2.6.0
Dorsal Vagal Complex (DVC)
In this chapter, we explore the anatomy and the functions of what the Polyvagal Theory calls the “Dorsal” – in the light of current science.
2.6.1
Does the Dorsal Vagal “Kill Babies”?
Of all the postulates of the PVT, the description of the Dorsal Vagal Complex (DVC) is the most fictional and unscientific regarding current scientific knowledge. Before we present the facts, let's go back thirty-two years to about 1992. Porges had just published Vagal Tonus: a Physiological Marker of Stress Vulnerability, in which he praised the vagal tonus as a health index. Soon after, he received a letter from a neonatologist explaining that he had learned from his teacher that too much vagal tone is lethal: it kills babies. Deeply shocked, Porges kept the letter in his pocket for months until he found a solution to exonerate the parasympathetic from this unacceptable accusation — two origins of the vagal nerve. A polyvagal solution. One branch is healthy; the other is deadly — good and new versus old and bad. There is no turning back.
Orienting a defensive world: mammalian modifications of our evolutionary heritage. A Polyvagal Theory (1995) is the printed version of his lecture in Dallas on October 8, 1994. In this narrative, which continues to grow and add detail over the next 30 years, the “dorsal” becomes the cause of all sorts of ills: fatal bradycardia, fainting, depression, dissociation — the list is long. In this chapter, perhaps to your astonishment, we will discover the life-supporting functions of the DVC according to current literature.
The Dorsal Vagal Complex (Bauer, 2005), or DVC, is a term that scientists rarely use. They prefer to be more specific and refer to each part individually: Nucleus of the tractus solitarii (NTS), dorsal motor vagal nucleus (DMVN, DVMN, or DMX), or area postrema (AP). All three structures have specific functions that are regularly involved in control circuits.
The NTS is part of complex circuits that regulate the heartbeat or digestion. Systematically grouping them into a complex called “dorsal” can create a fallacy of composition.
The nucleus tractus solitarius (NTS) is the central visceral hub. It receives multiple signals (not only vagal) and processes them before sending them up to higher centers and down to the viscera.
The dorsal motor vagal nucleus (DMVN) is an efferent, motor, parasympathetic branch that controls most of the viscera (except the heartbeat).
The area postrema (AP) receives and scans multiple pieces of information (e.g., vagal input, and blood) shared with the NTS, NA, and some higher centers.

This graphic shows a simplified view of two of the main feedback loops that connect the vagal brainstem centers to the visceral structures (heart, intestines, and stomach). Polyvagal theory considers only the first loop and ignores the second. We also see that the NTS has a central position in the Heart Rate Variability (HRV) regulatory loop. Finally, the NTS has multiple functions and different populations of neurons (@absb, 2024).
2.6.2
Anatomy of the Dorsal Vagal Complex
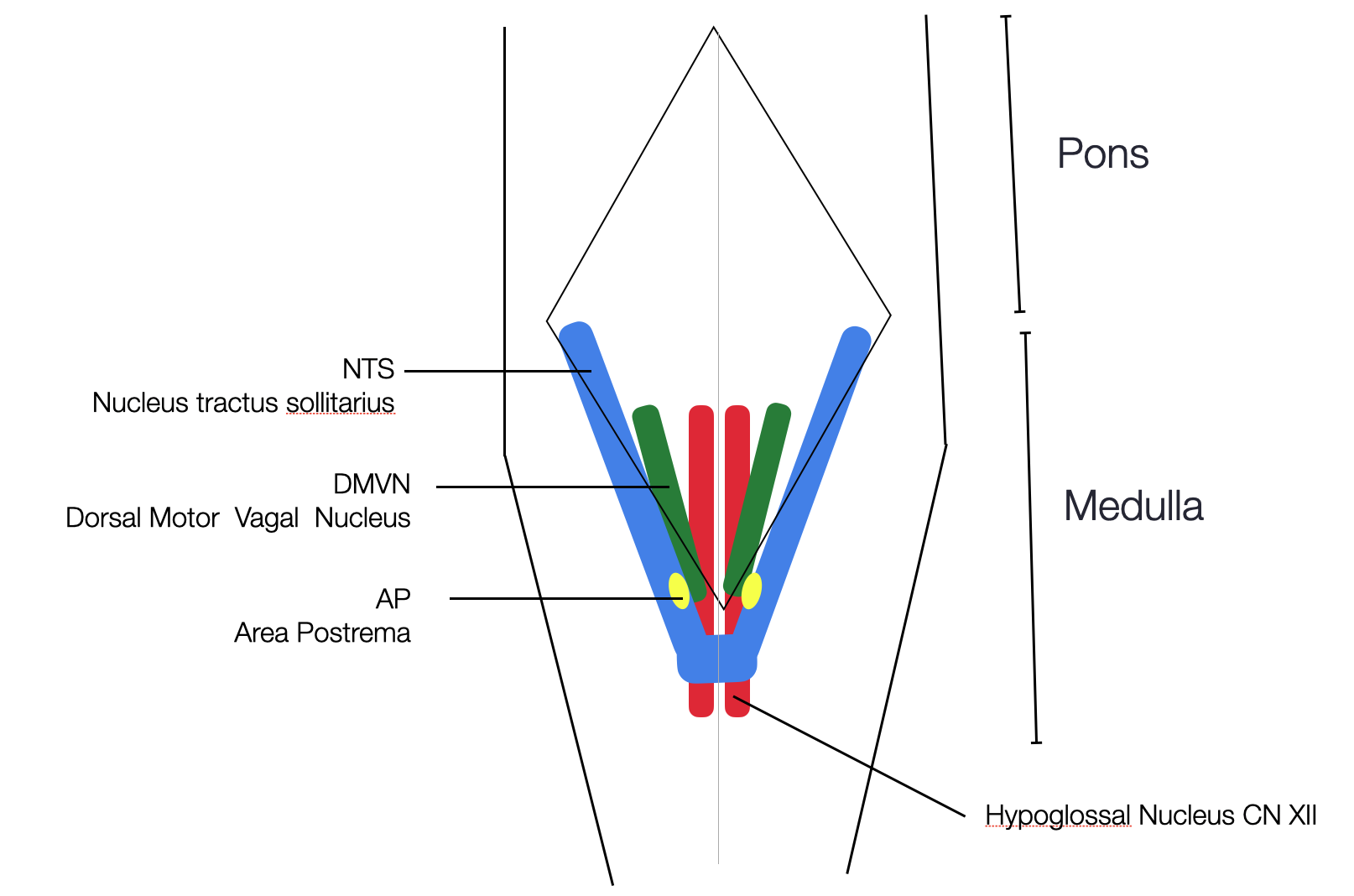
This simplified graphic is based on a 3D reconstruction of the morpho-functional topography of the human vagal trigone (Emmi, 2021). The two NTS merge in their lower part. In humans. N.B. We find two APs in humans and one central AP in mice /©absb, 2024).
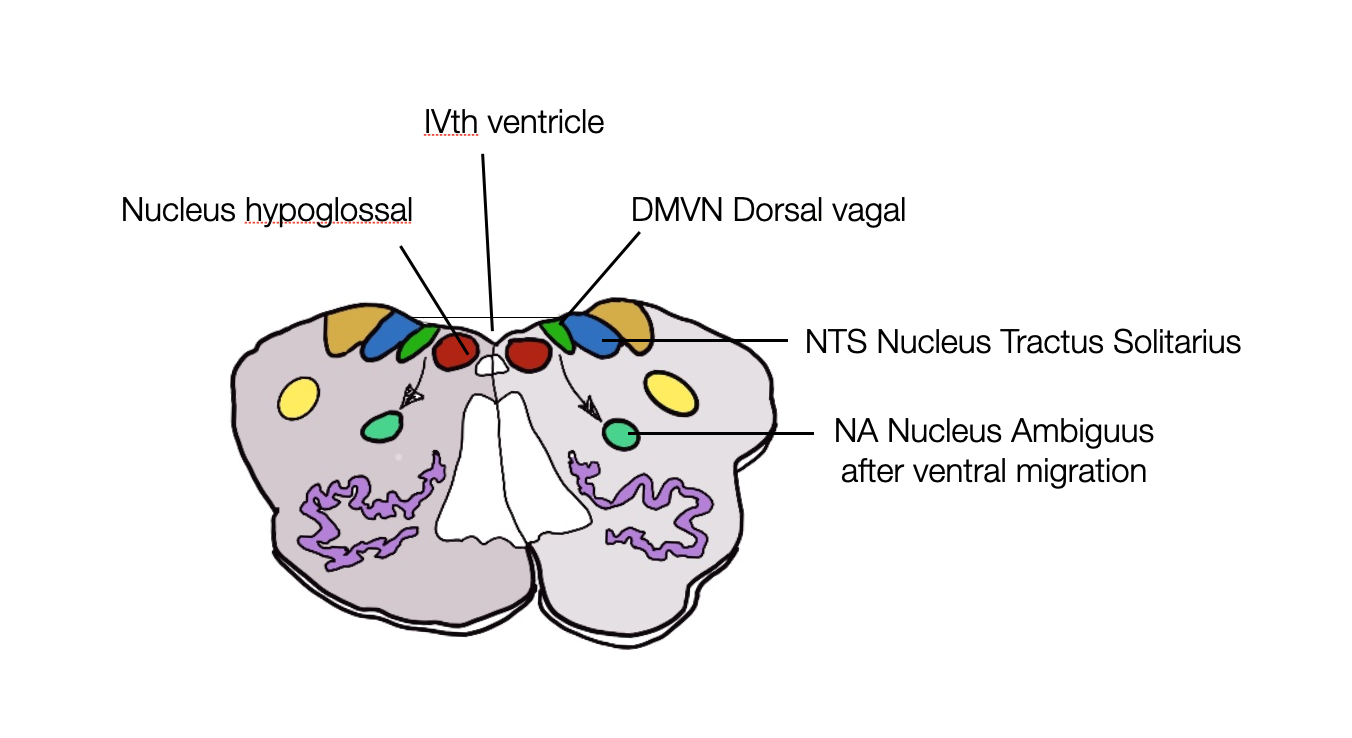
Cross-section of the brain stem: dorsal (NTS, DMVN, CN XII) vs. ventral (NA) nuclei @absb, 2024).
2.6.3
The Nucleus Tractus Solitarius (NTS)
It's easy to get caught up in the complexity of the NTS chapter. We'll try to simplify our presentation without sacrificing the richness of the role the NTS plays in visceral balance. The NTS is a collection of different subnuclei that form a Y- or V-shaped double column in the dorsal part of the inferior brainstem. Depending on the level of the coronal section of the medulla, the NTS appears as two separate circular structures or as a single central structure with all intermediate forms.
The discovery of the NTS was a great surprise for us, as we admired the neuronal architecture and the richness of the connections, in complete contrast to the dark descriptions presented by the PVT.
-
Afferent myelinated (A) and unmyelinated (C) fibers.
