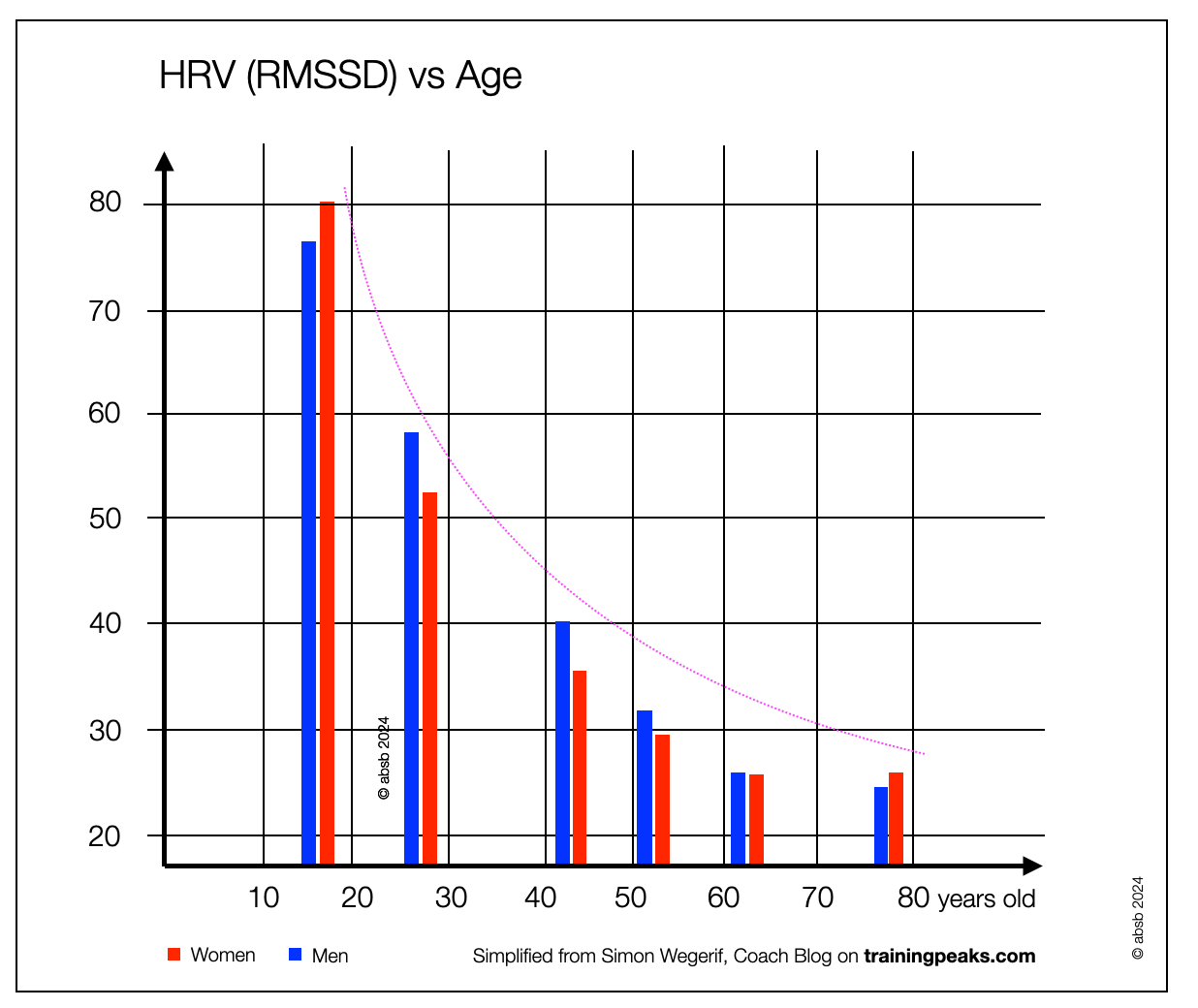
With age, HRV decreases — corresponding to a decrease in parasympathetic tone — but so does intrinsic rhythm. As a result, the increase in heart rate in the elderly generally remains constant or increases only slightly.
2.9.20
Does Heart Rate Variability Save Energy?
The PVT tries to prove a psychological role for HRV (or RSA). Grossman (2023) and Taylor (2022) have recently expressed doubts. The latter hypothesizes that “RSA could represent a relic of a system evolved in ancestral reptiles or at the origins of the tetrapods, as exemplified by the lungfishes.” Various authors (Hayano, 1996; Giardino, 2003; Elstad, 2012) have attempted to explain the importance of HRV in optimizing cardio-pulmonary gas exchange and circulatory efficiency. Ben-Tal et al. (2014) propose that RSA minimizes the work done by the heart while maintaining a desired average partial pressure of CO2.
Many studies are showing that RSA is diminished or absent in a variety of chronic diseases, including cardiovascular disease and heart failure. Enhancing respiratory sinus arrhythmia increases cardiac output in rats with left ventricular dysfunction (O'Callaghan, 2020), is a study investigating the benefit of RSA by rats with left ventricular dysfunction. “The benefits include blood pressure stabilization, cardiopulmonary gas exchange optimization, and increased cardiac energy efficiency.” Similar studies have been made without the same heart rate variability. Pacemaker create a monotone heart rate.
>> to the next chapter Hearing & Autism