Taste information (VII and X), chemo- and mechanoreceptors from the carotid (CN IX), aortic arch, and sinoatrial node (X),
Chemically and mechanically, information from the heart, lungs, airways, gastrointestinal system, pharynx, and liver via the nerves IX and X nerves and from facial cavities (Nose, palate, and sinus) via the nerve VII.
Central inputs. Holt (2019) lists 9 telencephalic, 9 diencephalic, 9 mesencephalic (midbrain), and 16 brain stem afferent sources to the NTS. The NTS receives visceral and psychological inputs, making it the most significant body—mind interface in cooperation with the hypothalamus.
The NTS displays a topographic organization of the centers receiving inputs from the visceral organs. Heart and lung information terminate in specific subnuclei.
-
The following cranial nerves are afferent (sensory) to the NTS:
I (olfactory nerve): information about the smell.
V (trigeminal nerve): sensation from the face, head, and oral cavity.
VII (facial nerve): taste sensation from the anterior two-thirds of the tongue.
IX (glossopharyngeal nerve): taste sensation from the posterior one-third of the tongue and general sensation from the pharynx.
X (vagus nerve): Carries information about the visceral sensation from the thoracic and abdominal organs and general sensation from the larynx, pharynx, and esophagus.
The NTS plays a critical role in regulating various autonomic functions, such as blood pressure, respiration, gastrointestinal motility, and reflexes related to vomiting, coughing, and swallowing.
-
Output
The NTS counts nearly as many output ways (to higher centers and brainstem) as the input ways previously evocated.
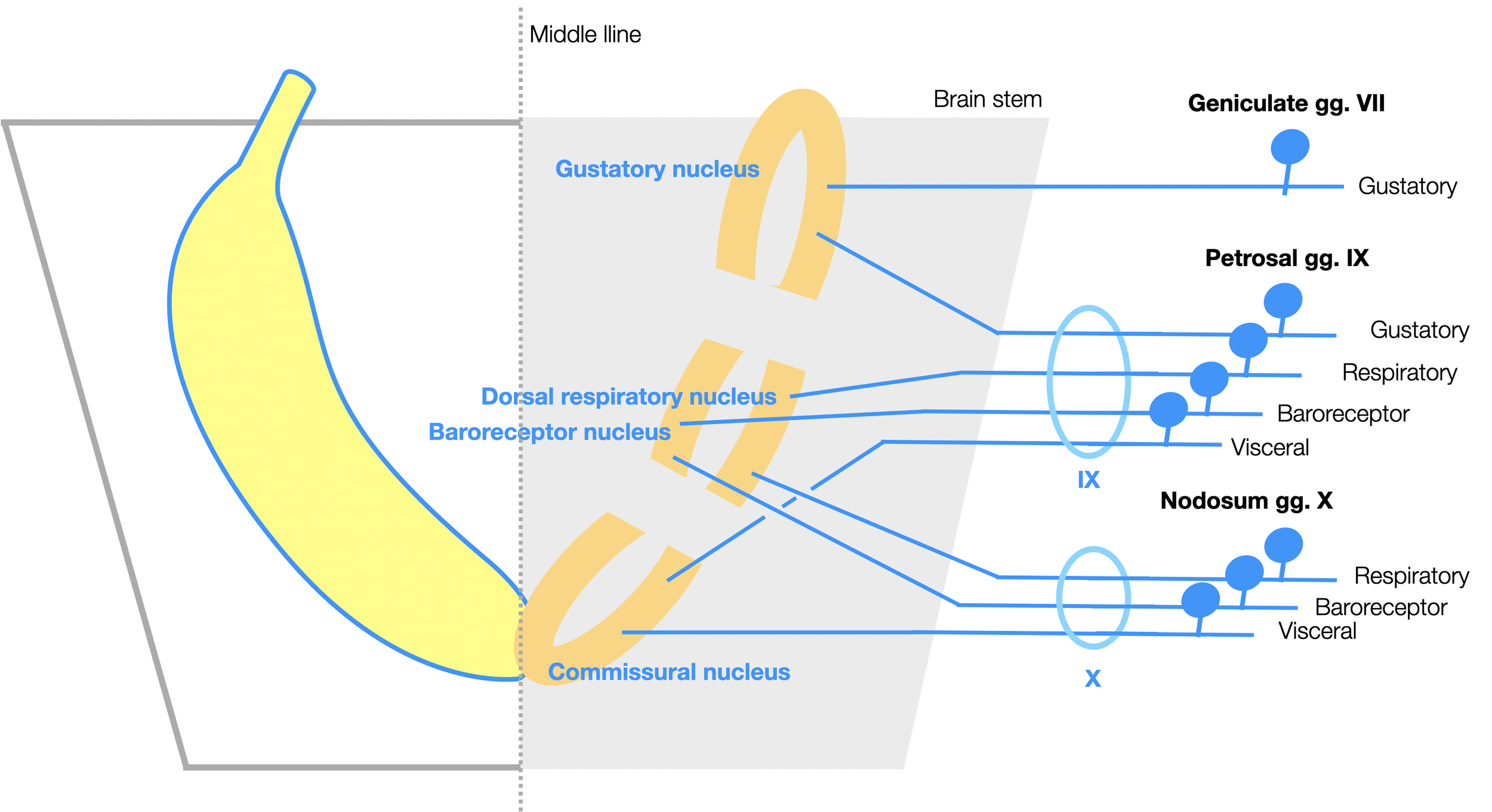
The Nucleus Tractus Solitarius. When viewed from above, the NTS looks like two bananas placed around the fourth ventricle, meeting at the lower end. Each side has three parts:
Rostral (anterior) part: gustatory nucleus (input from VII and IX).
Middle part: baroreceptor (blood pressure) and dorsal respiratory nuclei (input from IX and X).
Caudal part (posterior): commissural nucleus - connects with contralateral nucleus (visceral input from IX and X).
All afferent nerves to the NTS that we see here keep their cell bodies outside the brain in three ganglia (Geniculate, Petrosal, and Nodosum).
Our diagram is based on a graphic in Fitzgerald’s Clinical Neuroanatomy, 8th edition (Mtui, 2021, p. 217). We added the banana! (©absb, 2024).

This diagram shows how the NTS forms two vagal feedback loops, one with the NAext (regulation of the heartbeat) and a second with the DVMN (metabolic regulation of glucose and appetite).
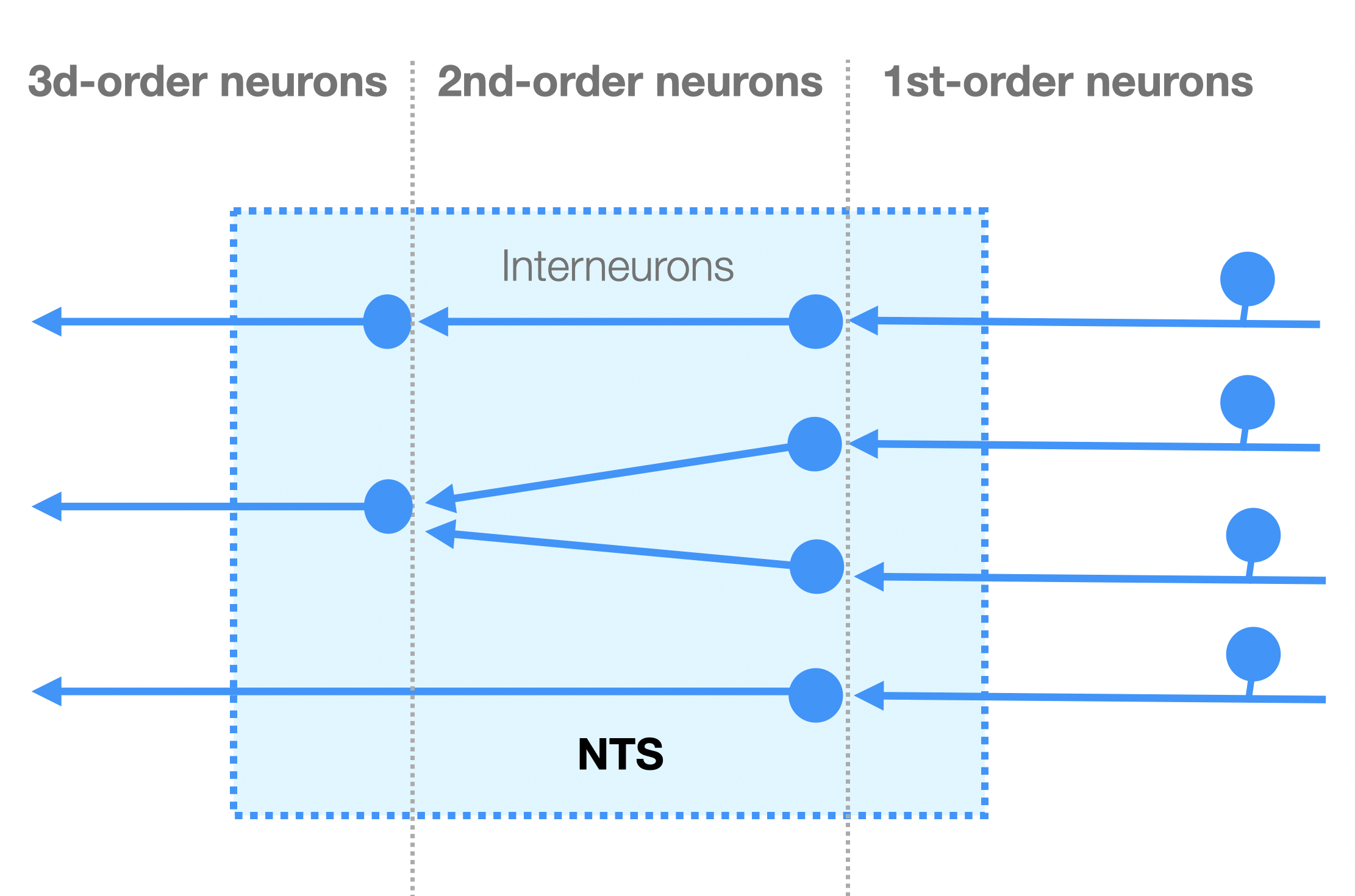
Let's compare this to the spinal reflex arc (second diagram). We see afferent fibers (in green) from the organs or skin, with their cell bodies in the ganglia connecting to interneurons (in yellow) before the signal returns to the muscles or organs.
Similarly, we find three levels of neurons between the periphery, the NTS (integration), and the efferent parasympathetic nerves.
1st order neurons are vagal, afferent sensory neurons with cell bodies in the ganglia outside the brain (nodose ganglion). Afferent fibers to the NTS make up 80% of the vagal nerve.
2nd order neurons, the interneurons in the NTS, play a crucial role in this process by acting as signal transducers (e.g., GABA or glutamate receptors).
3D-order efferent neurons synapse with neurons outside the NTS but inside the brain, which execute the final responses (e.g., bradycardia via the NAext or digestive action via the DMVN).
In a neuroanatomical sense, the NTS forms a complex of interneurons, modulating and computing myriads of information — an intermediary between afferent and efferent nerves. Like the area postrema (AP), it is primarily visceral–not vagal.

As a reminder, we show a spinal reflex arch. The double loop model presented earlier is functionally similar to the spinal reflex arch, assembling sensory and motor neurons with local modulation by interneurons (e.g., the NTS).
2.6.4
The NTS is the Visceral Hub of the Brainstem
As Andresen and Paton (2004) explain in Differentiation of autonomic reflex control begins with cellular mechanisms at the first synapse in the nucleus tractus solitarius, the NTS provides the central nervous system (CNS) with a continuous update on the status of the visceral organs: respiratory, cardiovascular, and gastrointestinal (see also Andresen & Paton in Llewellyn-Smith, 2011). Visceral and sensory inputs from cranial nerves V, VII, IX, and X synapse in the NTS. As a vast visceral hub, the NTS uses a sophisticated system of glutamatergic (excitatory) and GABAergic (inhibitory) “second-order” neurons to filter, integrate, and process information coming from the viscera (e.g., heart, lungs, and digestive system). From there, efferent pathways project to parasympathetic centers (DVMN and nucleus ambiguus), sympathetic centers (RVLM and CVLM), respiratory centers, and supramedullary centers (e.g., hypothalamus, insula, and prefrontal cortex). Thus, the NTS is the primary regulator of the ventral vagal parasympathetic nervous system.
From specific chemoreceptor and mechanoreceptor information via the IX and X cranial nerves and inputs from respiratory and higher centers, the NTS is the driving force for the NAext (“ventral vagal”). The NAext doesn't calculate; it's the executor.
Far from being a simple transit station that generates reflex responses to afferent inputs, the NTS has many interneurons, either excitatory (glutaminergic) or inhibitory (GABAergic), that modulate the inputs. The NTS mediates complex reflexes that require joint action at multiple levels: the gag reflex, the carotid sinus reflex, the aortic reflex, the cough reflex, the baroreceptor and chemoreceptor reflexes, several respiratory reflexes, and reflexes within the gastrointestinal system regulating motility and secretion. The dorsal respiratory group (DRG), part of the NTS, helps limit lung inflation and maintains the response to hypoxia (Appenzeller, 2022, p. 312).
NTS neurons also play an essential role in coordinating respiratory and sympathetic adjustments in response to inputs such as baroreceptors, pulmonary stretch receptors, and peripheral chemoreceptors (Zoccal, 2014).
This diagram, based on a graphic from The Nucleus of the Solitary Tract (Andresen, Paton) in Central Regulation of Autonomic Functions (Llewellyn-Smith, 2011), provides a simplified understanding of the integrative function of the NTS. GABA and glutamate 2nd order neurons allow modulation of the massive flow of afferent information.
The NTS is highly complex, considering all afferent and efferent outputs. Multiple processes of integration and modulation (e.g., glutamate and GABA) occur through the presence of second- and third-order neurons. In The Ins and Outs of the Caudal Nucleus of the Solitary Tract (Holt, 2022), the author cites about fifty recent studies of the NTS involving no less than 27 neurochemical groups: neuropeptides, small molecules (GABA, glutamate, norepinephrine), and intracellular proteins. Functional subclasses of NTS neurons don't always overlap with well-defined subdivisions (cytoarchitecture).
Glial Modulation of Energy Balance: The Dorsal Vagal Complex Is No Exception (Troadec, 2022).
A brain stem map for visceral sensations (Ran, 2022).
2.6.5
The Dorsal Vagal Complex Regulates the Appetite
Peripheral neural pathway: Vagal sensory receptors respond to gastrointestinal production of GLP-1 by sending a signal to the NTS.
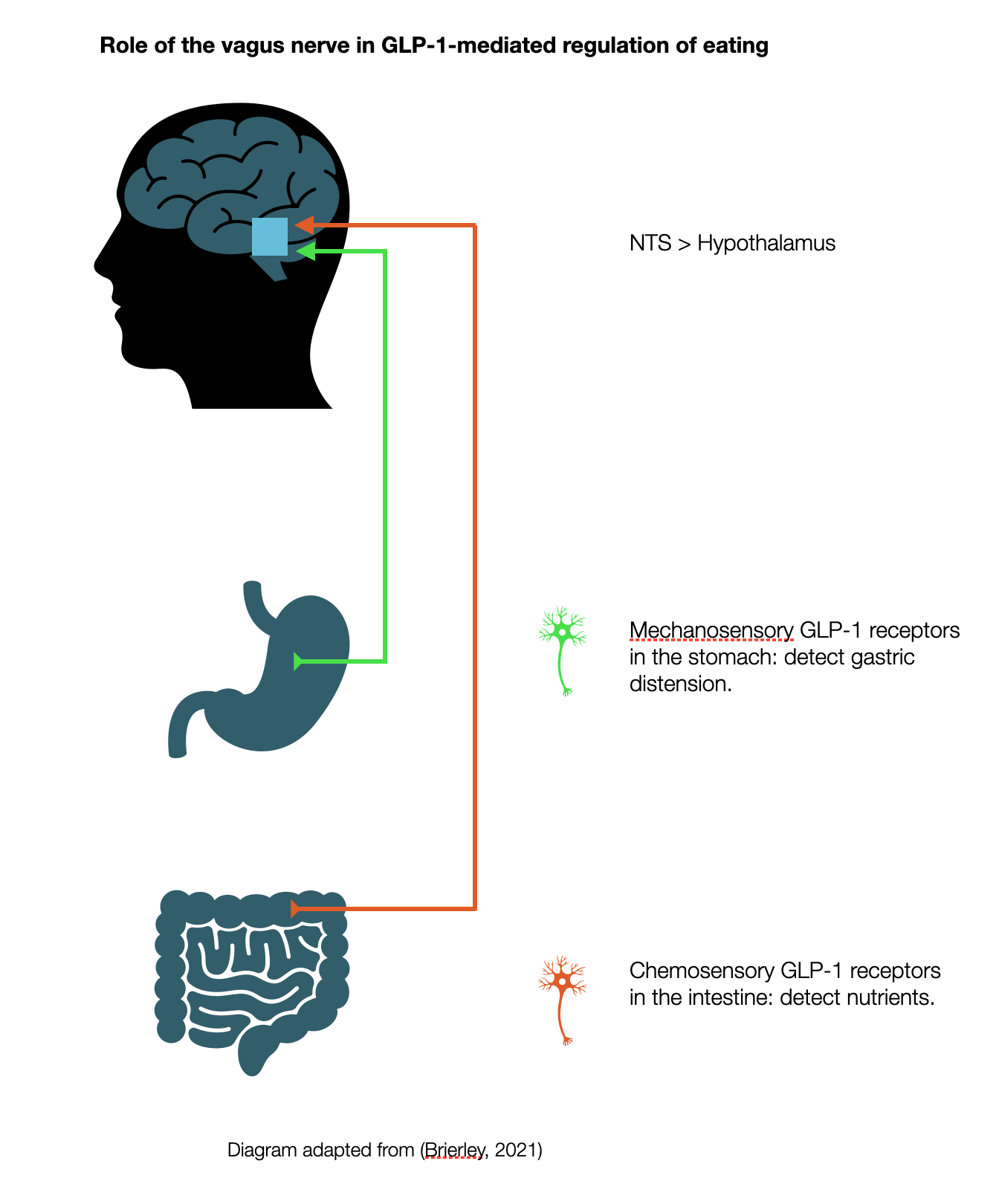
Ozempic, Wygovy, Tirzapide- Recently, everyone has been talking about the "magic pill" (Hari, 2024) to help people lose weight. But did you know that this "magic pill" mimics the physiological functions supported by the Dorsal Vagal Complex (DVC)? These are all brand names for semaglutide, a GLP-1 receptor agonist. GLP, or glucagon-like peptide, is a hormone and neurotransmitter secreted by the gastrointestinal system after a meal. GLP-1 sends signals to the NTS via the vagal nerve (neuronal) and to the organs via the bloodstream (hormonal). This neuropeptide produces satiety, regulates appetite, increases insulin release, decreases glucagon, and decreases gastric motility and emptying (slowing digestion and release of glucose).
Central and peripheral GLP-1 systems independently suppress appetite
Endogenous GLP-1 is produced by both endocrine cells in the gut and preproglucagon (PPG) neurons in the NTS (Brierley, 2021).
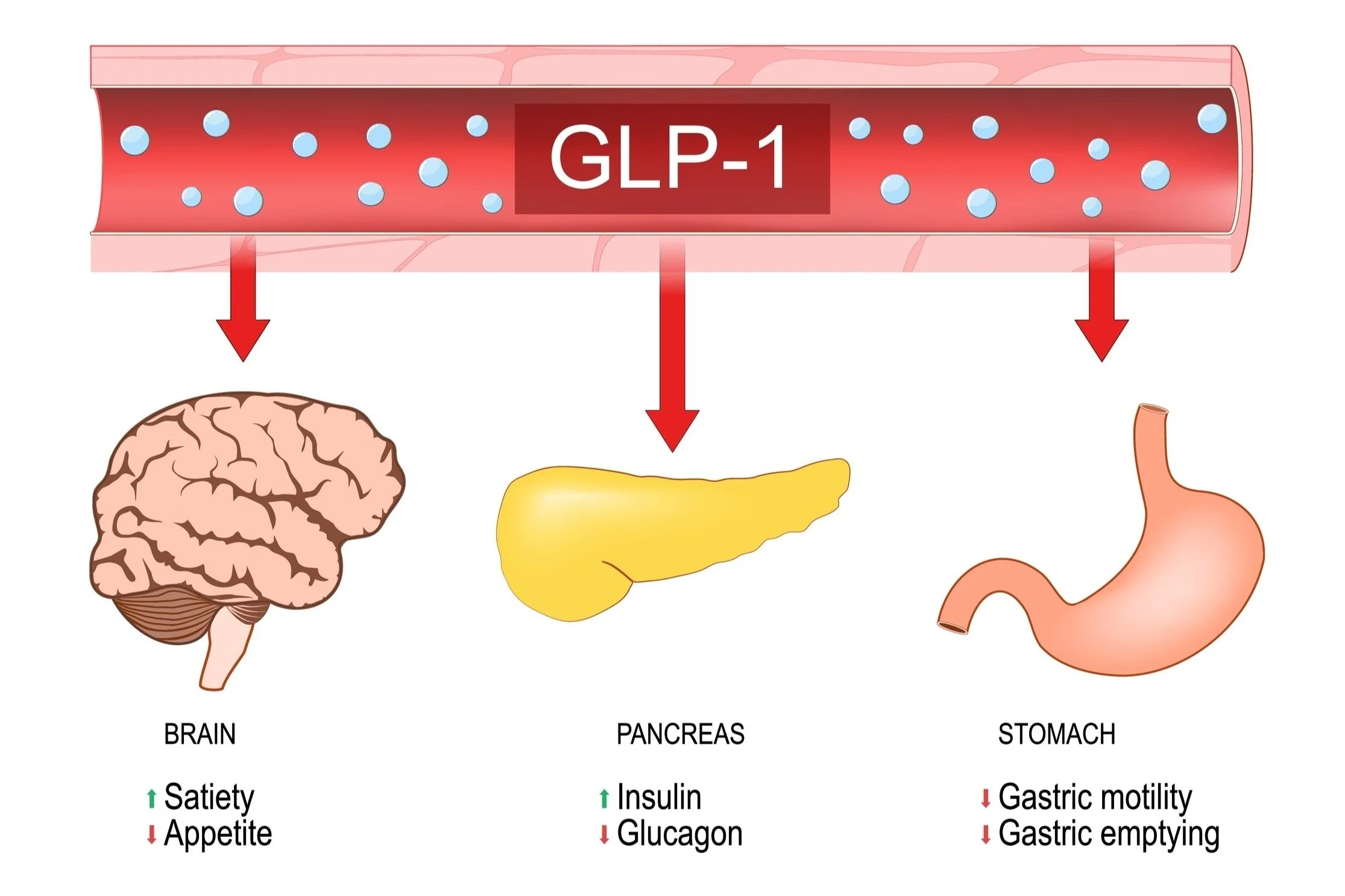
Hormonal pathway (bloodstream): The gut produces GLP-1, which targets various organs (e.g., brain, pancreas, and stomach).
2.6.6
Dorsal Vagal Complex and Weight Control
Ghrelin vs. GLP-1 in Humans
The gastrointestinal tract transmits feeding regulatory signals to the brain via neural and hormonal pathways. Two competing peptides, the orexigenic gastric peptide ghrelin, and the anorectic gut peptide GLP-1, signal to the NTS via the vagus nerve. Blood levels of ghrelin rise 1–2 hours before the start of a meal and fall 30 minutes after the end. Ghrelin increases gastric motility and gastric emptying. Blood levels of GLP-1 peak 30 minutes after a meal, delaying gastric emptying. The administration of a ghrelin 30' before GLP-1 abolishes the effect of the second and vice versa. In the DVMN, we find two distinct populations of neurons: those that increase gastric motility and emptying and those that decrease it. In this way, the DVMN is the stomach's master and consequently significantly influences glycemia. Slowing down gastric traffic allows for a much smoother rise in blood sugar after a meal (postprandial glycemia), which benefits weight and diabetes control.
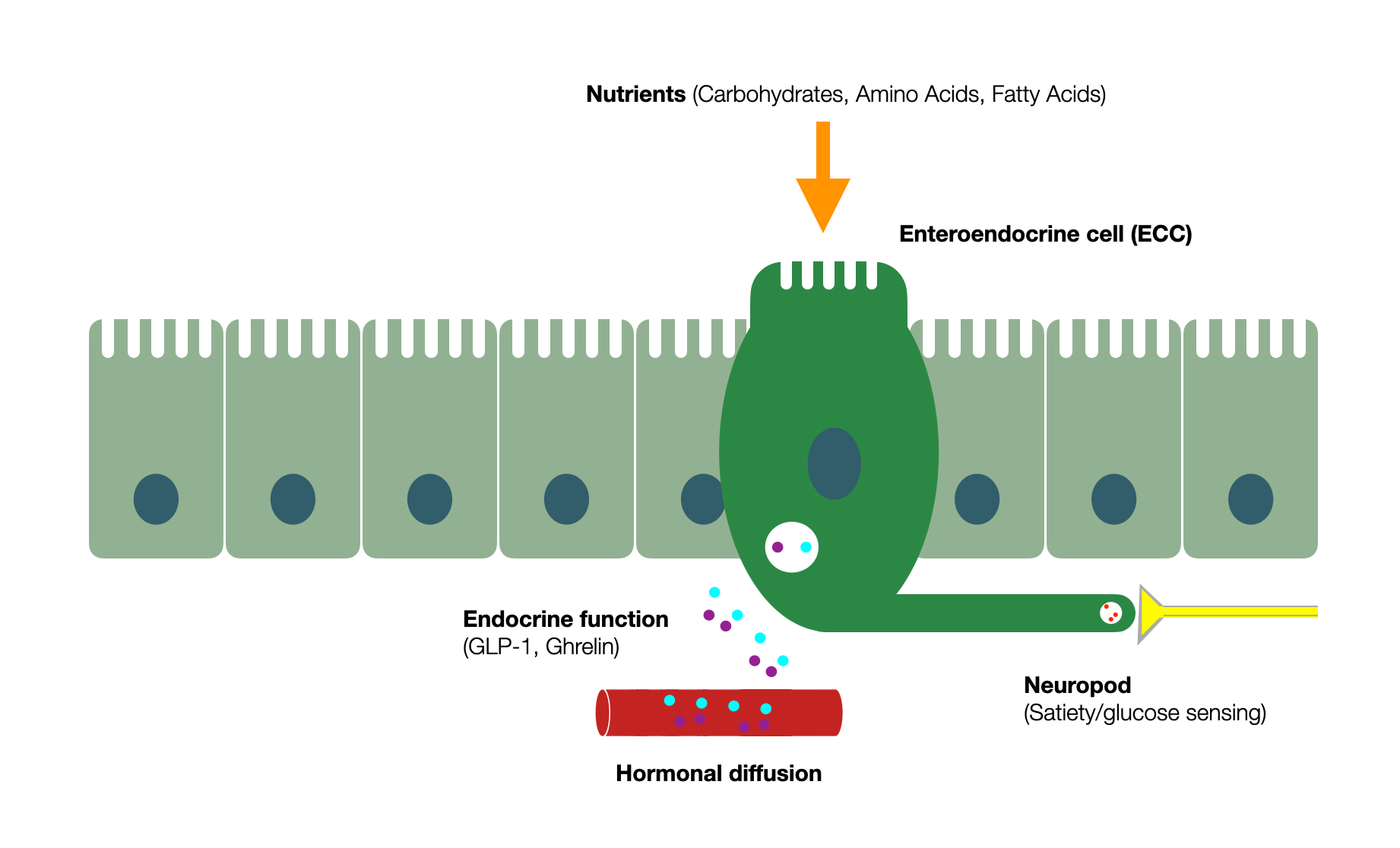
Diagram adapted from Enteroendocrine cell regulation of the gut-brain axis (Barton, 2023)(@absb, 2024).
2.6.7
Dorsal Vagal Complex and Interoception
Our guts constantly communicate with our brains; two-way surveillance is called “interoception.” While some see interoception as “the perception of afferent information from anywhere in the body” (Sugawara, 2024), others limit this term to visceral sensory information. The almost unconscious perception of our internal states manifests itself in our behavior, mood, activity level, and motivation (Büttiker, 2021). The primary purpose of interoception is to maintain homeostasis.
Whether James and Lange or Sherington introduced this concept a hundred years ago is debatable. Craig, in Interoception: the sense of the physiological condition of the body (2003), describes an interoceptive vs. exteroceptive (cutaneous and proprioceptive) system—the latter controlling somatic motor activity.
Primates have a unique brain system that monitors the body's internal state, separate from the system that senses external stimuli such as touch and movement (proprioception). This internal monitoring system in the dorsal posterior insula detects detailed body sensations such as pain, temperature, itch, sensual touch, muscle and organ sensations, blood flow, hunger, thirst, and need for air. In humans, the right anterior insula processes this information to create our sense of self and emotional awareness.
(Craig, 2003). Interoceptive training impacts the neural circuit of the anterior insula cortex (Sugawara, 2024)
Somatic experiencing: using interoception and proprioception as core elements of trauma therapy (Payne, Levine, Crane-Godreau, 2015). In collective publication: Interoception, contemplative practice, and health.
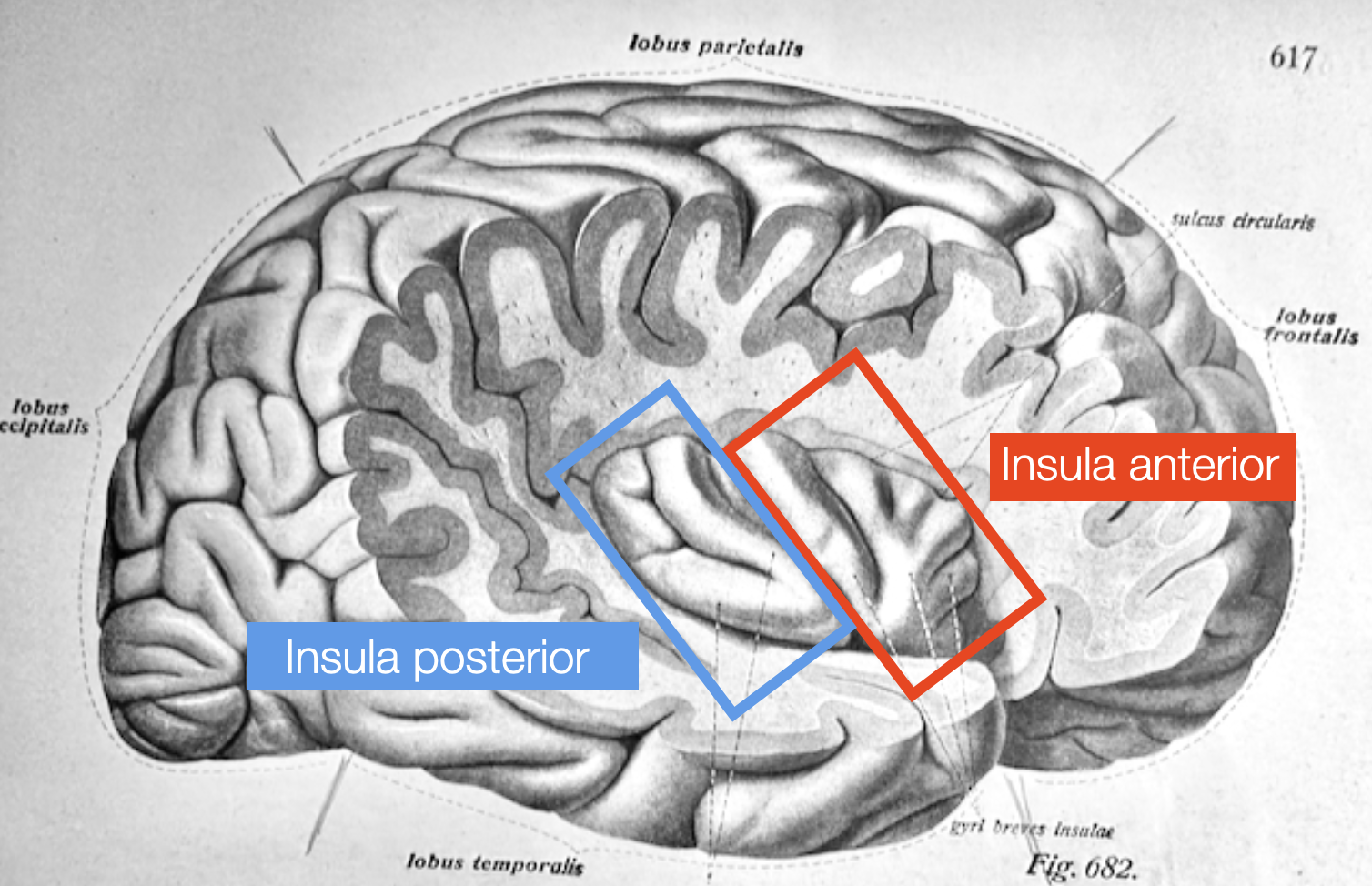
Anatomical view of the insula, after partial removing of temporal and frontal lobes. Adapted from (Sobotta, 1922)(@absb, 2024).
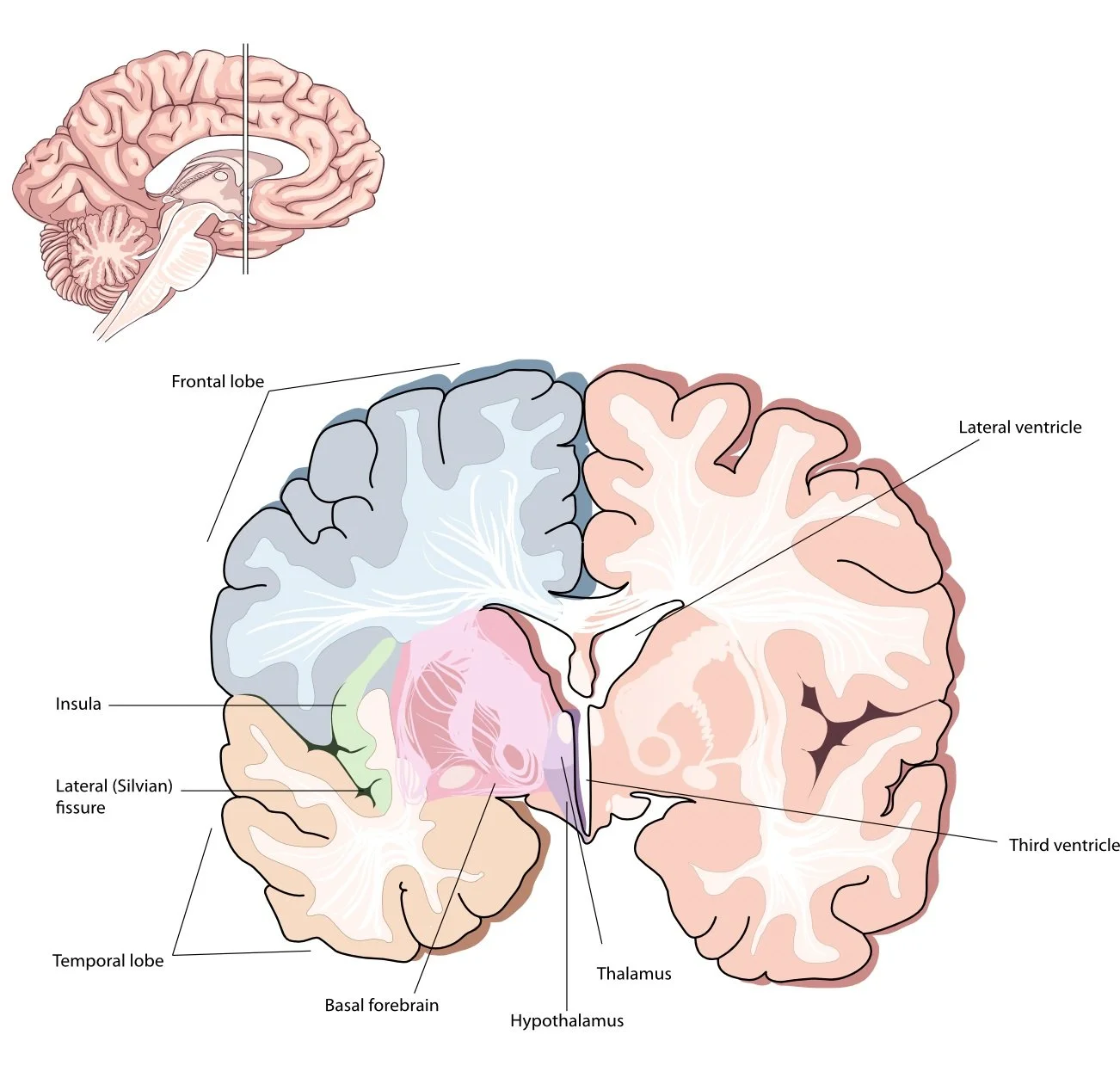
This is another view showing the insula deep hidden behind the temporal and frontal lobes.
2.6.8
The Dorsal Vagal Motor Nucleus (DVMN) and the Heart
This Dorsal Vagal Motor Nucleus (DVMN) is one of the two sources of vagal parasympathetic output. While the DVMN primarily controls cardiovascular activities in non-mammals, cardiac vagal neurons have gradually migrated from a dorsal to a ventral position adjacent to the nucleus ambiguus — the external formation (NAext). In addition, surrounding glial cells (Schwann) create a thick insulation layer of myelin, which allows for faster transmission of signals (B-fibers). Non-myelinated parasympathetic fibers originating from the DVMN have slower conduction (C-fibers). However, Booth (2021), working with larger mammals — sheep instead of mice and rats — was able to identify DVMN efferent fibers with a B-fiber velocity. The DVMN targets smooth muscles, glands, and the heart's ventricles. Its primary effect is to decrease the force of contraction (negative inotropy) and the excitability of the heart muscle.
In Polyvagal Perspectives (2024) writes that “recent studies document the important influences of cardioinhibitory inotropic vagal fibers originating from the dorsal motor nucleus of the vagus” referring to Gourine (2016), Machhada (2015, 2016, 2017, and 2020). “These studies describe the protective function of these pathways in the calm state as well as interactions with sympathetic inotropic influences. In addition, the literature documents experimental procedures during which the inotropic impact of the vagus, the reduction of contractility, occurs independent of changes in heart rate.” Later: “However, there is another literature contradicting the positive attributes of the vagus and linking vagal mechanisms to life-threatening responses, such as bradycardia (and potentially hypotension through diminished contractility) that could lead to sudden neurogenic death (e.g., Richter, 1957).”
Fact-checking
Checking the sources shows that Porges incorrectly cited the studies.
In On the Phenomenon of Sudden Death in Animals and Man, Richter describes how trimming away the whiskers and facial hair leads to premature death–“destroying possibly their most important means of contact with the outside world, seemed disturbing enough, especially to wild rats, to cause their deaths.” Richter adds the restraint in the hand and the confinement in the glass jar as essential factors. Later “the EKG records indicated that the rats succumbing promptly died with a slowing of the heart rate rather than with an acceleration.” But Richter goes on and explains the death by “hopelessness.” “After elimination of the hopelessness, the rats do not die.” Richter explicitly pleads for a psychological instead of a physiological cause. However, Richter concludes that sudden death “occurs apparently as a result of hopelessness; this seems to involve overactivity primarily of the parasympathetic system.”
Richter didn’t mention hypotension or diminished contractility. As he says, the autopsy showed a “large heart distended with blood,” which is expected in a cardiac failure. Richter didn’t evoke a “neurogenic death” either. One more time, Porges wants readers to believe that the DVMN is responsible for slowing the heartbeat and causing a “sudden neurogenic death.” This is polyvagal vocabulary, not the words of Richter.
Porges (2024) quotes Machhada (2016) and Gourine (2016), suggesting that the decrease of contractility of the left heart ventricle under the influence of the DVMN may be fatal. Isn’t it the last effort to save the myth of the lethal dorsal vagal?
Previous studies mentioned in the article (Gourine, 2016) used electrical stimulation to decrease ventricular contractility in animals under anesthesia. New techniques offer different results.
In Cardiac Vagus and Exercise, Gourine (2019) concludes that “vagal stimulation enhances cardiac contractility and prolongs exercise endurance.”
In Control of ventricular excitability by neurons of the dorsal motor nucleus of the vagus nerve (Machhada, 2015), the authors conclude that the “activity of the DVMN vagal preganglionic neurons is responsible for tonic parasympathetic control of ventricular excitability, likely to be mediated by nitric oxide.” The DVMN protects the heart against ventricular fibrillation (Brack, 2011) through a NO (nitric oxide) mechanism. It has antiarrhythmic effects on the heart (Machhada, 2015) (Ng, 2014) and decreases the contractility of the left ventricle without changing the heart rate (Machhada, 2016). After myocardial infarction, it protects left ventricular myocytes from lethal ischemia/reperfusion injury (Mastitskaya, 2012). In Origins of the vagal drive controlling left ventricular contractility (Machhada, 2016) the authors conclude that the inhibition of the DVMN increases left ventricle contractility.
However, in Vagal determinants of exercise capacity, Machhada, Gourine, et al. (2016) show that elite endurance athletes have exceptionally high parasympathetic vagal tone. Stimulation of the DVMN increases exercise capacity, while silencing the DVMN dramatically impairs exercise capacity. The authors conclude that the DVMN directly increases cardiac contractility of the left ventricle and prolongs exercise endurance.
In Vagal determinants of exercise capacity (Machhada, 2017) and Optogenetic Stimulation of Vagal Efferent Activity Preserves Left Ventricular Function in Experimental Heart Failure (Machhada, 2020), the authors come to the same conclusion: “In rats, genetic silencing of the largest population of brainstem vagal preganglionic neurons residing in the brainstem’s dorsal vagal motor nucleus dramatically impairs exercise capacity, while optogenetic recruitment of the same neuronal population enhances cardiac contractility and prolongs exercise endurance.”
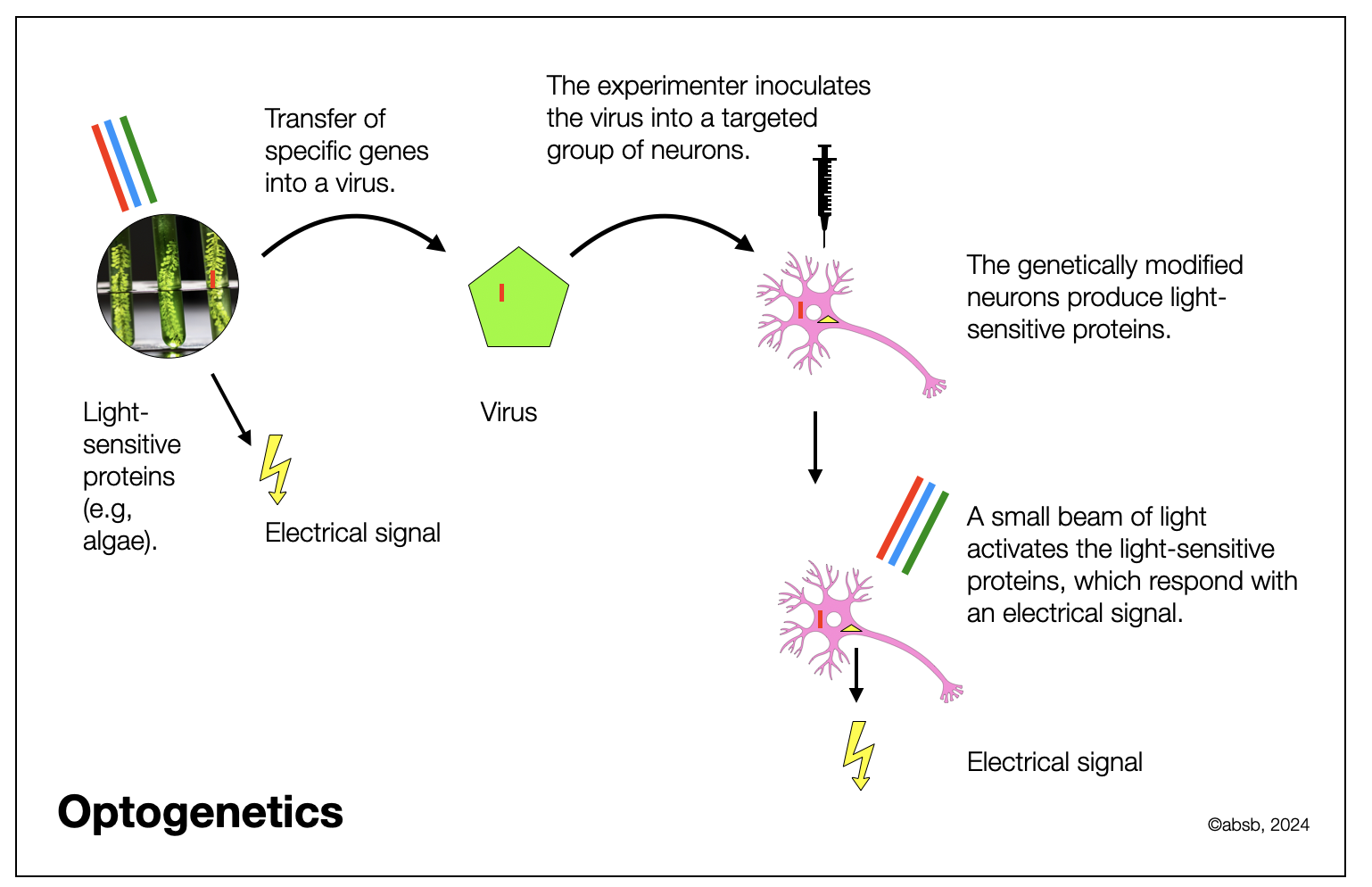
Optogenetics
Optogenetic stimulation is a new technique (see diagram below) that stimulates genetically modified (light-sensitive) cells through light instead of electrical stimulation. Targeting the right group of neurons is much easier, and the stimulation occurs in a waked state (post-anesthesia). Changing the technique may explain the new results: the DVMN may improve rather than decrease ventricular contractility. Why didn’t Porges take those findings into account?
As another consequence, after decades of debate among experts, new research using optogenetics (Machhada, 2015, 2016), (Veerakumar, 2022), and (Jalil, 2023) has demonstrated that the NAext and not the DVMN is the main vagal brake of cardiac rhythm (negative chronotropic) and atrioventricular conduction velocity (dromotropic).
Machhada et al. (2020) recently described other beneficial effects of the DVMN:
Optogenetic stimulation of the DVMN protects left ventricular function in ischemic infarction
In response to dorsal solid vagal stimulation, the authors observed modest bradycardia (8.5%), improved left ventricular (LV) contractile responses to β-adrenoceptor stimulation, and improved exercise capacity in healthy rats.
Optogenetic stimulation of the DVMN for 15 min/two days over four weeks after provoked myocardial infarction preserved left ventricular and exercise capacity. In particular, it protects the heart from so-called “reperfusion injury,” which occurs when blood starts circulating again (perfusion) after removing the blockage. Reperfusion often triggers an intense inflammatory process that gets out of control.
Finally, according to Moreira (2018), long-term activation of the DVMN reduces blood pressure in hypertensive rats.
So, are we done discussing the specific roles of NAext and DVMN? Not quite! A recent study, Dorsal Motor Vagal Neurons Can Elicit Bradycardia and Reduce Anxiety-like Behavior (Strain, 2024), challenges both the polyvagal theory and the orthovagal model. Optogenetic stimulation of the dorsal vagal motor nucleus (DVMN or DMVN) in mice at 20 Hz elicited significant bradycardia, reducing the heart rate by approximately 56% through muscarinic receptors. Chemogenetic stimulation of the DVMN also suppressed heart rate (around 65.5% of resting rate) for up to eight hours. Interestingly, DVMN activation reduced anxiety-like behavior in the mice.
The study further demonstrated that DVMN neurons were significantly more excitable than NAext cardiac neurons, exhibiting spontaneous ongoing activity in ex vivo conditions. The authors attribute the differences in response between studies with cats or rats versus mice to variations in vagal tone, noting that cats and rats do not respond as strongly to stimulation. Additionally, this study's stimulation frequency (20 Hz) may play a role.
In their conclusion, the authors reference the James-Lange theory (James, 1884), which suggests that anxiety does not cause an increase in heart rate; instead, the perception of an increased heart rate is what constitutes anxiety. They also cite a recent study (Hsueh, 2023)—see the chapter on emotions—showing that optogenetically increasing heart rate in mice can induce anxiety-like behaviors in risky situations (context-specific). Further research is needed to determine whether similar results would be observed in larger mammals, such as sheep, following optogenetic DVMN stimulation.
While some of these findings support the polyvagal hypothesis of dorsal bradycardia, other aspects highlight the DVMN's unexpected role as a visceral-emotional pacifier—an intriguing result for polyvagal theory. We are eager to see more studies in this area.
2.6.9
The Orthovagal Model: A Challenge to the Polyvagal Theory
The Polyvagal Theory
The PVT describes three groups separated by history (phylogeny and ontogeny), anatomy, and physiology. The “smart vagal” or ventral vagal complex (VVC) is part of a self-regulatory loop that includes:
Cranial nerves V, VII, IX, X and XI
The heart
Face, neck, and throat regions
Social interaction (from the outside)
The respiratory centers and the digestive system are not shown on this diagram because they do not play a major role in the polyvagal world. This “vagal loop” doesn't include the NTS, the most important visceral hub in the brainstem. How does the respiratory and baroreceptor information get to the NAext if not through the NTS, which is a part of the “dorsal”? There is a cognitive dissonance here.
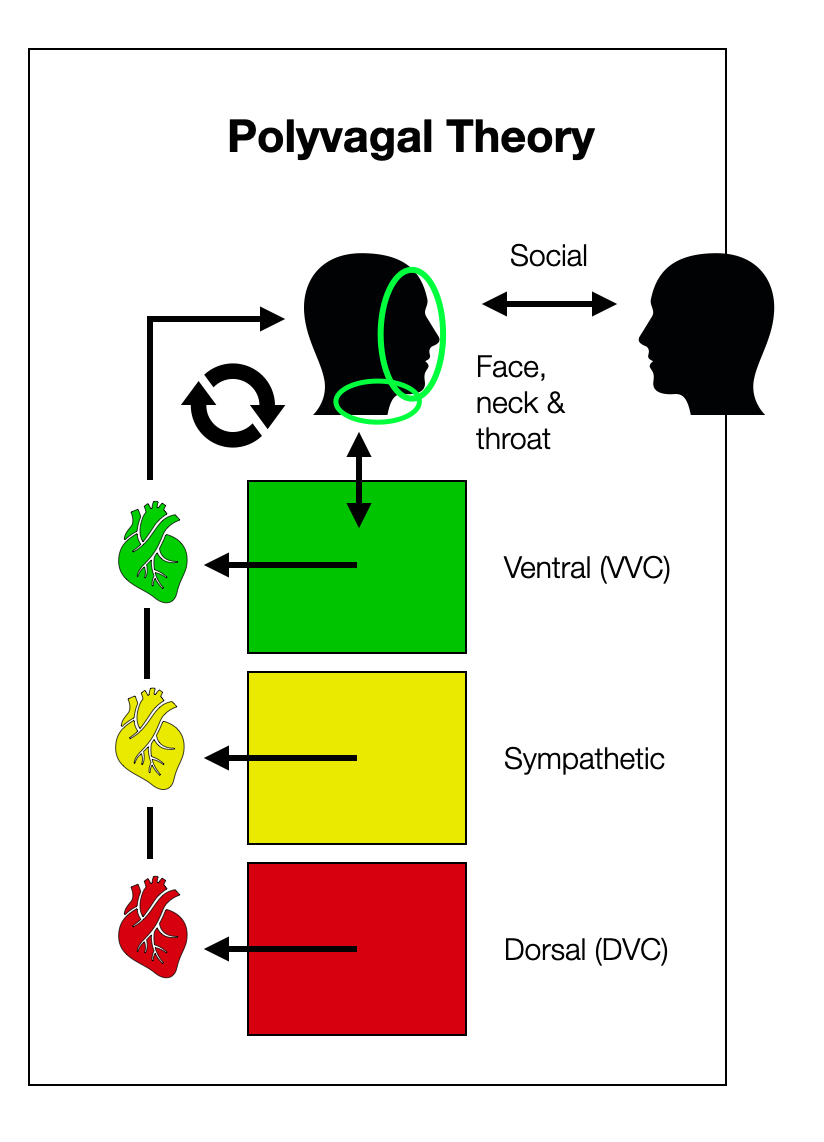
The polyvagal model always has in mind a multi-hierarchical dimension (physiological, psychological, phylogenetic, and ontogenetic), dating the three systems (dorsal, sympathetic, and ventral) according to when they supposedly “punched the evolutionary clock.”
The Orthovagal Model
Recognize the banana-shaped NTS with its four sections — gustatory, respiratory, cardiac, and visceral. No matter the scale — we made the NTS very big! — This diagram shows how the viscera, brainstem centers, and higher centers interact.
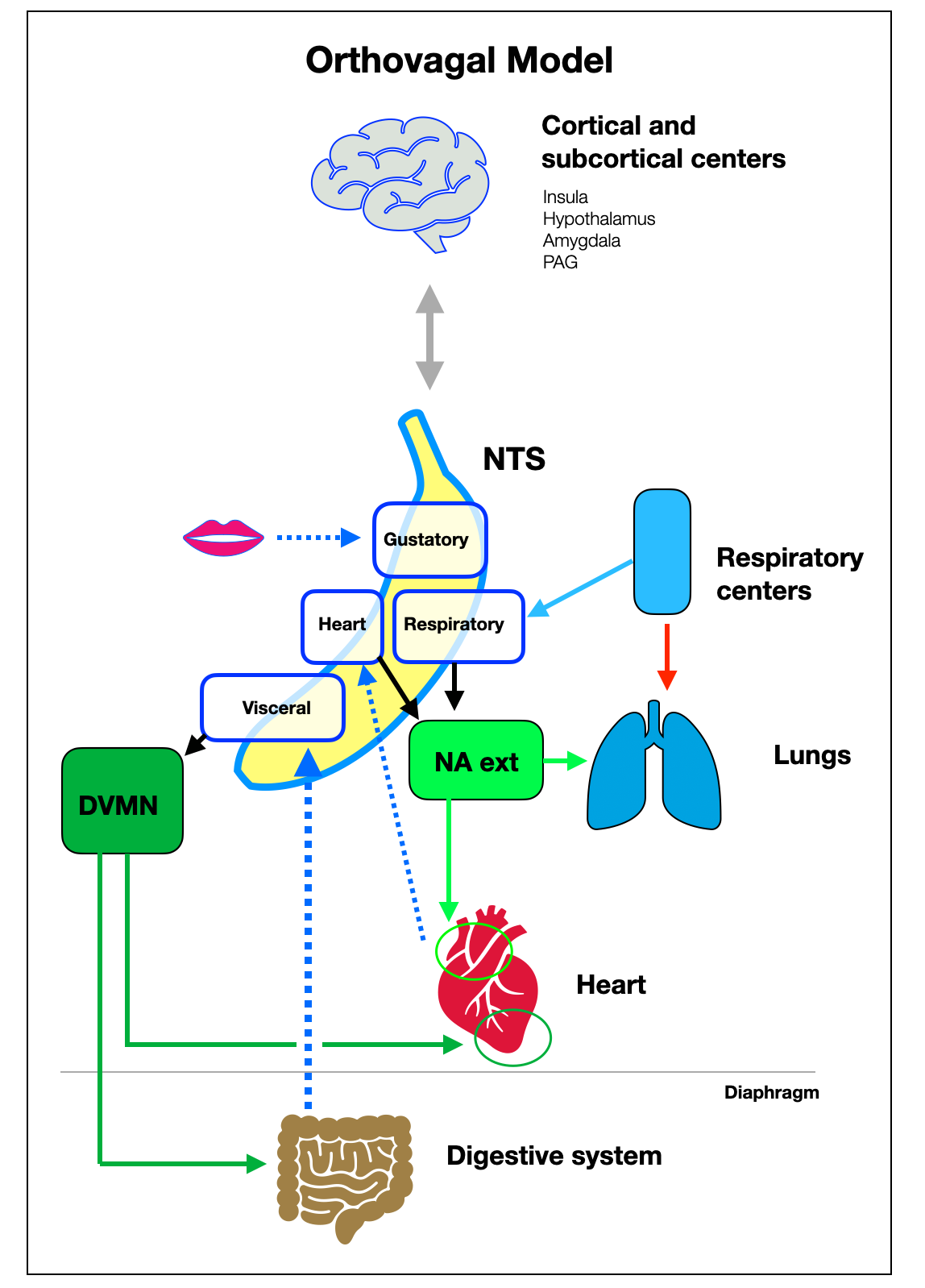
Here, again, are the two main visceral loops:
The digestive loop (Digestive System > NTS > DVMN)
The cardiac loop (baro- and chemoreceptors > NTS > NAext)
We also see how the respiratory and baroreceptors send signals to the NTS, resulting in the variable ventral parasympathetic output (HRV). The DVMN (dorsal parasympathetic) innervates the ventricles (protection + inotropy).

2.6.10
Research about the Dorsal Vagal Motor Nucleus
Acetylcholine muscarinic receptors
Decreased excitability: Control of ventricular excitability by neurons of the dorsal motor nucleus of the vagus nerve (Machhada, 2015)
Decreased contractility: Origins of the vagal drive controlling left ventricular contractility (Machhada, 2016)
General overview
Cardiac vagal preganglionic neurones: An update (Gourine, Machhada, Trapp, Spyer, 2016): Distinct groups of cardiac vagal preganglionic neurons with different patterns of activity control nodal tissue (controlling the heart rate and atrioventricular conductance) and the ventricular myocardium (modulating its contractility and excitability).

Nitric oxide (NO)
Direct evidence of nitric oxide release from neuronal nitric oxide synthase activation in the left ventricle as a result of cervical vagus nerve stimulation (Brack, Patel, Mantravardi, Coote, Ng, 2009)
Vagus nerve stimulation protects against ventricular fibrillation independent of muscarinic receptor activation (Brack, Coote, Ng, 2011)
Vagal modulation of cardiac ventricular arrhythmia (Ng, 2014)
Protection of the heart in acute ischemia /reperfusion injury:
Nitric oxide mediates the vagal protective effect on ventricular fibrillation via effects on action potential duration restitution in the rabbit heart (Brack, Patel, Coote, Ng, 2007)Cardioprotection evoked by remote ischaemic preconditioning is critically dependent on the activity of vagal pre-ganglionic neurons (Mastitkskaya, 2012)
2.6.11
The Dorsal Vagal Motor Nucleus and the Immune System

The DVMN regulates metabolic homeostasis by controlling heart rate, gastrointestinal motility and secretion, pancreatic secretion, hepatic glucose production, and other visceral functions. It is also an important component of a neural reflex the inflammatory reflex that controls innate immune responses and inflammation during pathogen invasion and tissue injury (such as ischemic heart failure) (Pavlov, 2012). The Vagus Nerve in the Neuro-immune Axis: implications in the Pathology of the Gastrointestinal Tract (Bonaz, 2017) describes how the sympathetic nervous system and the vagal nerve work in synergy to inhibit the release of tumor necrosis factor-alpha (TNFα). Imagine a bomb hitting a house during a war, creating a certain radius of destruction. Sappers rush to the scene with picks and shovels to clear the rubble. Maybe they knock down a few walls that are in danger of collapsing. But if they go too far, they risk destroying other buildings that are still intact. Similarly, in the body, after tissue destruction (injury, cancer, or heart attack), the cleanup cells (macrophages) can exaggerate and create huge zones of destruction. Inflammation is essential for effective antimicrobial defense and response to tissue injury, but the process must be regulated and resolved to avoid excessive tissue damage and chronic inflammation.
Following a viral infection (e.g., Pfeiffer's disease, COVID-19, or influenza), the immune system can maintain chronic inflammatory processes, leading to debilitating conditions such as long COVID or long influenza. Autoimmune diseases (such as multiple sclerosis, irritable bowel disease, or rheumatoid arthritis) are also the result of an immune system overreaction. The DMVN and the sympathetic nervous system can help fight chronic inflammation.
In a recent study, Caravaca et al (2022) show how vagal electrical signals accelerate the resolution of inflammation. Current research's novelty is identifying the pro-versus anti-inflammatory molecular processes with increasing precision and how to target them with precision.
Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin (Borovikova, 2000).
Vagus nerve stimulation enhances the cholinergic anti-inflammatory pathway to reduce lung injury in acute respiratory distress syndrome via STAT3
(Li, 2021). Acute respiratory distress syndrome (ARDS) is characterized by acute, diffuse, and inflammatory lung injury. Numerous studies have shown that the cholinergic anti-inflammatory pathway (CAIP) is critical in regulating ARDS-associated lung injury. The mechanism of action of the CAIP involves the afferent transmission of inflammatory stimuli to the nucleus solitarius (NTS). This results in the excitation of the dorsal motor vagal nucleus (DMVN), which in turn causes the release of acetylcholine.
Consequently, acetylcholine binds to the acetylcholine receptor, α7nAchR, which plays a critical antagonistic role against inflammation. Vagus nerve stimulation (VNS) is a relatively mature method of activating CAIP by promoting the release of acetylcholine, which acts on α7nAchR. Recent studies have shown that α7nAchR can inhibit the secretion of pro-inflammatory cytokines and chemokines in inflammatory cells and reduce lung inflammation and injury.
Insular cortex neurons encode and retrieve specific immune responses (Koren, 2021) and send the information through the autonomic system (DMVN) and sympathetic RVLM (medulla).
Your brain could be controlling how sick you get — And how you recover (Kwon, 2023) explains how cutting the vagus nerve makes the anti-inflammatory effect of the drug administered to the brain disappear.
2.6.12
Dorsal Vagal Motor Nucleus and Metabolism

In humans and rodents, GLP-1 levels are reduced in obesity. GLP-1 is also produced centrally by neurons in the caudal brainstem. Circulating levels of GLP-1 are low under fasting conditions and increase threefold to fourfold in response to a meal. Nutrient sensing in the gut is performed by specialized enteroendocrine cells that are sparsely distributed in the gastrointestinal epithelium. L-cells are enteroendocrine cells that account for 95% of circulating GLP-1. Intraperitoneal injection of GLP-1 has been shown to promote satiety, inhibit gastric emptying, and improve glucose tolerance. Intestinal GLP-1 exerts its effects primarily through vagal sensory afferents, providing a direct link between the gut and the brain. These sensory neurons are the stomach's mechanoreceptors (pressure sensitive) and chemoreceptors (nutrient sensitive) in the intestine. The need for an intact vagus nerve to mediate all the effects of GLP-1 has been demonstrated in human subjects who have undergone vagotomy.
See also Insulin Signals Through the Dorsal Vagal Complex to Regulate Energy Balance (Filippi, 2014) and (Drucker, 2022) (Kabahizi, 2022) (Kanoski, 2016) (Ludwig, 2021) on the involvement of the DVC in the satiety system.
In summary, all of this recently gathered information regarding fat and glucose metabolism doesn't fit the polyvagal description of the DVC — another confirmation of the hallucinatory nature of the PVT.
Metabolism and Obesity
In 2021, semaglutide (marketed as Wegovy) became the first once-weekly medication in its class to be approved by the FDA for chronic weight management. According to social media, Oprah Winfrey, Elon Musk, and Amy Schumer have taken Ozempic, another semaglutide. Semaglutide is a glucagon-like peptide-1 (GLP-1) receptor agonist. It works by increasing the release of insulin from the pancreas after a meal and decreasing the release of glucagon, a hormone that raises blood sugar. Semaglutide helps control blood glucose levels, leading to weight loss. In the largest clinical trial, adults taking semaglutide lost an average of nearly 15% of their initial body weight.
In addition to its effect on blood sugar, GLP-1 affects weight through two mechanisms:
Acting on the hunger centers in the brain (hypothalamus and medulla) to reduce hunger, appetite, and cravings.
By slowing the gastric emptying rate, it prolongs fullness and satiety after eating.
Why do we need external stimulation of GLP-1 receptors? Couldn't our bodies do it themselves?
Afferent vagal sensory pathways (to the NTS) and specialized centers in the NTS and hypothalamus are part of the satiety system that sets limits on food intake. According to Haspula (2023), “the DVC, located in the hindbrain, is referred to as the brainstem satiety center. “ The role of the vagus nerve in GLP-1-mediated regulation of eating (Brierley, 2022) describes how glucagon-like peptide 1 (GLP-1), a gastrointestinal hormone and centrally produced neuromodulator, has received particular attention as a gut-brain signaling molecule.
2.6.13
The Area Postrema
This small structure is located just below the fourth ventricle in the brainstem. It monitors blood and cerebrospinal fluid, assesses signs of intoxication, and initiates nausea and vomiting with the NTS, DVMN, and NA. After relaying information to the NTS, distinct neural outputs are sent to coordinate vomiting responses, including swallowing, salivation, cardiovascular, respiratory, and gastrointestinal, in a structured manner. Some of these outputs include diaphragm contraction via abdominal muscles, external intercostal muscles while internal intercostal muscles relax, esophageal contraction, lower esophageal sphincter relaxation, gastric dysrhythmia, and duodenal retroperistaltic activity in the duodenum and stomach (Price, 2008).

This cross-section of the spinal cord shows the location of the area postrema. In humans, this is a paired structure. In mice, the AP is a block, which can be confusing when looking at different diagrams of the mammalian AP. The DVMN and NTS are below the AP (©absb, 2024).
2.6.14
Dorsal Vagal Complex and Depression
Recognizing the role of the vagus nerve in depression from microbiota-gut-brain axis (Tan, 2022). In this study, the authors explore depression from a broad perspective, linking the microbiome with the autonomic nervous system and immune system. The afferent pathways (80% of the vagus nerve) that synapse in the NTS (nucleus tracts solitarius) inform the brain about the state of the gut. Descending fibers originating in the DVMN (dorsal vagal motor nucleus) are anti-inflammatory and protect the gut. In depression, vagal tone is reduced, and the gut microbiome is disrupted. However, it is not clear which parts of the vagal nerve are involved.
Polyvagalists keep writing that activating the DVC causes depression. They ignore the current state of science on this topic. Depression is related to serotoninergic and adrenergic mechanisms. It doesn't mean that the amount of serotonin in the gut or blood is relevant, as some believe. Serotonin doesn't cross the blood-brain barrier; only serotonin precursors do. Noradrenaline (norepinephrine) produced in the locus coeruleus (brainstem) has multiple functions: concentration, learning, motivation, and more. Modern antidepressants enhance both synaptic neurotransmitters.
2.6.15
Dorsal Vagal Complex and Polyvagal Theory
As seen above, neither the NTS nor the DVMN deserves to be collectively described as “old mammalian,” “primitive,” or “vestigial." Individually, both centers have stimulated brand-new discussions and research on body weight regulation, the immune system, and brain-gut interactions. Thanks to modern technology, researchers can explore these areas with more power than they could thirty years ago.
Without dwelling too long on the polyvagal argument, let us briefly consider some of Porges' assertions over the last three decades. In the introduction to Clinical applications of the polyvagal theory: the emergence of polyvagal-informed therapies (Porges, 2018, p. XXi), Porges states about the DVC: “The shutdown system, when activated in mammals, leads to fainting, defecation, and, as I would later learn, dissociation.”
Let us examine these few statements:
In The Pocket Guide to the Polyvagal Theory (2017, p. 10-11), Porges begins his book with a few definitions, which we will quote at length because they summarize the core of PVT.
Death feigning/shutdown system.
In mammals, under certain conditions, the nervous system reverts to a primitive defense response characterized by appearing to be inanimate.
This defense pattern is frequently observed in vertebrates, such as reptiles and amphibians, that evolved before the phylogenetic emergence of mammals.
However, mammals are great consumers of oxygen, and the immobilization required in feigning death is associated with a decrease in the capacity to oxygenate the blood and an inability to deliver sufficient oxygenated blood to the brain to support consciousness.
This massive depression of autonomic function is due to activation of the dorsal vagal circuit, which depresses respiration (apnea) and slows heart rate (bradycardia) (…)
The nervous system may revert to the ancient immobilization defense system (…) life threat that would include features of death feigning such as fainting (vasovagal syncope), defecation, and dissociation.
Let's analyze these four lines:
Death feigning, also called thanatosis (Greek: Thanatos = death), is a deceptive behavior used by all kinds of animals, from beetles to snakes to opossums to mice. It is not specific to mammals or non-mammals. The purpose is to simulate death, since many predators don't eat dead prey. Rapid, shallow breathing — not apnea — characterizes this behavior. Animals that can't escape (e.g., a mouse in a cage with a cat) may show high or low cardiovascular tone, depending on the species (Hofer, 1970).
Regarding immobilization, Porges confuses the somatic and visceral domains: immobilization is a behavior of the striated muscles (somatic), and bradycardia belongs to the autonomic domain. Both are controlled by the ventral lateral PAG (periaqueductal gray matter) of the midbrain.
The DVMN doesn't affect breathing. The DRG (dorsal respiratory group) in the NTS plays a modest respiratory role as part of a large complex of brainstem nuclei. Finally, the NTS and some respiratory centers (pre-Bötziger) directly inform the ventral parasympathetic (NAext), which is integrated into an autonomic loop.
Fainting. A sudden drop in arterial pressure generally causes fainting after a sudden change in position (orthostatic) or when a person experiences a stressful event. Examples include seeing blood, emotional stress, pain, and physical or emotional trauma. The stressful event stimulates a body reflex called the vasovagal response. The heart slows down, pumps less blood, and blood pressure drops. Then, the brain doesn't get enough oxygenated blood, and the person passes out. However, in PVT there is no place for the positive role of the sympathetic centers (RVML and CVLM (Karemaker, 2022)) that regulate blood pressure in the medulla. They are affected by a change of position or the slightest physical exercise, with or without threat. Fainting is a vasodepressor (sympathetic) phenomenon rather than a vagal response. In the hypothetical case of fainting due to bradycardia, the nucleus ambiguus (NAext) and not the DVMN — the unmyelinated parasympathetic — would slow the heart rate.
In summary, Porges and his followers have invented a negative entity called “the Dorsal” and given it the characteristics of a person (“personification”). Many lyrical fictional descriptions are made, and role-playing (Dana) is staged. This may be entertaining, but it has nothing to do with the anatomical and physiological reality of the brain stem.
Allan N. Schore, in The Effects of Early Relational Trauma on Right Brain Development and Infant Mental Health (2001), suggests the negative role of the dorsal vagal many times. In 2021, he again cites Porges ): «I suggest that the early postnatal stage of human development is a critical period for a transition from subcortical to cortical face processing systems and from the dorsal vagal to the experience-dependent maturation of the right-lateralized ventral vagal social engagement system of Porges (2011).»
Fear and the Defense Cascade (Kozlowska, 2015) extensively quotes Porges as an expert. It presents the term “anxiety bradycardia” as caused by the DVMN. It wrongly quotes Hofer (1970), who doesn't use the term “dorsal” vagal action” and remains cautious in explaining arrhythmias that are “thought to be parasympathetic in etiology.” In the coming decades, much research will be done on the molecular mechanisms of arrhythmias. The DVMN protects against them.
What does “Older Vagal Shutdown” mean?
In Love’s Brain: A Conversation with Stephen Porges (Loizzo, 2018), Porges uses the expression «older vagal shutdown response that drives the faint or freeze reflex»: a repetitive affirmation – ad nauseam.
Our comment:
To call the DVC “old” is incorrect. The DVC, like the nucleus ambiguus (NA), the amygdala, and many other brain structures, is “conserved.” Would you call your car old because it still has four wheels, front brakes, rear brakes, and a driving wheel, similar to the Ford Model T produced by the Motor Ford Company on October 1, 1908? Moving the respiratory-cardiac function to a ventral position doesn't make the DVC older: having so many essential functions already (e.g., metabolic regulation, cardio-protection, and immune regulation) justifies clever outsourcing. Moreover, the NTS (sensory part of the DVC) and the ventral nucleus ambiguus (external formation) are connected by direct reciprocal connections.
“Shutting down” is not a function of the DVC. On the contrary, the DVC protects the heart, regulates digestive and metabolic processes (up and down), and provides the anti-inflammatory reflex. A shutdown — meaning a deactivation of the sympathetic system — is commanded by higher neural centers. As seen elsewhere, the trigeminal cardiac reflex can be fatal after stimulation of the face with cold water or surgical intervention on the face, in which case the NAext is the source of the cardiac slowing, not the DVC.
“Fainting” is also not caused by the DVC. It is mainly caused by a drop in arterial pressure (a drop in sympathetic tone) and not by a slowing of the heartbeat (bradycardia). Several centers are involved in the medulla and higher (Guyenet, 2006).
The “freeze reflex” is a motor behavior — not an autonomic one — activated primarily at the vlPAG level (midbrain). It is a brief or prolonged cessation of movement that can be externally observed (Roseberry, 2017).
2.5.16
More Facts about the Dorsal Vagal Complex
Vasovagal syncope. Good literature on this subject is scarce. Neural Circulatory Control in Vasovagal Syncope (Van Lieshout, 1997)states: “In most of our cases, a faint is preceded by increasing heart rate but decreasing blood pressure, due to decreasing return of blood to the ventricles.” Sympathetic withdrawal may also be the predominant phenomenon. In psychogenic fainting, muscle contraction may optimize venous return. This supports the hypothesis of volumetric depression.
Defecation. The autonomic nervous system can activate urgent defecation, but not through the vagal nerve, which ceases to innervate the colon from the middle of the transverse (horizontal) colon. Whether such a rapid reaction — e.g., in the case of hanging — is sympathetic or parasympathetic remains to be seen.
Every year in the USA, 1000–1500 babies die from aspiration of meconium, a dark substance expelled from the rectum into the amniotic fluid. In utero, however, defecation is a normal function (Ramón y Cajal, 2005). Therefore, researchers prefer another explanation, as presented in Meconium Aspiration Syndrome: A Role for Fetal Systemic Inflammation (Lee, 2016).
Dissociation. The PVT conflates immobility, dissociation, and shutdown. With the development of psychotraumatology, various authors (e.g., Fanselow, Niejenhuis, or Levine) have begun to study animal models to better understand human responses to threat. While these models are partially applicable, they largely underestimate the flexibility of the human neocortex. Dissociation shouldn't be confused with immobility, confusion, or blurring. As seen in The Haunted Self: Structural Dissociation and the Treatment of Chronic traumatization (van der Hart, 2006), humans can adopt a detached mind and simulate normality (displaying an “apparently normal part of the personality” or ANP). This normality is the psychological camouflage that victims need to survive. Human dissociation is not necessarily associated with immobility but rather with detachment (depersonalization and derealization) or dissociative identity disorder. The different parts of the personality may exhibit different behaviors or energy states (e.g., regulated, aggressive, or submissive) without reference to a specific phylogenetic level.
>> to the next chapter Sympathetic